What Is Butyrate and Where Does It Come From?
Your large intestine hosts trillions of bacteria, and some of the most important work they do is invisible: fermenting dietary fiber into short-chain fatty acids (SCFAs). Three SCFAs dominate this process — acetate, propionate, and butyrate — with acetate accounting for roughly 60% of total production, propionate about 25%, and butyrate approximately 15%. That 15% share sounds small, but butyrate has an outsized biological impact relative to its concentration.
Butyrate is a four-carbon fatty acid produced primarily by gram-positive anaerobic bacteria, including Faecalibacterium prausnitzii, Eubacterium rectale, and species within the Roseburia and Clostridium genera. These microbes thrive on fermentable fiber — the portions of plant foods your own digestive enzymes cannot break down. When fiber reaches the colon undigested, these bacteria convert it into butyrate through a process called saccharolytic fermentation.
Butyrate influences multiple biological systems at once. It serves as the primary fuel source for the cells lining your colon, it crosses into circulation to modulate immune signaling, and emerging research shows it reaches the brain where it may alter gene expression in ways that protect against neurodegeneration. Researchers at Weill Medical College of Cornell University have described butyrate as a "multi-functional molecule" capable of targeting multiple pathways simultaneously — something few pharmaceutical compounds achieve.
Quick Fact: Butyrate operates through at least three distinct mechanisms: it inhibits histone deacetylase enzymes (changing which genes get expressed), activates G protein-coupled receptors on immune cells, and provides direct metabolic fuel to colonocytes. No other gut metabolite covers this much biological ground.
