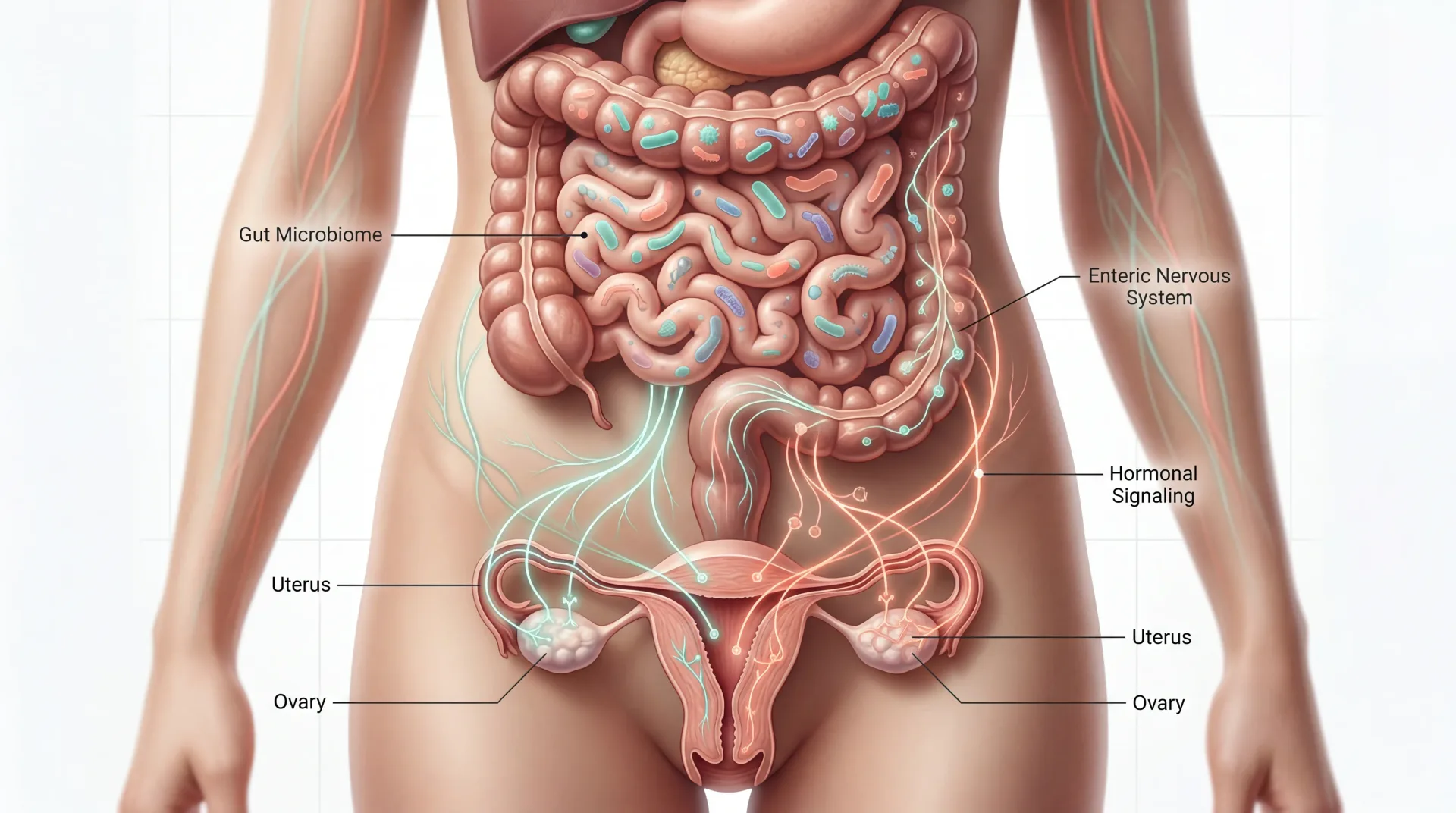
Roughly 70% of Your Immune System Lives in Your Gut — and It Talks to Your Ovaries
Most people think of gut bacteria as helpers that break down food and keep digestion on track. That picture is incomplete. The trillions of microorganisms lining your intestinal walls form what researchers now describe as a full-fledged endocrine organ, one that manufactures hormones, shapes immune responses, and sends chemical signals to tissues far removed from the digestive tract — including the uterus, ovaries, and fallopian tubes.
