Your body runs on two fuel tanks
Your cells carry two main fuel sources: glucose (from carbohydrates) and fatty acids (from stored or dietary fat). A healthy metabolism toggles between them depending on what is available and what the body needs at any given moment. After a meal rich in carbohydrates, insulin rises, glucose floods into cells, and your body burns mostly sugar. Several hours into a fast or during a long walk, insulin drops, fat stores open up, and fatty acid oxidation takes over.
Researchers call this toggle metabolic flexibility — a term first formalized by Kelley and Mandarino as "the capacity to switch from predominantly lipid oxidation during fasting to the suppression of lipid oxidation and increased glucose uptake under insulin-stimulated conditions." It sounds technical, but the basic idea is straightforward: your metabolism should be responsive, not rigid.
Scientists measure this by tracking the respiratory quotient (RQ) — the ratio of carbon dioxide produced to oxygen consumed. An RQ of 0.7 means the body is burning almost entirely fat. An RQ near 1.0 means it is burning almost entirely glucose. A metabolically flexible person shows clear RQ shifts between meals and fasting, between rest and exercise, between daytime activity and sleep.
Quick take: Metabolic flexibility is your body's ability to efficiently switch between burning fat and glucose based on what fuel is available. Poor metabolic flexibility is linked to obesity, type 2 diabetes, and accelerated aging.
How fuel switching actually works
The molecular machinery behind fuel switching operates mainly at the level of your mitochondria — the organelles that generate most of your cellular energy. Two competing fuel pathways converge inside them, and a handful of enzymes act as traffic controllers deciding which fuel gets oxidized.
When glucose is plentiful after a carbohydrate-rich meal, glycolysis breaks it down into pyruvate, which enters mitochondria via the pyruvate dehydrogenase complex (PDC). This process increases malonyl-CoA concentrations, which block carnitine palmitoyltransferase-1 (CPT-1), the gatekeeper enzyme that shuttles fatty acids into mitochondria. The result: glucose gets burned, fat oxidation is suppressed.
Reverse the conditions — skip a meal, exercise for an hour, sleep through the night — and insulin drops. Adipose tissue releases free fatty acids into the bloodstream. CPT-1 reopens because malonyl-CoA levels fall. Fatty acids flood into mitochondria and become the dominant fuel. Pyruvate dehydrogenase kinase 4 (PDK4) simultaneously shuts down glucose oxidation by deactivating PDC, completing the switch.