Your Colon Runs on Butyrate
Most cells in your body depend on glucose for energy. Colonocytes — the epithelial cells lining your large intestine — are different. They derive approximately 70% of their energy from butyrate oxidation, making them among the most microbiome-dependent cells in the human body. Remove that microbial fuel supply, and the consequences are stark.
A landmark study published in Cell Metabolism demonstrated exactly what happens when colonocytes lose access to butyrate. Researchers at the University of North Carolina compared colon tissue from germ-free mice (raised without any gut bacteria) to tissue from conventionally colonized animals. The germ-free colons showed a 16-fold decrease in NADH/NAD+ ratios, a 56% reduction in ATP levels, and a 70% drop in oxidative phosphorylation. No other organ tested — not the liver, heart, kidney, or testis — showed comparable deficits. The colon was uniquely starved.
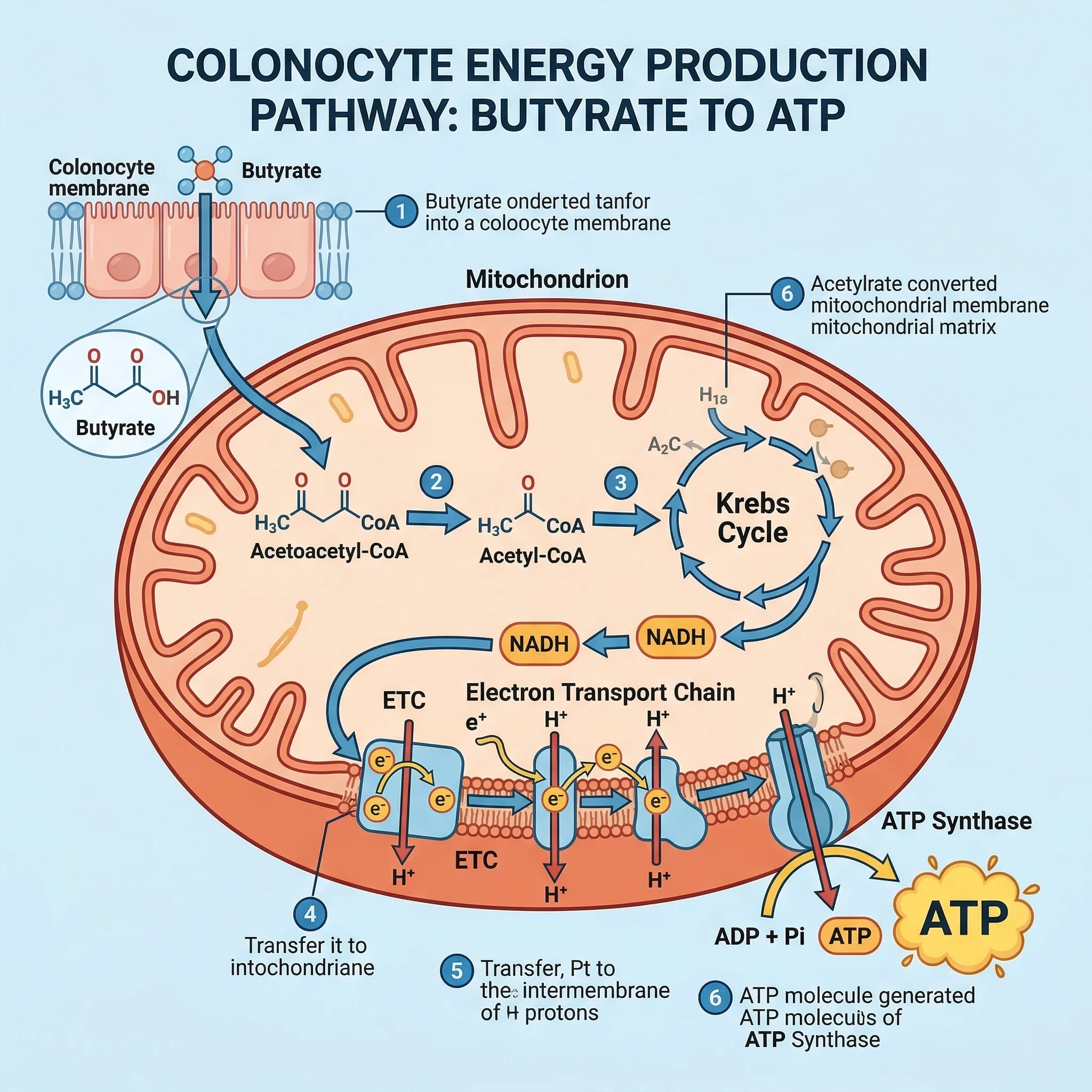
Even more striking: those energy-starved colonocytes triggered massive autophagy — the cell's emergency self-recycling program. Transmission electron microscopy revealed autophagosomes in roughly one of every germ-free colonocyte, compared to one in every thirty cells in normal mice. The cells were digesting their own components to stay alive.
When researchers added butyrate back to those germ-free colonocytes, mitochondrial respiration recovered. The rescue depended specifically on fatty acid oxidation — blocking that pathway with the drug etomoxir eliminated the benefit entirely. An HDAC inhibitor drug, by contrast, had no effect on energy recovery. This confirmed that butyrate's role as cellular fuel is distinct from and more fundamental than its epigenetic signaling activity.
| Metric | Normal Mice | Germ-Free Mice | Change |