That is not a metaphor. Your gut bacteria produce short-chain fatty acids (SCFAs) like butyrate, propionate, and acetate through the fermentation of dietary fiber. These metabolites do far more than fuel intestinal cells. They regulate the activity of immune cells called regulatory T cells (Tregs), which are the same cells that must establish tolerance at the uterine lining for an embryo to implant successfully. When the microbial community producing those metabolites shifts out of balance — a state called dysbiosis — the downstream effects ripple into hormonal regulation, inflammation control, and reproductive tissue integrity.
Research now connects gut dysbiosis to three of the most common reproductive health challenges women face: polycystic ovary syndrome (PCOS), endometriosis, and unexplained infertility. The relationship is not one-directional. Hormonal shifts during the menstrual cycle, pregnancy, and perimenopause reshape the gut microbiome just as the gut microbiome reshapes hormone levels. That two-way conversation points toward interventions that conventional gynecological care has mostly ignored.
Key takeaway: Your gut microbiome functions as an endocrine organ that directly influences estrogen metabolism, androgen levels, insulin signaling, and immune tolerance — all of which govern reproductive health outcomes.
The Hidden Hormone Recycling Plant Inside Your Intestines
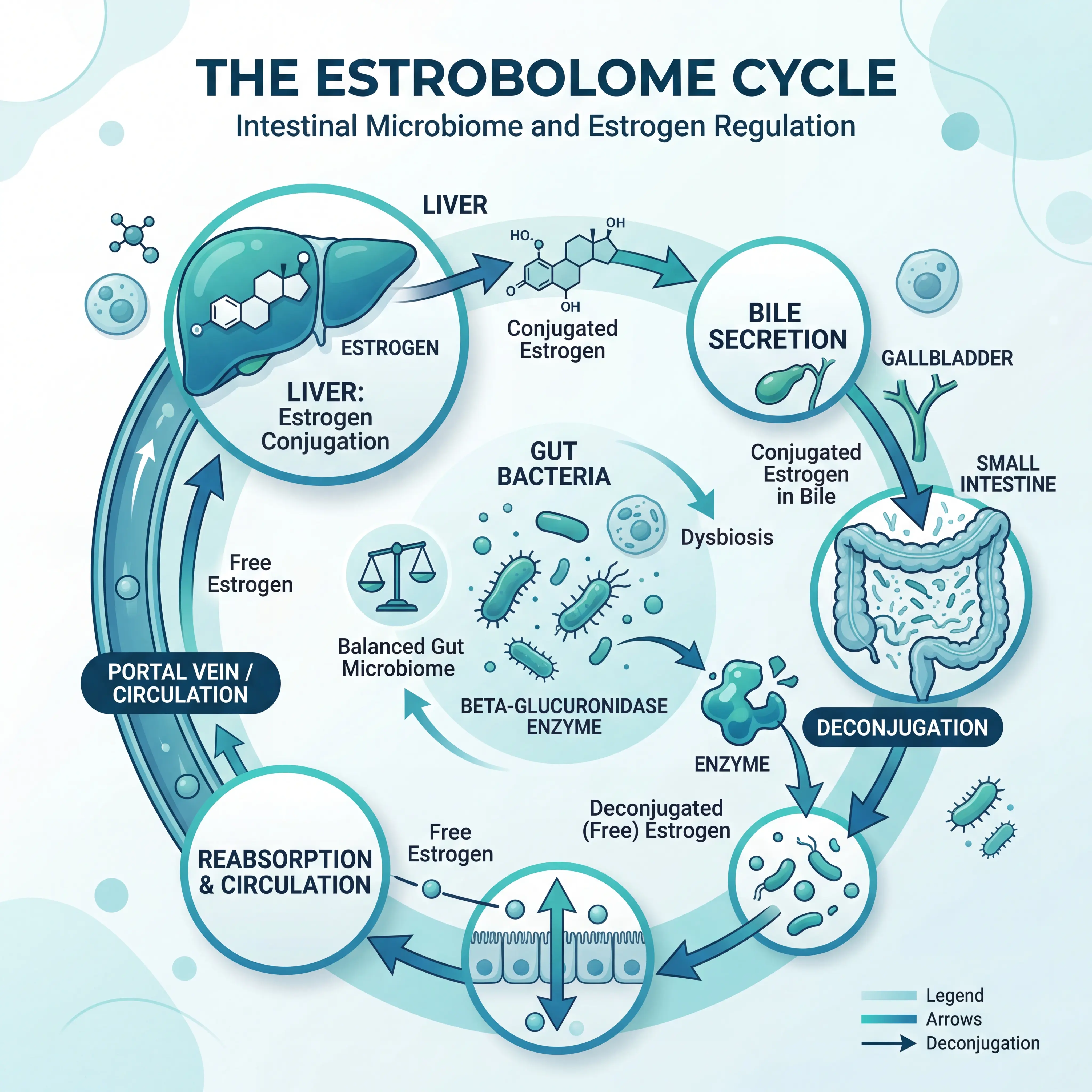
Inside your gut lives a specialized collection of bacterial genes collectively called the estrobolome. These genes encode enzymes — most notably beta-glucuronidase — that determine how much estrogen your body reabsorbs versus how much it eliminates. The process works like an internal recycling plant: your liver packages used estrogen into water-soluble conjugates and dumps them into the intestines via bile. From there, gut bacteria equipped with beta-glucuronidase strip the packaging off, converting conjugated estrogen back into its active form so it can re-enter circulation.
