Same drug, opposite effect: how dosage flips naltrexone's purpose
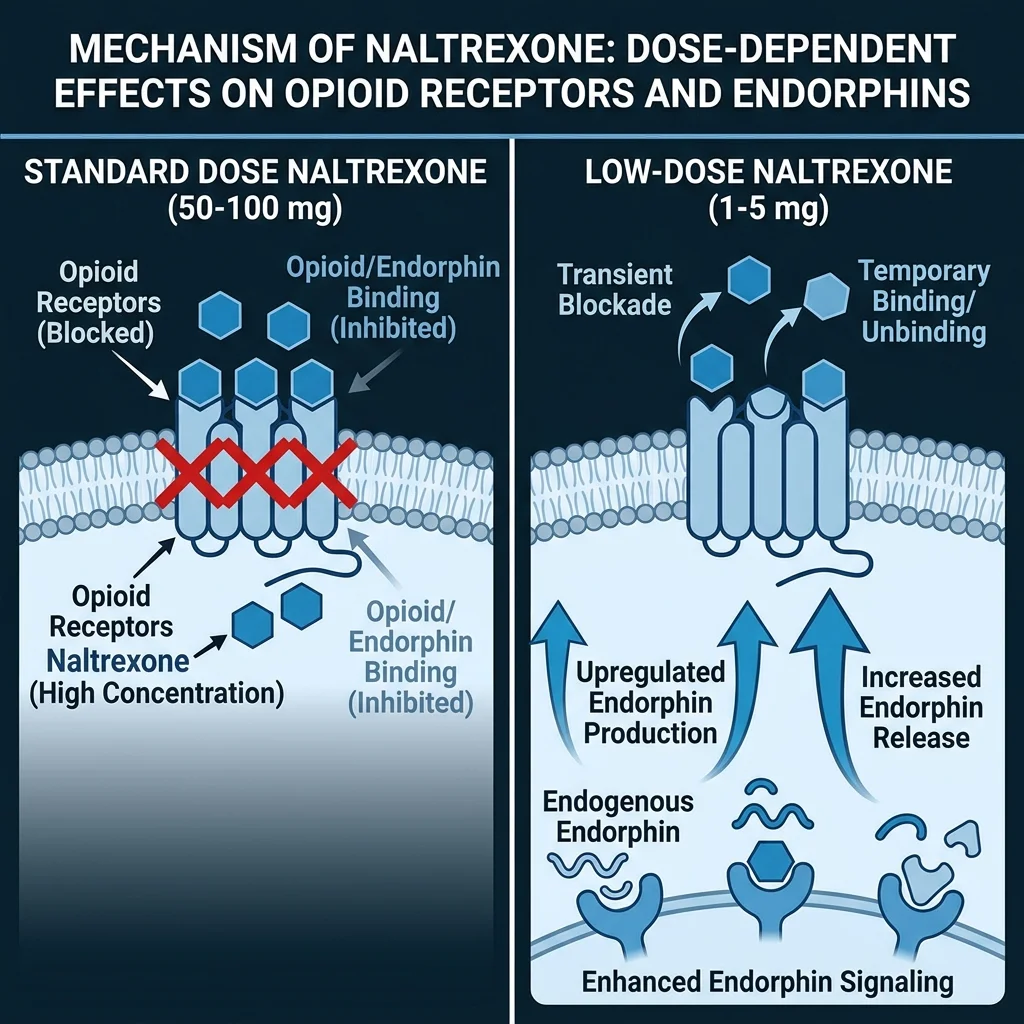
Naltrexone was synthesized in 1963 and gained FDA approval in 1984 for a single purpose: blocking opioid receptors to treat addiction. At the standard 50-100mg daily dose, it saturates mu-, delta-, and kappa-opioid receptors so thoroughly that drinking alcohol loses its rewarding buzz and heroin produces no high. That part of naltrexone's story is well-established pharmacology.
The strange part starts at roughly one-tenth that dose.
At 1-5mg daily, naltrexone does something pharmacologists call hormesis — a phenomenon where a substance produces qualitatively different effects at low versus high concentrations. Think of it like a dimmer switch versus an on/off switch. Standard-dose naltrexone slams the opioid system shut. Low-dose naltrexone (LDN) barely touches it — blocking receptors for a few hours, then stepping away. That brief, partial blockade appears to trigger the body to compensate by producing more endorphins and upregulating opioid receptor expression.
Dr. Bernard Bihari, a New York physician, was the first to test this idea clinically. In the mid-1980s, he began prescribing 1.5-3mg naltrexone as adjunct therapy for AIDS patients, reasoning that the opioid rebound effect might boost immune function. That was four decades ago. The first published trial in humans didn't arrive until 2007. In the years between, LDN spread through patient communities and alternative medicine practitioners long before the research could catch up.