|---|---|---|---|
| NADH/NAD+ ratio | Baseline | 16-fold decrease | Severe energy deficit |
| ATP levels | Baseline | 56% reduction | Cellular energy crisis |
| Oxidative phosphorylation | Baseline | 70% decrease | Mitochondrial shutdown |
| Fatty acid oxidation | Baseline | 31% reduction | Metabolic compensation |
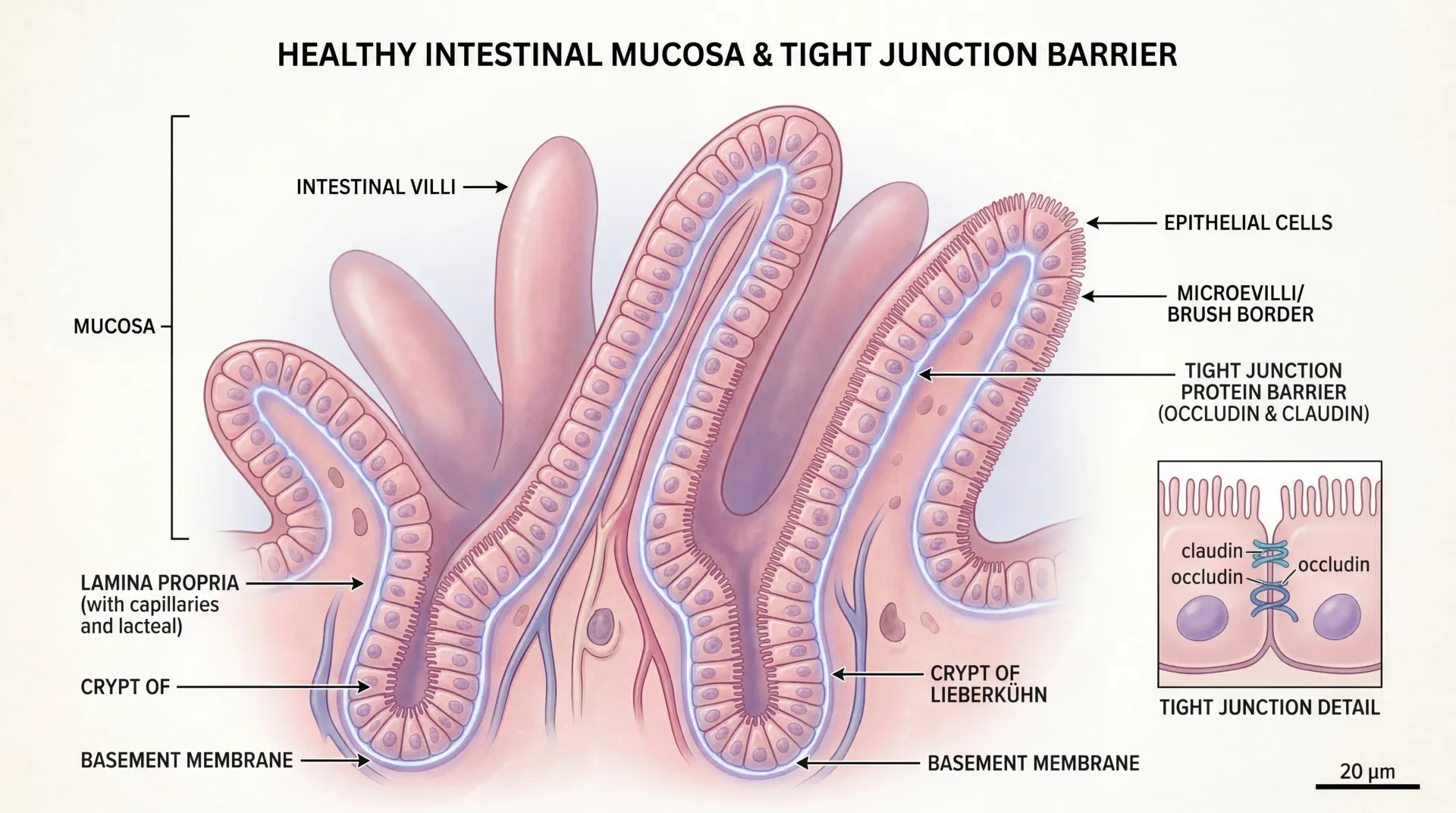
Beyond raw energy, butyrate maintains the physical integrity of the intestinal barrier. It upregulates tight junction proteins — specifically increasing claudin-1, claudin-3, and claudin-4 expression while reducing claudin-2 (a "leaky" claudin associated with increased permeability). It stimulates mucin-2 (MUC2) production, thickening the protective mucus layer that keeps bacteria from contacting epithelial cells directly. And it boosts antimicrobial peptide production, including the cathelicidin LL-37, through histone acetylation at the gene promoter.
For anyone dealing with leaky gut symptoms, this matters because butyrate addresses the problem at multiple levels simultaneously — energy for cell turnover, structural proteins for barrier tightness, and mucus for physical separation from luminal bacteria.
A feedback loop reinforces this relationship. Research from the University of Tennessee published in iScience (2024) found that colonocytes consuming butyrate actively promote the growth of butyrate-producing bacteria. The mechanism involves oxygen gradients: when colonocytes oxidize butyrate efficiently, they consume oxygen and create hypoxic conditions at the mucosal surface — exactly the environment that strict anaerobic butyrate producers need to thrive. When this cycle breaks down (as in colorectal cancer or ulcerative colitis, where colonocytes shift to glycolysis), butyrate-producing bacteria decline, and even high-fiber diets fail to restore them.