When the estrobolome functions normally, this recycling maintains estrogen within a range that supports ovulation, endometrial health, and bone density. Problems emerge at both extremes. Research published in the Journal of Molecular Endocrinology explains that when dysbiosis reduces beta-glucuronidase activity, too little estrogen re-enters circulation. The result can be irregular cycles, difficulty with ovulation, and metabolic disruptions linked to estrogen deficiency. Flip the equation — an overgrowth of beta-glucuronidase-producing bacteria — and circulating estrogen climbs too high, fueling estrogen-dependent conditions like endometriosis.
The estrobolome does not operate in isolation, though. The same bacterial communities that metabolize estrogen also produce SCFAs that regulate the synthesis of progesterone and estradiol in ovarian granulosa cells. A study examining porcine granulosa cells found that butyrate directly influenced progesterone secretion through the cAMP signaling pathway — lower concentrations stimulated production while higher concentrations suppressed it. This means the metabolic output of your gut flora is simultaneously tuning multiple hormonal dials that affect your cycle.
| Estrobolome State | Beta-Glucuronidase Activity | Circulating Estrogen | Potential Reproductive Impact |
|---|---|---|---|
| Balanced | Normal | Optimal range | Regular ovulation, healthy endometrium |
| Low diversity / dysbiosis | Reduced | Below optimal | Anovulation, irregular cycles, metabolic syndrome |
| Overgrowth of GUS-producers | Elevated | Above optimal | Endometriosis, estrogen-driven conditions |
So interventions that restore microbial diversity in the gut — from dietary fiber increases to targeted probiotics — can theoretically shift the estrobolome back toward balanced estrogen recycling. That is a different strategy than prescribing hormonal birth control to mask symptoms. It aims at the upstream cause instead of the downstream effects.
A Meta-Analysis of Nearly 2,000 Women Reveals Consistent Gut Changes in PCOS
Polycystic ovary syndrome affects somewhere between 5% and 20% of women of reproductive age worldwide, depending on the diagnostic criteria used. It is defined by some combination of excess androgens, irregular ovulation, and polycystic ovarian morphology. The underlying cause remains unclear, which makes management frustrating. Genetics, insulin resistance, inflammation, and hormonal imbalance all interact, and the gut microbiome appears to sit near the center of that mess.
The most comprehensive analysis to date — a systematic review and meta-analysis published in BMC Medicine examining 28 studies with 1,022 PCOS patients and 928 controls — found that women with PCOS consistently have lower gut microbial diversity. The Shannon diversity index, which measures both species richness and evenness, was significantly decreased (standardized mean difference of -0.27). More telling than the overall diversity drop was the pattern of which bacteria disappeared and which flourished.