What makes LDN unusual in pharmacology is that these aren't smaller versions of the same effect. The low dose activates an entirely different receptor pathway than the standard dose, acting on immune cells rather than just opioid receptors. That dual identity is what makes LDN interesting to researchers and maddening to anyone trying to pin down what it actually does.
Your immune system's off switch might be stuck
The mechanism that gets most attention in LDN research has nothing to do with opioid receptors. LDN acts as an antagonist of Toll-like receptor 4 (TLR4), a receptor found on microglia — the brain and spinal cord's resident immune cells. When functioning normally, microglia patrol for threats. When they get stuck in an activated state, they become a problem.
Imagine a smoke detector that goes off during an actual fire, but then never stops shrieking even after the fire is out. Chronically activated microglia keep pumping out inflammatory signals — TNF-alpha, interleukin-6, nitric oxide — even when there's no active threat to justify the response. That sustained inflammation shows up in conditions like fibromyalgia, chronic fatigue, and several autoimmune diseases.
LDN appears to quiet those overactive microglia. Specifically, it disrupts the TRIF portion of TLR4's downstream signaling cascade, reducing the production of TNF-alpha and interferon-beta. This isn't the same mechanism as taking an aspirin or a steroid. Traditional anti-inflammatories target the end products of inflammation. LDN may work further upstream, at the cellular command level.
Researchers at Stanford confirmed something that supports this mechanism indirectly. In fibromyalgia patients, baseline erythrocyte sedimentation rate (ESR) — a crude marker of systemic inflammation — predicted who would respond to LDN. The correlation was strong: r=0.58 (p=0.0001) for the LDN group versus r=0.06 for placebo. Patients with more inflammation got more relief. That doesn't prove the mechanism, but it fits the theory neatly.
TLR4 receptors also sit on mast cells and type 1 macrophages — immune cells outside the nervous system that release their own cocktail of inflammatory mediators. So LDN's immunomodulatory reach may extend beyond the brain into the broader immune system. Whether that matters in practice is a different question, and the clinical evidence doesn't give a clean answer.
What 80% remission in Crohn's and mixed MS results tell us
Crohn's disease is where LDN's evidence looks strongest. In multiple small trials, over 80% of participants showed significant improvement, and these weren't just self-reported symptom changes. Researchers documented reduced objective markers of inflammation, including improved endoscopic severity scores — meaning the actual tissue inflammation visible on camera diminished. For a cheap generic medication with few side effects, those numbers caught attention.
Multiple sclerosis tells a more complicated story. A 2006 open-label pilot by Dr. Miriam Gironi and colleagues tested LDN in 40 patients with primary progressive MS over six months. The drug was well tolerated, reduced spasticity significantly, and beta-endorphin levels in patients' blood mononuclear cells rose — a real-time biochemical confirmation that the opioid rebound mechanism was actually happening in living patients.