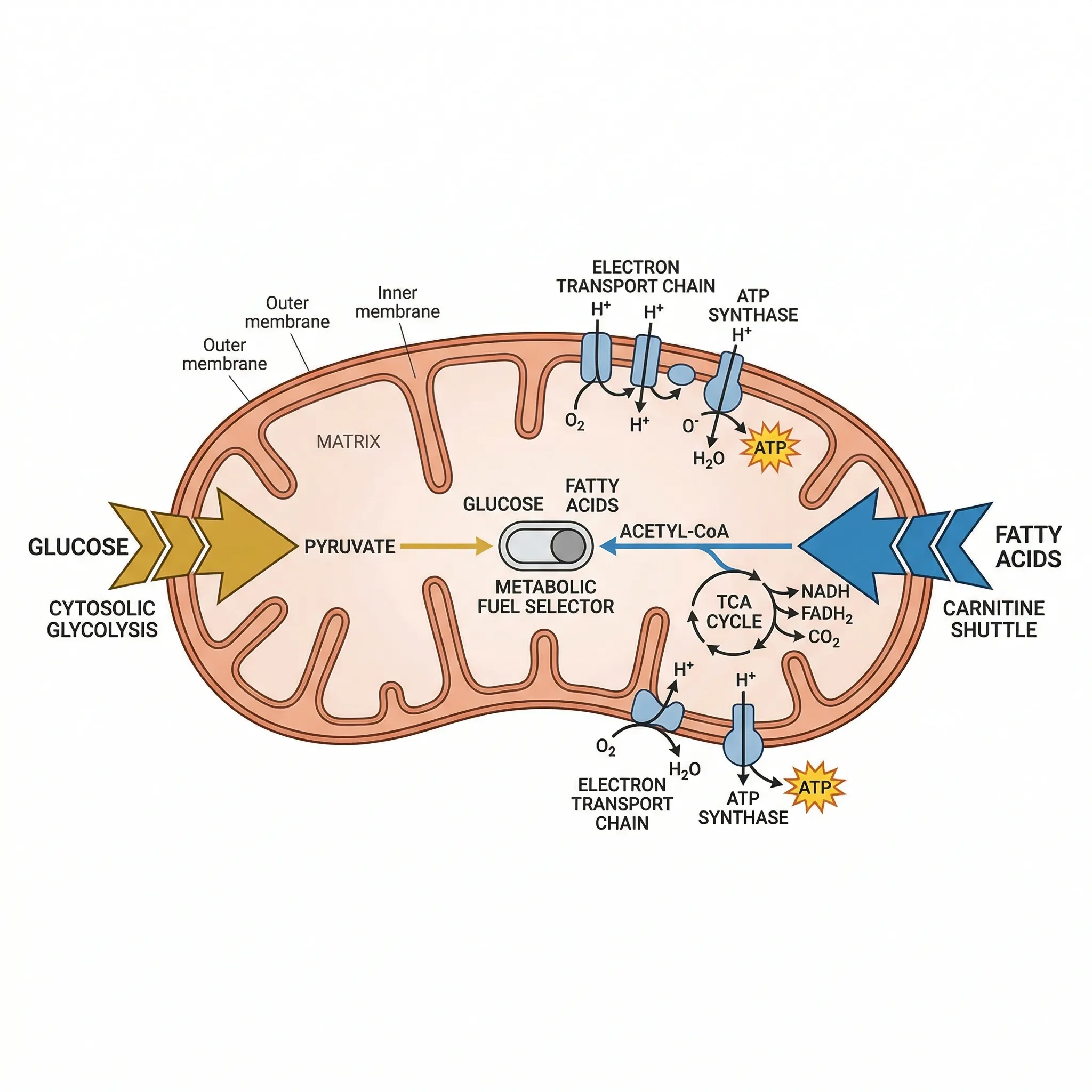
This is not a binary on-off switch. Your body almost always burns a mixture of both fuels. The ratio shifts along a continuum, and that shift happens continuously throughout the day. Goodpaster and Sparks (Cell Metabolism, 2017) describe it as a spectrum of adaptability — healthy people show wide RQ swings over 24 hours, while metabolically inflexible people show dampened, flat RQ profiles.
Skeletal muscle drives most of this fuel switching. It accounts for 60 to 80 percent of insulin-stimulated glucose metabolism, making it the largest consumer of both glucose and fat during daily activity and exercise. When skeletal muscle mitochondria work well, the whole system stays responsive. When they decline — through inactivity, chronic overfeeding, or aging — the system gets sluggish.
When the switch gets stuck
Metabolic inflexibility is what happens when your body loses the ability to transition smoothly between fuel sources. Instead of cleanly shifting to fat burning during a fast, cells keep trying to burn glucose. Instead of ramping up glucose oxidation after a meal, they stay partly reliant on fatty acids. The RQ stays in a narrow, muddled middle range.
Several overlapping factors drive this:
| Factor | Mechanism | Consequence |
|---|---|---|
| Insulin resistance | Cells resist insulin signaling, reducing glucose uptake | Less glucose enters cells for oxidation, trapping the body in partial fat-burning mode even after meals |
| Ectopic lipid accumulation | Fat deposits inside muscle and liver cells (ceramides, diacylglycerols) | Disrupts insulin signaling pathways, creating a vicious cycle of inflexibility |
| Mitochondrial decline | Reduced mitochondrial number, content, or function | Lower capacity to oxidize any substrate efficiently |
| Adipose tissue dysfunction | Fat tissue fails to properly store or release fatty acids | Excess free fatty acids flood other tissues at inappropriate times |
| Sedentary lifestyle | Muscles lose oxidative capacity without use | Reduced enzyme activity (citrate synthase, CPT-1) needed for fuel switching |
The cascade is self-reinforcing. Galgani, Moro, and Ravussin (2008) showed that when skeletal muscle cannot match fat oxidation to lipid uptake, fat accumulates inside muscle cells as triglycerides. Excess lipid accretion then produces ceramides and diacylglycerols — lipid species that impair insulin signaling through increased serine phosphorylation of insulin receptors. This lipotoxicity makes the cell even more insulin resistant, which further impairs fuel switching.