The Gut-Brain Connection: Butyrate Beyond Digestion
The notion that a fatty acid produced in the colon could influence brain function sounds implausible until you consider the anatomy. The gut-brain axis — a bidirectional communication highway linking the enteric nervous system to the central nervous system — gives microbial metabolites like butyrate several routes into the brain: the vagus nerve, immune signaling molecules in the bloodstream, and direct passage across the blood-brain barrier.
One of the most cited findings in this field comes from research showing that germ-free mice have increased blood-brain barrier permeability compared to conventionally colonized animals. Their BBB was leaky — and treatment with either butyrate-producing bacteria or sodium butyrate directly restored tight junction protein expression (claudin-5 and occludin) in brain endothelial cells. The BBB tightened back up. This suggests that the structural integrity of the barrier separating your blood from your brain tissue depends, at least in part, on metabolites produced by bacteria living in your colon.
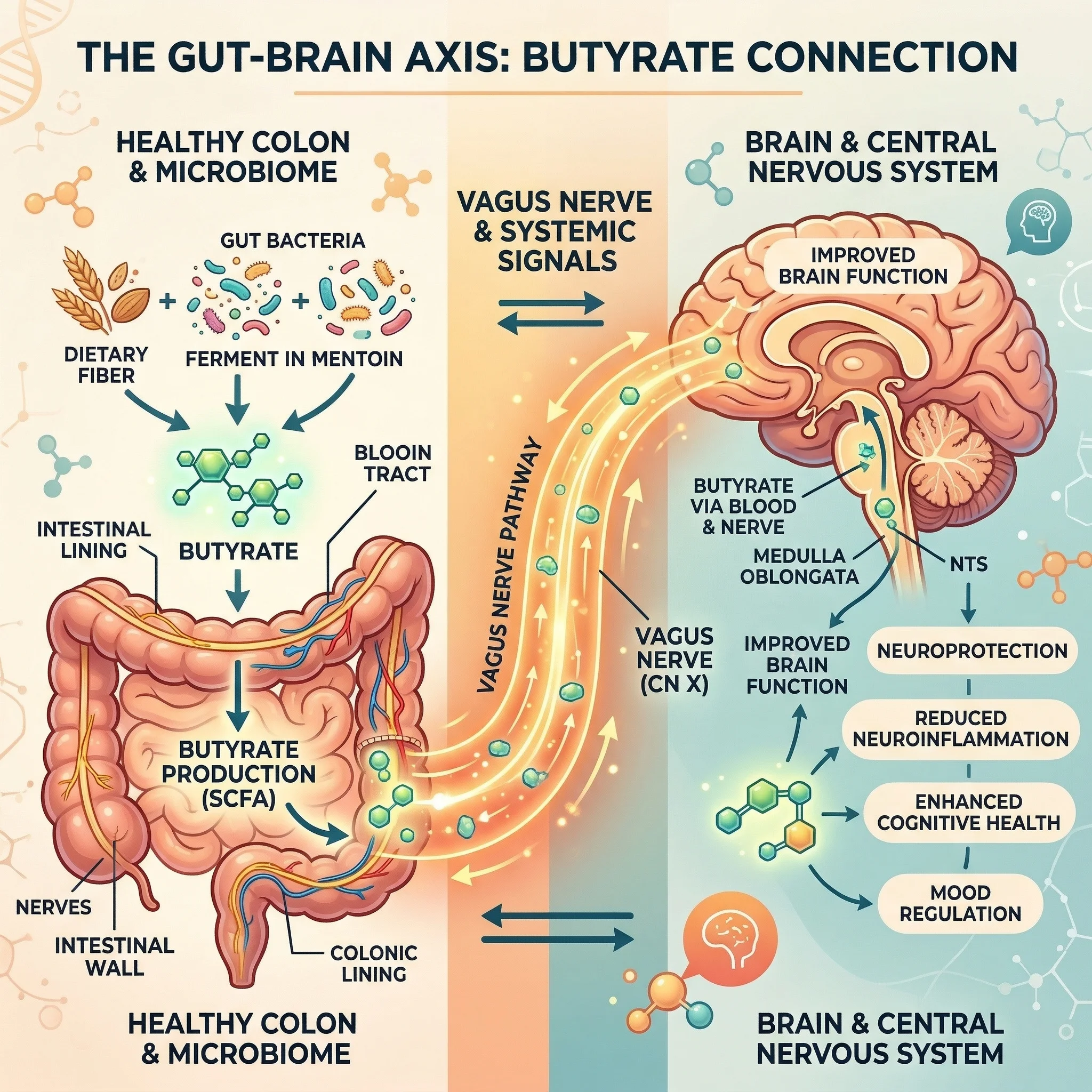
Butyrate's brain effects operate largely through HDAC inhibition — the same epigenetic mechanism it uses throughout the body, but with particularly significant neurological consequences. By inhibiting histone deacetylases, butyrate loosens the chromatin packaging around DNA, allowing transcription of genes that would otherwise remain silenced. In the brain, this translates to increased production of neurotrophic factors — specifically BDNF (brain-derived neurotrophic factor), GDNF (glial cell-derived neurotrophic factor), and NGF (nerve growth factor). These proteins support neuronal survival, growth, and synaptic plasticity.