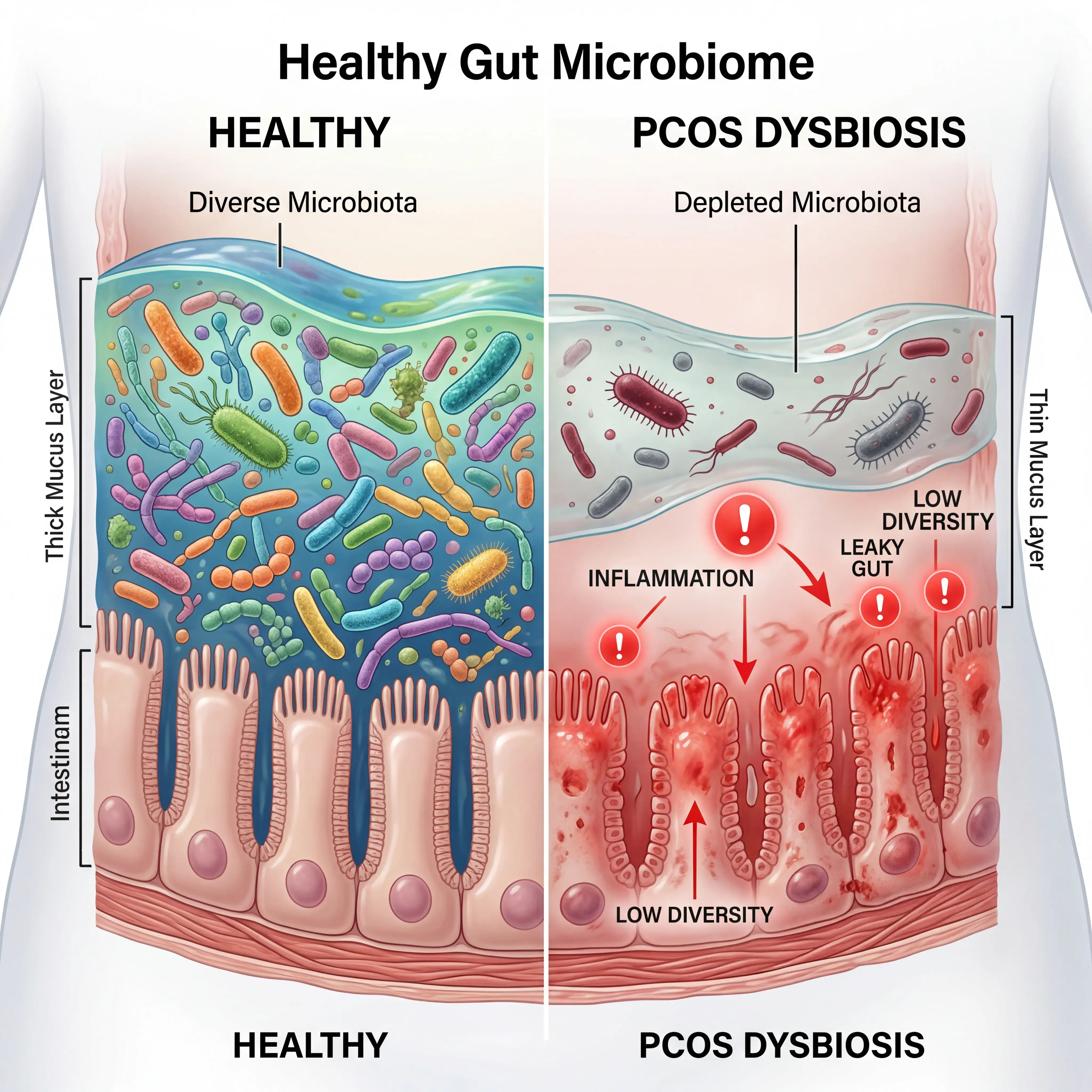
Women with PCOS showed consistent depletion of Lachnospira and Prevotella — both producers of anti-inflammatory short-chain fatty acids. In their place, pro-inflammatory genera like Bacteroides, Parabacteroides, Fusobacterium, and Escherichia/Shigella were enriched. The researchers described this as a fundamental shift in the gut ecosystem away from anti-inflammatory bacterial communities toward pro-inflammatory ones.
The strongest evidence for a causal role comes from a study published in Nature Medicine. Qi and colleagues demonstrated that when they performed fecal microbiota transplantation from women with PCOS into mice, those mice developed hallmark features of the syndrome — disrupted ovarian cycles, insulin resistance, and altered bile acid metabolism. That experiment moved past correlation. It showed that the microbial community itself can drive PCOS-like pathology, operating through what the researchers termed the gut microbiota-bile acid-interleukin-22 axis.