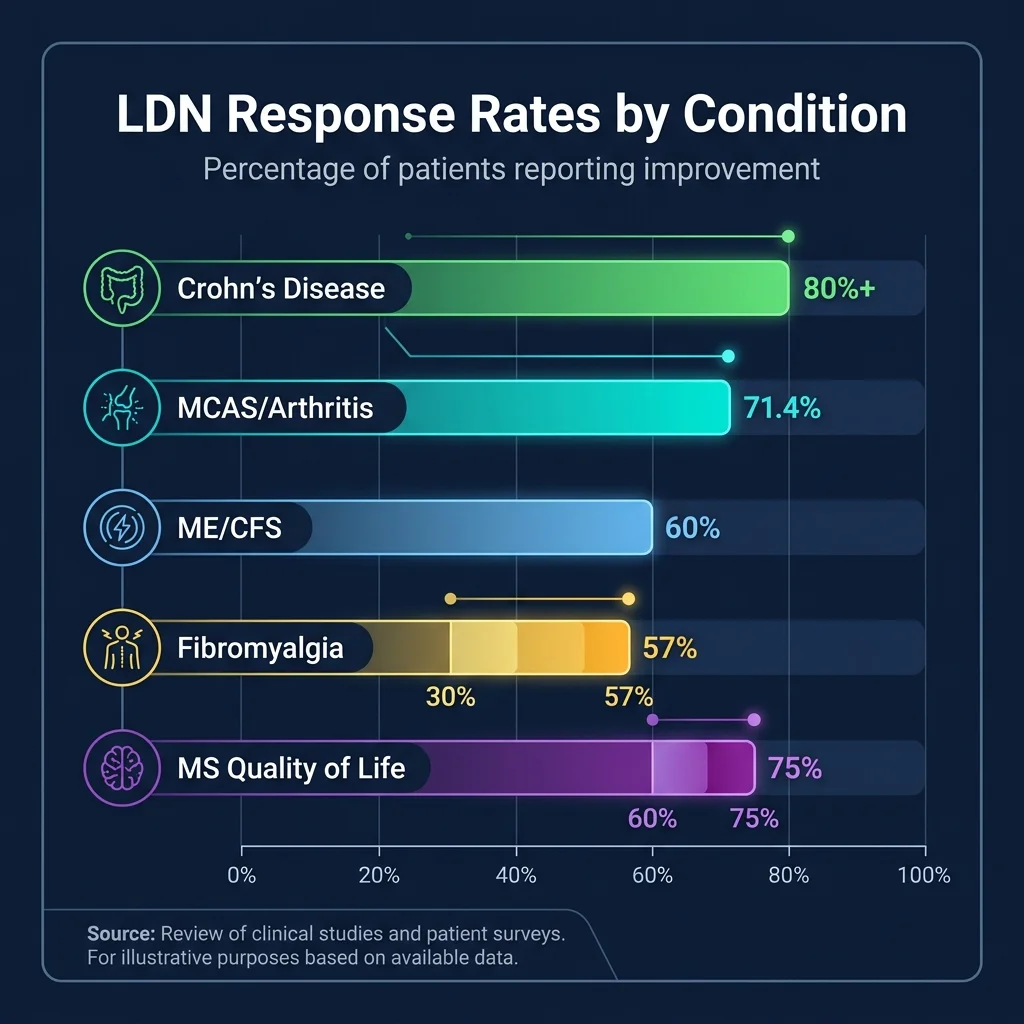
A larger retrospective study surveyed 215 MS patients (87% relapsing-remitting) who took LDN for a median of 804 days. The results read well: 77% reported no side effects, 60% experienced less fatigue, and 75% endorsed improved quality of life. But retrospective surveys with self-selected patients are far from definitive.
The controlled evidence is genuinely split. One RCT with 60 MS patients found significant improvement in mental health quality of life scores. Another, larger RCT with 96 patients found no significant difference between LDN and placebo on the same measures. A 10-year retrospective comparing LDN-only versus LDN plus glatiramer acetate showed no between-group difference in disease progression, with half of both cohorts maintaining stable disease.
Beyond Crohn's and MS, the autoimmune evidence thins considerably. A 2025 scoping review identified 68 studies spanning fibromyalgia, MS, CRPS, Crohn's, dermatological conditions, and cancer, but most were case reports or small uncontrolled series. For conditions like rheumatoid arthritis, lupus, or Hashimoto's thyroiditis — conditions that come up constantly in LDN patient communities — published controlled evidence barely exists.
Conditions with strong inflammatory components show encouraging early signals. But "early" has described LDN research for nearly two decades now, and the gap between what patients report and what controlled trials can confirm hasn't narrowed much.
The fibromyalgia paradox: patients swear by it, RCTs can't prove it
Fibromyalgia has attracted more LDN research than any other condition, and the results are a mess. The earliest data looked promising. Dr. Jarred Younger's group at Stanford ran two trials culminating in a double-blind crossover study of 30 women where 57% showed clinically significant pain reduction on 4.5mg LDN. Half rated themselves "much improved" or "very much improved."
Then the larger, more rigorous trials arrived, and the picture shifted. Bested and colleagues in Denmark conducted a randomized, double-blind, placebo-controlled crossover study with 58 fibromyalgia patients. The primary outcomes — FIQR scores and summed pain intensity — showed effect sizes of 0.15 and 0.13 respectively. Their conclusion was blunt: "no clinically relevant analgesic efficacy."
A systematic review published in 2024 by Rassi-Mariani and colleagues pooled 7 RCTs comprising 406 patients and concluded that LDN was "not effective in managing chronic pain." Meanwhile, a separate meta-analysis by Vatvani et al. had reported more favorable results — until Bruun and colleagues published a 2026 re-analysis identifying data extraction errors in the original. After correction, the pooled odds ratio for 30% pain improvement dropped from 3.32 to 2.47, and a re-analysis of pain score changes yielded a non-significant standardized mean difference.

Yet real-world practice tells a different story. A 2025 retrospective cohort of 93 chronic pain patients from a single Canadian pain practice found 53.8% reporting subjective benefit from LDN. Among the 27 fibromyalgia patients, 55.6% said their symptoms improved. The highest response rates appeared in patients with mast cell activation syndrome and arthritis-related conditions, both at 71.4%.