What makes this difficult to detect is that metabolic inflexibility often develops years before blood sugar levels rise enough for a diabetes diagnosis. Cleveland Clinic notes that insulin resistance can be present without any symptoms at all — the pancreas compensates by producing more insulin, masking the problem until it can no longer keep up.

Exercise: the fastest way to restore fuel switching
If metabolic inflexibility is partly a disease of disuse, exercise is the most direct fix. The data holds up across populations — lean, obese, type 2 diabetic. Exercise improves fuel switching in all of them.
One of the clearest demonstrations comes from Battaglia and colleagues at East Carolina University (2012). They put lean and obese men on a 70% fat diet for three days, then measured fatty acid oxidation in muscle biopsies. Lean subjects increased complete palmitate oxidation by 27.3%. Obese subjects? A 1% increase — essentially nothing. Their muscles could not respond to the increased fat availability.
Then both groups did ten consecutive days of aerobic exercise (one hour daily at 70% VO2peak). After seven days of training, both groups increased fatty acid oxidation to the same extent. The exercise erased the gap. As the researchers put it, exercise training "enables the skeletal muscle of obese individuals to respond similarly to their lean counterparts when confronted with an increase in dietary lipid."
A larger 2021 study published in Diabetologia used 24-hour whole-room calorimetry to measure metabolic flexibility across three groups: active healthy individuals, sedentary obese people without diabetes, and obese people with type 2 diabetes. Sleep RQ values differed across the spectrum — 0.823 for the active group, 0.860 for the obese non-diabetic group. After ten weeks of supervised aerobic training (four sessions per week, 45 minutes at 75% VO2max), the type 2 diabetes group showed significantly improved post-breakfast RQ responses, indicating restored ability to switch to carbohydrate oxidation after a meal.
| Exercise type | How it helps metabolic flexibility | Evidence strength |
|---|---|---|
| Zone 2 endurance (60-70% max HR) | Increases mitochondrial density and fatty acid oxidation enzymes (citrate synthase, HAD) | Strong — multiple RCTs |
| High-intensity interval training | Improves both glucose uptake and fat oxidation capacity; stimulates mitochondrial biogenesis | Strong — improves VO2max which correlates with metabolic flexibility |
| Resistance training | Increases muscle mass (the body's largest metabolic organ); improves insulin sensitivity | Moderate — fewer direct MetFlex studies, but strong indirect evidence |
| Combined aerobic + resistance | Addresses both oxidative capacity and muscle mass simultaneously | Strong — 12 weeks of combined training restored MetFlex in T2D patients to healthy levels |
The speed of improvement is worth noting. Citrate synthase activity — a marker of mitochondrial content — increased measurably after just seven days of training in the Battaglia study. This aligns with broader exercise physiology showing that mitochondrial remodeling begins within days of starting a training program, not weeks or months. You do not need to become a marathon runner to see benefits.
A reasonable starting program: three to four sessions of moderate-intensity aerobic exercise per week (brisk walking, cycling, swimming at a pace where conversation is possible but not easy), plus two sessions of resistance training targeting major muscle groups. This combination addresses mitochondrial capacity (aerobic work) and the size of your metabolic engine (resistance work). If you are new to exercise, even daily walking — the kind explored in detail in our guide to triggering autophagy through fasting and exercise — activates many of the same cellular cleanup pathways that support fuel switching.
Dietary strategies that retrain your metabolism
Diet affects metabolic flexibility, but the relationship is more complex than popular nutrition advice suggests. The data does not point to one "best" diet. Rather, certain eating patterns train specific aspects of fuel switching.
Time-restricted eating and intermittent fasting. Extending overnight fasting periods (12-16 hours) forces a metabolic transition from carbohydrate dependence to fat oxidation. Healthy people show a clear RQ decline during overnight fasting — dropping from around 0.89 after the evening meal to 0.80 or lower by morning. Metabolically inflexible individuals show a blunted version of this drop, as documented in Tokuyama and colleagues' whole-room calorimetry study. Regularly practicing time-restricted eating provides repeated "training" for this overnight switch.
Macronutrient cycling. Alternating between higher-carbohydrate and higher-fat meals (across different days, not within the same meal) challenges the metabolic machinery to adapt. The Galgani review notes that after one to a few days on a changed macronutrient ratio, a metabolically flexible person's 24-hour RQ will adjust to match the dietary food quotient. Someone with impaired flexibility takes longer to make this adjustment — or never fully makes it.