Insulin resistance in PCOS ties into this directly. Gut bacteria regulate insulin sensitivity through multiple mechanisms, including SCFA production, bile acid modification, and control of intestinal permeability. When the gut barrier weakens — a consequence of dysbiosis — bacterial fragments called lipopolysaccharides (LPS) leak into the bloodstream, triggering low-grade systemic inflammation that directly worsens insulin resistance. Insulin resistance, in turn, drives the ovaries to produce excess androgens, creating the hyperandrogenism that defines so much of the PCOS experience.
| Microbial Change in PCOS | Direction | Functional Consequence |
|---|---|---|
| Lachnospira, Prevotella | Depleted | Reduced SCFA production, weakened gut barrier |
| Akkermansia, Bifidobacterium | Depleted | Impaired mucin layer integrity, reduced immune regulation |
| Bacteroides, Fusobacterium | Enriched | Pro-inflammatory signaling, altered bile acid metabolism |
| Desulfovibrio | Enriched | Hydrogen sulfide production, metabolic disruption |
| Bacteroides vulgatus | Enriched | Bile acid deconjugation linked to hormonal dysregulation |
196 Million Women Have Endometriosis — and Their Gut Bacteria Tell a Different Story
Endometriosis involves endometrial-like tissue growing outside the uterus, most commonly on the ovaries, fallopian tubes, and pelvic peritoneum. It affects roughly 10% of reproductive-age women globally — approximately 196 million — and between 35% and 50% of women who seek treatment for infertility receive an endometriosis diagnosis. The condition is estrogen-dependent and inflammatory by nature, which places it squarely within the influence of the gut microbiome.
A 2025 systematic review and meta-analysis in Frontiers in Microbiology pooled data from 11 studies encompassing 1,727 women (433 with endometriosis, 1,294 controls) and found statistically significant differences in gut microbial diversity. The Shannon Index showed a standardized mean difference of 0.39, while the Simpson Index revealed an even larger difference in microbial richness (SMD of 0.91). These numbers represent a real and measurable divergence between the gut ecosystems of women with endometriosis and those without.
But how does an imbalanced gut actually promote endometrial lesion growth? The mechanism runs through at least two parallel tracks. First, the estrobolome connection: when beta-glucuronidase-producing bacteria overgrow, they recycle too much estrogen back into circulation. Since endometriotic lesions depend on estrogen for growth — they express high levels of aromatase and low levels of the enzymes that would inactivate estrogen — this excess fuel directly feeds lesion expansion. Second, gut dysbiosis weakens the intestinal barrier, allowing bacterial fragments to cross into the bloodstream and activate peritoneal macrophages, which in turn ramp up the inflammatory environment where endometriotic tissue thrives.
The inflammatory cascade does not stop there. Immune dysfunction in endometriosis involves elevated pro-inflammatory cytokines — IL-6, IL-8, TNF-alpha, and IL-1beta — alongside compromised immunosurveillance that would normally clear misplaced endometrial cells. Gut-derived inflammation amplifies this problem by tipping the balance of T helper cells away from the regulatory (Treg) phenotype that suppresses inflammation and toward the Th17 phenotype that promotes it. A 2025 review on the gut-endometrial axis identified this Th17/Treg imbalance as one of the shared mechanisms linking gut dysbiosis to endometriosis, PCOS, and implantation failure.
The bacterial contamination hypothesis adds another layer. Researchers have found significant Escherichia coli contamination in both menstrual blood and peritoneal fluid of women with endometriosis. The lipopolysaccharides from these bacteria may act as a chronic inflammatory stimulus at the sites where endometrial tissue implants, essentially creating a microenvironment that welcomes rather than rejects ectopic growth.
Worth noting: Commensal gut bacteria that produce SCFAs appear to have a protective effect against endometriotic lesion formation. When these protective species are lost through dysbiosis, the balance tips toward an inflammatory environment that promotes lesion growth and survival.