How do you reconcile 53.8% of real-world patients feeling better with RCTs showing non-significant results? One possibility: LDN works well for a subset of patients but the effect washes out in group averages. The dosing question matters here. Dr. Norman Marcus and colleagues demonstrated through careful titration that the maximally effective dose is idiosyncratic, ranging from 0.1 to 6.0mg across 41 patients. Many RCTs used a fixed 4.5mg dose — which might be the wrong dose for a substantial fraction of participants.
Fibromyalgia researchers estimated that the dose effective in 50% of patients (ED50) was 3.88mg and the ED95 was 5.40mg, but only 11 of 25 subjects were classified as responsive at all. The arithmetic points to a drug that genuinely helps some people and does little for others, with no reliable way yet to predict who falls into which group — beyond that tantalizing ESR correlation.
The weight loss claim that borrowed someone else's data
Search for "low-dose naltrexone weight loss" and you'll find no shortage of clinic websites, TikTok videos, and wellness blogs touting LDN as a weight management tool. The evidence behind these claims is thinner than you'd expect, largely because it borrows from a different drug at a different dose.
Naltrexone does have a legitimate, FDA-approved role in weight management — but at standard doses, combined with another drug. The combination of naltrexone (32mg/day) and bupropion (360mg/day), marketed as Contrave, targets the brain's appetite-regulating POMC neurons. In the COR-BMOD trial, NB32 plus behavioral modification produced 9.3% weight loss versus 5.1% for placebo over 56 weeks. Across phase 3 trials, responders achieved a mean 11.7% body weight reduction.
But that's 32mg of naltrexone, not 4.5mg. And it's combined with bupropion, which contributes its own appetite-suppressing mechanism by blocking norepinephrine and dopamine reuptake.
What about naltrexone alone for weight? A meta-analysis in The Lancet's eClinicalMedicine found naltrexone monotherapy produced just -0.47kg of weight loss — less than half a kilogram. The combination with bupropion reached -4.06kg. For context, semaglutide achieved -8.48kg in the same analysis.

The part nobody mentions: no randomized controlled trials exist for LDN at 1-5mg specifically for weight loss. Zero. The weight claims appear to borrow from the naltrexone/bupropion data (which uses 7 times the dose, plus a second drug) or from the theoretical mechanism — opioid receptor blockade reduces the rewarding properties of food, which may decrease preference for palatable foods.
Some patients do report weight changes while taking LDN for pain or autoimmune conditions. Whether that reflects a direct drug effect, reduced inflammation affecting metabolism, increased activity from pain relief, or something else entirely hasn't been studied in controlled conditions. Prescribing LDN specifically for weight loss currently means prescribing based on mechanism-of-action speculation, not clinical evidence.
Vivid dreams, mild nausea, and a $35 monthly tab
One thing the research does agree on: LDN doesn't cause much trouble. Whether a study finds the drug effective or useless, the side effect profile stays mild.
The most distinctive side effect is vivid dreams, reported by approximately 37% of participants in Younger's trials. Not nightmares in most cases — just unusually vivid or memorable dream content. This effect appears quickly (sometimes the first night) and tends to diminish over weeks. It's also the most commonly reported side effect during placebo administration in these trials, so some of it may reflect expectancy.
In the largest real-world dataset, Aalto and colleagues' 2025 retrospective of 93 patients found 49.5% experienced at least one adverse effect. Nausea led the list (18 patients), followed by fatigue (7), vivid dreams (6), and insomnia (6). Of the 46 patients reporting side effects, 22 said they were transient. No serious adverse events were recorded in this cohort or in any published trial.
| Side effect | Approximate frequency | Typical duration |
|---|---|---|
| Vivid dreams | 6-37% | Decreases over weeks |
| Nausea | 19% | Often transient |
| Fatigue | 7-8% | Variable |
| Insomnia | 5-6% | Often resolves with morning dosing |
| Headache | Slightly above placebo | Variable |
One caveat the safety data can't answer: long-term chronic use. Every published study runs weeks to months. The Norwegian Institute of Public Health's systematic review flagged this directly: "it is extremely uncertain whether low-dose naltrexone has been investigated in a sufficient number of patients to identify rare side effects." Standard-dose naltrexone has a long safety track record, but the unique mechanisms activated at low doses could theoretically carry their own risks. Immune suppression, for instance, could theoretically reduce immunosurveillance — though no such effect has been reported at any naltrexone dosage.