Fiber and resistant starch. Slowly digestible carbohydrates produce a more gradual glucose and insulin response, reducing the sharp malonyl-CoA spikes that completely shut down fat oxidation after meals. A 2025 study in the European Journal of Nutrition found that mice consuming slowly digestible carbohydrates showed improved measures of metabolic substrate utilization at the whole-body level. Foods like oats, legumes, and cooled potatoes — covered in depth in our article on resistant starch and blood sugar metabolism — help smooth out the glucose-fat transition.
| Dietary approach | Effect on metabolic flexibility | What the research says |
|---|---|---|
| Time-restricted eating (12-16h fast) | Trains the fasting-to-fed transition | Repeated overnight fasting exposure promotes fat oxidation capacity |
| Higher protein intake | Supports muscle mass (the metabolic engine) | Muscle mass independently predicts metabolic flexibility |
| Fiber-rich, slow-digesting carbs | Reduces sharp insulin spikes that lock out fat oxidation | Slow-digesting carbs improved whole-body substrate utilization (2025 mouse study) |
| Periodic higher-fat days | Challenges fat oxidation pathways to stay active | 3-day high-fat diet increased FAO by 27% in lean subjects (Battaglia 2012) |
| Avoiding chronic overfeeding | Prevents ectopic lipid accumulation that causes lipotoxicity | Chronic surplus leads to ceramide buildup and insulin signaling impairment |
One counterintuitive finding: a randomized controlled trial by Lundsgaard and colleagues found that a six-week healthy diet (rich in fruits, vegetables, legumes, fiber, nuts, and fatty fish) did not improve metabolic flexibility compared to a Western-style diet when measured by standard ΔRQ during a clamp — despite improving other health markers. The takeaway: diet alone, without changes in physical activity or energy balance, may not be sufficient to measurably restore metabolic flexibility. The strongest evidence favors combining dietary changes with exercise.
Sleep, stress, and circadian timing
Sleep is not just recovery time — it is a metabolic testing ground. During a typical night, your body transitions through approximately seven hours of fasting. Fuel oxidation should shift steadily toward fat burning as the night progresses.
But research from the University of Tsukuba using high-resolution whole-room calorimetry uncovered something unexpected: RQ during sleep does not simply decline. It follows a U-shaped curve. RQ falls after bedtime, reaches a nadir (its lowest point, around midnight to 2 a.m.), and then starts rising again before awakening — even though you have not eaten. In metabolically inflexible men, this overnight RQ decline was blunted. Their bodies could not fully shift to fat burning during sleep.

The researchers found that the time course of RQ during sleep closely tracked core body temperature (correlation r = 0.913), suggesting that both rhythms are governed by the same circadian pacemaker. Age mattered: in subjects just ten years older, the overnight RQ decrease was blunted, independent of BMI or body fat percentage. Women reached their RQ nadir earlier than men, paralleling known sex differences in circadian body temperature patterns.
This circadian connection runs deeper than sleep timing. Bass and colleagues (Nature Reviews, 2016) showed that disrupted circadian clocks impair nearly every aspect of metabolic homeostasis — from insulin secretion timing to adipose tissue lipid cycling. Shift workers, people with irregular sleep schedules, and those who eat late at night all show markers of reduced metabolic flexibility. The molecular metabolic machinery has its own clock, and ignoring it comes at a cost.
Consistent sleep and wake times matter more than most people realize for metabolic health. Late-night eating compresses the overnight fasting window and prevents the full RQ decline that characterizes healthy sleep metabolism. Addressing sleep quality may be as important as exercise for people whose metabolic flexibility has declined with age — a connection that ties into broader strategies for supporting sleep quality through nutritional approaches.