From Conception to Implantation: The Gut's Invisible Role in Getting Pregnant
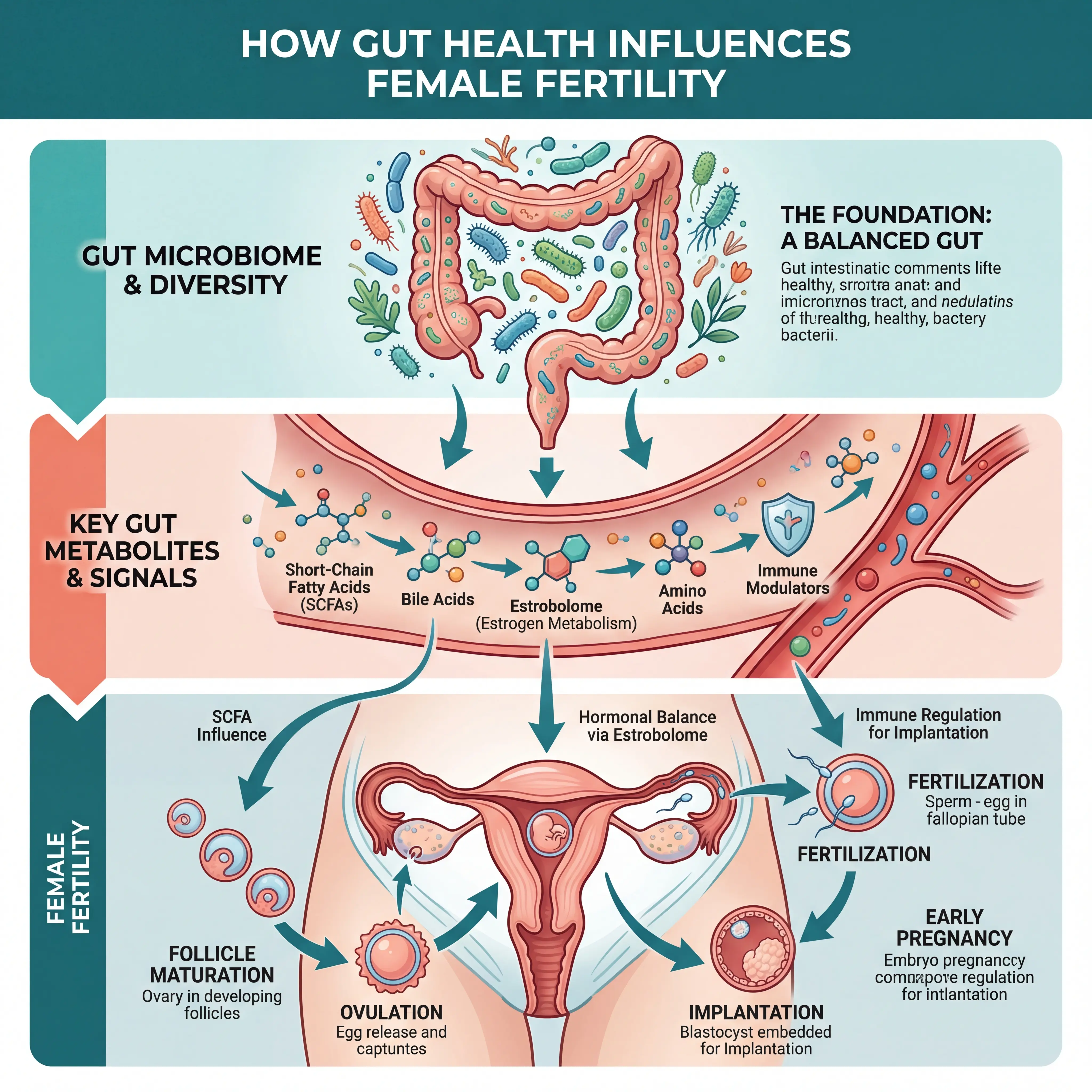
Fertility depends on a chain of precisely timed biological events: follicle maturation, ovulation, fertilization, embryo migration, and implantation into a receptive endometrium. What researchers are now discovering is that the gut microbiome influences virtually every step in that chain, not by acting directly on the reproductive organs but by shaping the systemic hormonal, metabolic, and immune conditions that those organs require to function.
Consider implantation — the moment an embryo burrows into the uterine lining. Successful implantation demands a finely tuned immune response. The body must tolerate a genetically foreign entity (the embryo carries paternal antigens) without triggering the rejection response it would mount against a pathogen. This tolerance depends on regulatory T cells, which are generated in part through the influence of gut-derived SCFAs. Research has shown that dysbiosis of the maternal gut microbiota enhances inflammatory responses that increase the risk of fetal rejection in early pregnancy.