The monthly cost averages about US$35 from compounding pharmacies, since no commercial 4.5mg formulation exists. That price assumes no insurance coverage, which is typical for off-label prescriptions. Compared to FDA-approved fibromyalgia medications that can run over $100/month, it's inexpensive. The drug has no known abuse potential — producing no euphoria and carrying no addiction risk. Cheap, safe, and non-addictive isn't a bad sales pitch even when the efficacy data is thin.
Who should avoid it: anyone taking opioid medications (LDN could reduce their effectiveness or trigger withdrawal), individuals with severe liver disease, and anyone whose condition requires reliable immunosuppression — the immunomodulatory effects could theoretically interfere with transplant medications or cancer immunotherapy, though this hasn't been studied.
Getting a prescription for a drug with no approved indication
There is no FDA-approved use of naltrexone at any dosage for chronic pain, autoimmune disease, or inflammation. All LDN prescribing is off-label — legal, common in medicine, but it means your physician is relying on their clinical judgment rather than regulatory validation.
The practical barriers start at the pharmacy. Naltrexone is commercially available only as 50mg tablets. Getting a 4.5mg dose requires a compounding pharmacy, which not every patient has convenient access to. Some patients attempt to create their own doses by splitting tablets or dissolving them in water — an approach that introduces unintended dosing variability and that no researcher recommends.

If you're considering LDN, a productive conversation with your doctor might cover these specifics:
- What you've tried already and why you're seeking alternatives
- That you understand LDN is off-label and experimental, with mixed evidence
- The specific condition you're hoping to treat — evidence varies dramatically by condition
- Whether you take opioid medications or immunosuppressants (potential contraindications)
- Your willingness to try a titration protocol rather than jumping to a fixed dose
Regarding dosing: the research increasingly suggests that the effective dose varies between individuals, potentially ranging from 0.1mg to 6.0mg. Most prescribers start at 1.5mg and increase gradually to 4.5mg, but the data from Marcus and colleagues suggests that some patients respond only to very specific doses and that both too little and too much can be ineffective. If a fixed 4.5mg dose doesn't work, that may not mean LDN doesn't work for you — it may mean you haven't found your dose.
Expect patience. In clinical trials, meaningful separation from placebo wasn't observed until at least one month of treatment, with two months generally needed to gauge effectiveness. Bedtime dosing is standard, though patients who experience insomnia often switch to morning administration.
Physicians who prescribe LDN know the awkward truth: the evidence is mixed. The Norwegian Institute of Public Health reviewed all available controlled evidence and rated it "very low quality" — not because the drug is dangerous, but because studies were uniformly small, short, and methodologically limited. Well-designed RCTs with adequate sample sizes remain the missing piece. Until they arrive, prescribing LDN means operating in that uncomfortable clinical space between "it probably doesn't harm" and "we can't prove it helps."
Frequently Asked Questions
Can I take LDN while on opioid pain medication?
This combination carries real risk. Even at low doses, naltrexone could block enough opioid receptors to reduce pain medication effectiveness or, in opioid-dependent individuals, trigger withdrawal symptoms. All published LDN trials have excluded patients taking opioid analgesics. If you currently use opioids, discuss this conflict directly with your prescriber before considering LDN.
How is LDN different from the naltrexone in Contrave for weight loss?
Contrave contains 32mg of naltrexone combined with 360mg of bupropion in an extended-release formulation. LDN uses 1-5mg of naltrexone alone. The dose, formulation, and mechanism of action differ substantially. Contrave's weight loss effects cannot be extrapolated to the individual components because the extended-release combination modifies how each drug is absorbed, distributed, and metabolized.
Why isn't LDN FDA-approved if it's been used since the 1980s?
FDA approval requires large, expensive clinical trials funded by the entity seeking approval — typically pharmaceutical companies. Naltrexone is a generic medication with no patent protection, which means no company has financial incentive to fund the trials. The research that does exist has been funded by smaller grants from foundations and university research funds. This funding gap explains why evidence remains limited despite decades of clinical use.
Is the compounding pharmacy version of LDN safe?
Compounding pharmacies produce LDN because no commercial manufacturer makes a 4.5mg formulation. Quality depends on the pharmacy. Look for a pharmacy accredited by the Pharmacy Compounding Accreditation Board (PCAB) or one that provides certificates of analysis. Self-dosing by splitting 50mg tablets introduces significant variability and is not recommended by any researcher in the field.
How long should I try LDN before deciding it doesn't work?
Clinical trials suggest that at least two months of consistent use may be needed before meaningful effects emerge. Some prescribers recommend a three-month trial. If a fixed dose produces no benefit, discuss a titration approach with your doctor — the evidence shows that response can be highly dose-specific.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.