Myths vs. evidence
| Claim | Reality |
|---|---|
| "Metabolic inflexibility is caused by bad mitochondria" | The relationship is not that simple. Galgani et al. (2008) showed that people with frank mitochondrial dysfunction (myopathies) actually have increased fuel oxidation. Mitochondrial content matters, but sedentary behavior may explain the deficits attributed to "dysfunction." Exercise rapidly reverses these decrements. |
| "You need to eat keto to become metabolically flexible" | Ketogenic diets do train fat oxidation pathways. But metabolic flexibility means switching both ways — to fat and to glucose. Chronic very-low-carb eating can actually impair glucose oxidation capacity, creating inflexibility in the opposite direction. |
| "If you can fast 16 hours comfortably, you're metabolically flexible" | Subjective comfort during fasting does not equal measured metabolic flexibility. Whole-room calorimetry studies show that some people who tolerate fasting still have blunted RQ shifts. Actual measurement requires indirect calorimetry or at minimum continuous glucose monitoring during controlled conditions. |
| "Thin people are always metabolically flexible" | Not necessarily. Research shows that offspring of parents with type 2 diabetes had impaired fat oxidation responses to high-fat meals despite normal weight and normal insulin sensitivity, suggesting a genetic component to metabolic inflexibility that operates independently of body composition. |
| "Supplements can fix metabolic inflexibility" | No supplement has been shown to directly restore metabolic flexibility. However, compounds that support mitochondrial function — like those discussed in research on urolithin A and mitochondrial health — may support the cellular machinery involved in fuel switching when combined with exercise and dietary changes. |
Frequently Asked Questions
How do I know if I am metabolically inflexible?
The gold standard is indirect calorimetry, which measures your RQ under fasting and fed conditions. Outside a lab, certain patterns suggest reduced metabolic flexibility: difficulty going more than a few hours without eating, persistent energy crashes after meals, difficulty losing weight despite calorie restriction, and reliance on frequent carbohydrate intake to maintain energy. Continuous glucose monitors can also provide indirect clues — large glucose spikes and slow returns to baseline suggest poor insulin sensitivity, which is closely linked to metabolic inflexibility.
Can you improve metabolic flexibility at any age?
Yes. While the Tokuyama sleep study showed age-related declines in overnight RQ shifting, the exercise intervention data is encouraging at all ages. The Diabetologia trial included middle-aged adults with type 2 diabetes who improved their post-meal metabolic flexibility after ten weeks of aerobic training. Shoemaker et al. (2023) also found that even older adults with sarcopenia showed differences in metabolic flexibility that correlated with exercise capacity, suggesting that building and maintaining muscle mass preserves fuel-switching ability. Starting exercise at any age produces measurable mitochondrial improvements within days.
Is metabolic flexibility the same as being "fat adapted"?
Not quite. Fat adaptation refers to training your body to rely more heavily on fat as fuel, often through ketogenic diets or prolonged endurance training. Metabolic flexibility is broader — it is the ability to switch efficiently in both directions, from fat to glucose and back. Someone who is fat adapted but has lost the ability to efficiently oxidize glucose after a carbohydrate meal is not metabolically flexible. The goal is range, not specialization.
Does metabolic flexibility affect weight loss?
It can. Metabolically inflexible individuals tend to stay in positive fat balance when they eat high-fat foods because they cannot increase fat oxidation to match fat intake. Over time, this promotes ectopic fat accumulation — fat stored inside muscles and organs rather than in subcutaneous fat tissue. Improving metabolic flexibility through exercise may make weight management easier because the body can actually burn the fuel types it receives rather than defaulting to storage.
What is the single most effective intervention for improving metabolic flexibility?
Exercise, and specifically aerobic exercise combined with resistance training. The evidence points one direction: exercise improves every measured aspect of metabolic flexibility — fasting fat oxidation, post-meal glucose handling, sleep RQ patterns, and mitochondrial capacity. No dietary intervention studied in isolation has matched the breadth of exercise effects on fuel switching. Diet and sleep modifications enhance the results, but exercise is the foundation.
Related Articles
- Resistant Starch: The Cheap Gut Health Hack for Blood Sugar and Metabolism — How slowly digestible carbohydrates affect glucose and insulin responses.
- Autophagy: How to Trigger It Through Fasting, Exercise, and Diet — The cellular cleanup process that overlaps with fasting-induced metabolic switching.
- Urolithin A: The Mitochondrial Anti-Aging Supplement Most People Haven't Heard Of — Supporting mitochondrial health, a key driver of metabolic flexibility.
- Biological Age Testing: How to Measure and Improve Your Pace of Aging — Metabolic flexibility is one marker of biological versus chronological age.
- Glycine: The Amino Acid That Improves Sleep, Collagen, and Longevity — Sleep quality supports the circadian rhythms that govern overnight fuel switching.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.