The fertility implications extend to assisted reproductive technologies as well. Studies examining women undergoing IVF have associated the microbial composition of both the gut and the reproductive tract with implantation success rates. Women with a non-Lactobacillus-dominant endometrial microbiome — where species like Gardnerella, Streptococcus, and Enterobacteriaceae dominate — show significantly lower implantation and ongoing pregnancy rates. The gut microbiome, which communicates with the reproductive tract microbiome through shared immune and metabolic pathways, may be the upstream regulator of this vaginal and endometrial microbial landscape.
Lifestyle factors that damage the gut microbiome have measurable fertility effects. A sedentary lifestyle reduces microbial richness and promotes pro-inflammatory Proteobacteria, while chronic psychological stress alters gut microbiota through HPA axis activation and cortisol secretion, raising intestinal permeability and circulating LPS. A 2025 review on gut-reproductive crosstalk identified diet, exercise, stress, and environmental toxin exposure as the four primary modifiable factors that shape the gut microbiome's influence on fertility.
| Fertility Stage | Gut Microbiome Influence | Mechanism |
|---|---|---|
| Follicle maturation | Estrogen and progesterone regulation via estrobolome and SCFAs | Beta-glucuronidase activity, butyrate-cAMP signaling |
| Ovulation | Insulin sensitivity affecting ovarian function | SCFA regulation of glucose metabolism, LPS-driven insulin resistance |
| Implantation | Immune tolerance at the endometrium | SCFA-driven Treg cell generation, Th17/Treg balance |
| Early pregnancy | Inflammatory control preventing rejection | Gut barrier integrity, systemic cytokine regulation |
Separating What the Science Shows From What the Internet Claims
This topic has attracted plenty of interest online, and with it, a fair amount of misinformation. Here is where the evidence stands compared to what gets overstated.
| Myth | Fact |
|---|---|
| Taking any probiotic will fix hormonal imbalances | Not all probiotics are equivalent. Generic strains have not been shown to meaningfully shift the estrobolome. Specific species like Lactobacillus and Bifidobacterium show more targeted effects, but the research is still early and strain-specific. |
| Gut health causes PCOS | The relationship is bidirectional, not unidirectional. Gut dysbiosis contributes to PCOS pathology through inflammation and hormonal disruption, but PCOS-related hormonal changes (excess androgens) also reshape the gut microbiome. Neither "causes" the other in a simple linear way. |
| A healthy gut can cure endometriosis | Improving gut health may reduce the inflammatory burden that drives endometriosis progression, but it cannot eliminate existing endometriotic lesions. Gut-targeted approaches are better understood as complementary to — not replacements for — conventional treatment. |
| Fermented foods alone are enough to restore the gut microbiome | Fermented foods contribute beneficial bacteria, but dietary fiber is the primary fuel for SCFA-producing bacteria. Without adequate fiber intake, even well-colonized bacteria lack the substrate to produce the metabolites that matter for reproductive health. |
| Gut testing can diagnose reproductive conditions | Gut microbiome profiling may eventually serve as a diagnostic biomarker, but current commercial tests lack the specificity and clinical validation to diagnose PCOS, endometriosis, or infertility. |
Seven Evidence-Backed Strategies to Strengthen the Gut-Reproductive Connection
The research linking gut and reproductive health is solid enough to act on. These strategies focus on restoring microbial diversity, boosting SCFA production, and lowering the chronic low-grade inflammation that throws hormones off track.
1. Prioritize dietary fiber diversity. Fiber is the single most important dietary lever for the gut microbiome. A fiber-rich, plant-based diet enhances SCFA-producing taxa including Faecalibacterium prausnitzii, Lactobacillus, and Bifidobacterium. Aim for 25-35 grams daily from a variety of sources — legumes, whole grains, vegetables, fruits, nuts, and seeds. Variety matters because different fibers feed different bacterial species.
2. Reduce processed food and refined sugar. Western-style diets high in saturated fats and refined sugars reduce microbial diversity and promote pro-inflammatory species like Bilophila wadsworthia. For women with PCOS, this has a compounding effect: the inflammation worsens insulin resistance, which worsens androgen excess.
3. Include polyphenol-rich foods. Polyphenols from berries, green tea, dark chocolate, and olive oil enrich beneficial microbes like Akkermansia muciniphila, which is specifically depleted in women with PCOS. These compounds also have direct anti-inflammatory properties that complement their prebiotic effects.
4. Move regularly at moderate intensity. Regular physical activity promotes microbial diversity and enhances SCFA production. Both aerobic exercise and resistance training have demonstrated positive effects on the gut microbiome. Sedentary behavior, by contrast, is associated with an expansion of Proteobacteria and impaired gut barrier function.
5. Manage stress deliberately. Chronic stress activates the HPA axis, raises cortisol, increases intestinal permeability, and reduces Bifidobacterium populations. Practices that lower cortisol — including vagus nerve stimulation techniques, meditation, and adequate sleep — protect the gut-reproductive axis.
6. Consider targeted probiotic strains. While the evidence is not yet definitive enough for universal prescription, specific Lactobacillus and Bifidobacterium strains have shown promise in small trials for improving gut diversity in the context of reproductive conditions. Consult a healthcare provider about strains with clinical evidence rather than grabbing a generic product.
7. Minimize unnecessary antibiotic exposure. Antibiotics are the fastest way to disrupt the estrobolome and gut microbial diversity. When antibiotics are medically necessary, following a course with targeted probiotic supplementation and increased prebiotic fiber may help accelerate microbial recovery.
Frequently Asked Questions
Can improving gut health help manage PCOS symptoms?
Evidence suggests it can be part of the picture. Research shows that women with PCOS consistently have reduced gut microbial diversity and fewer SCFA-producing bacteria. Dietary interventions that restore diversity — particularly increased fiber intake and reduced refined sugars — have been associated with improvements in insulin sensitivity and inflammatory markers, both of which drive PCOS symptoms. However, gut-focused strategies work best alongside standard care, not as standalone treatments.
Does the gut microbiome affect endometriosis pain levels?
The gut microbiome likely contributes to the inflammatory environment that amplifies endometriosis-related pain, though direct clinical trials are limited. Gut dysbiosis drives systemic inflammation through intestinal permeability and LPS translocation, which can heighten the inflammatory signaling at endometriotic lesion sites. Women with endometriosis also show altered gut microbial diversity according to multiple studies, suggesting that restoring gut balance may reduce inflammatory burden and potentially help with pain management.
Should I get a gut microbiome test if I am struggling with fertility?
Commercial gut microbiome tests can provide interesting information about your microbial composition, but they do not yet have validated clinical cut-offs for diagnosing fertility-related issues. The research connecting specific bacterial profiles to fertility outcomes is still in its early stages. A more practical first step is to focus on the dietary and lifestyle factors that are known to improve both gut health and fertility outcomes — adequate fiber, regular exercise, stress management, and reducing processed food intake.
How long does it take for gut microbiome changes to affect reproductive hormones?
Dietary changes can begin shifting gut microbial composition within days, but meaningful changes in diversity and metabolic output typically require several weeks to months of consistent habits. The downstream effects on hormonal balance — through estrobolome activity, SCFA production, and inflammation reduction — likely take even longer to manifest as measurable changes in reproductive hormones or cycle regularity. Patience and consistency matter more than dramatic short-term interventions.
Are fecal microbiota transplants being used to treat reproductive conditions?
Fecal microbiota transplantation (FMT) has been explored in animal research for PCOS — the same Nature Medicine study that showed FMT from PCOS patients could induce PCOS in mice also demonstrated that modifying the gut microbiota could reverse some features. In humans, FMT is currently approved only for recurrent Clostridioides difficile infections. Clinical trials for reproductive applications are in very early stages, and FMT is not recommended as a treatment for PCOS, endometriosis, or infertility outside of research settings.
Related Articles
- Inositol for PCOS, Anxiety, and Hormonal Balance: What the Research Shows — A deep dive into one of the most studied supplements for PCOS management, including its effects on insulin sensitivity and ovarian function.
- Endometriosis Symptoms, Natural Pain Relief, and Diet Strategies — Evidence-based dietary and lifestyle approaches for managing endometriosis symptoms alongside conventional treatment.
- Estrogen Dominance: Symptoms, Causes, and How to Rebalance Naturally — How excess estrogen develops and what you can do to restore hormonal equilibrium through diet, lifestyle, and targeted supplementation.
- Leaky Gut Syndrome: Symptoms, Causes, and Evidence-Based Healing — The mechanisms behind intestinal permeability and its connections to systemic inflammation and chronic disease.
- Butyrate: The Gut Metabolite That Heals Your Colon and Protects Your Brain — Everything you need to know about the short-chain fatty acid that plays a central role in gut barrier integrity and immune regulation.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.