Animal studies have demonstrated protective effects across several neurodegenerative conditions. In Huntington's disease models, butyrate prevented neuronal cell death and extended lifespan. In Parkinson's disease models, it protected dopaminergic neurons from toxicity. In Alzheimer's disease models, it restored histone acetylation patterns and improved expression of genes associated with learning and memory. A 2024 systematic review and meta-analysis of nine animal studies confirmed that butyrate administration substantially elevates BDNF levels and reduces cerebral infarct volume following ischemic stroke.
The human evidence, while still early, is moving in the same direction. A 2025 cross-sectional analysis of 2,078 adults aged 60 and older from the National Health and Nutrition Examination Survey found that those in the highest quartile of dietary butyrate intake scored significantly better on cognitive assessments — including processing speed (DSST), verbal fluency (AFT), and memory (CERAD) — compared to those in the lowest quartile. The researchers identified a nonlinear dose-response relationship, with an inflection point around 1.73 grams per day of dietary butyrate.
| Neurological Condition | Butyrate's Observed Effect | Evidence Level |
|---|---|---|
| Alzheimer's Disease | Restored histone acetylation, improved learning genes | Animal studies |
| Parkinson's Disease | Protected dopaminergic neurons from toxicity | Animal studies |
| Huntington's Disease | Prevented neuronal cell death, extended lifespan | Animal studies |
| Ischemic Stroke | Reduced infarct volume, elevated BDNF | Meta-analysis (9 studies) |
| Cognitive Decline (aging) | Higher intake linked to better test scores | Human cross-sectional (n=2,078) |
Butyrate also activates a receptor called GPR109a that is expressed on microglia — the brain's resident immune cells. Researchers have found this receptor is upregulated in the substantia nigra of Parkinson's disease patients, where it colocalizes with microglia. Activation of GPR109a produces anti-inflammatory effects, which may help explain why butyrate-producing bacteria and high-fiber diets are associated with reduced neuroinflammation in animal models.
How Butyrate Tames Chronic Inflammation
Chronic low-grade inflammation underlies many of the conditions butyrate appears to protect against — from inflammatory bowel disease to neurodegeneration. Butyrate's anti-inflammatory action centers on a transcription factor called NF-κB, which acts as a master switch for inflammatory gene expression. When NF-κB is activated, it drives production of pro-inflammatory cytokines including IL-1β, TNF-α, IL-6, IL-8, and COX-2. Butyrate inhibits NF-κB activation, simultaneously reducing this entire inflammatory cascade while upregulating anti-inflammatory mediators like IL-10 and TGF-β.
This dual action — suppressing inflammatory signals while boosting anti-inflammatory ones — is why butyrate shows therapeutic potential in conditions where the immune system is overactive. Clinical evidence backs this up in inflammatory bowel disease. One trial found that adding 60 grams of oat bran (providing 20 grams of dietary fiber) daily to the diet of patients with quiescent ulcerative colitis produced a significant increase in fecal butyrate concentration and meaningful improvement in abdominal symptoms within just four weeks. A separate multicenter trial demonstrated that combining topical 5-aminosalicylic acid with sodium butyrate improved disease activity scores more significantly than 5-ASA treatment alone.
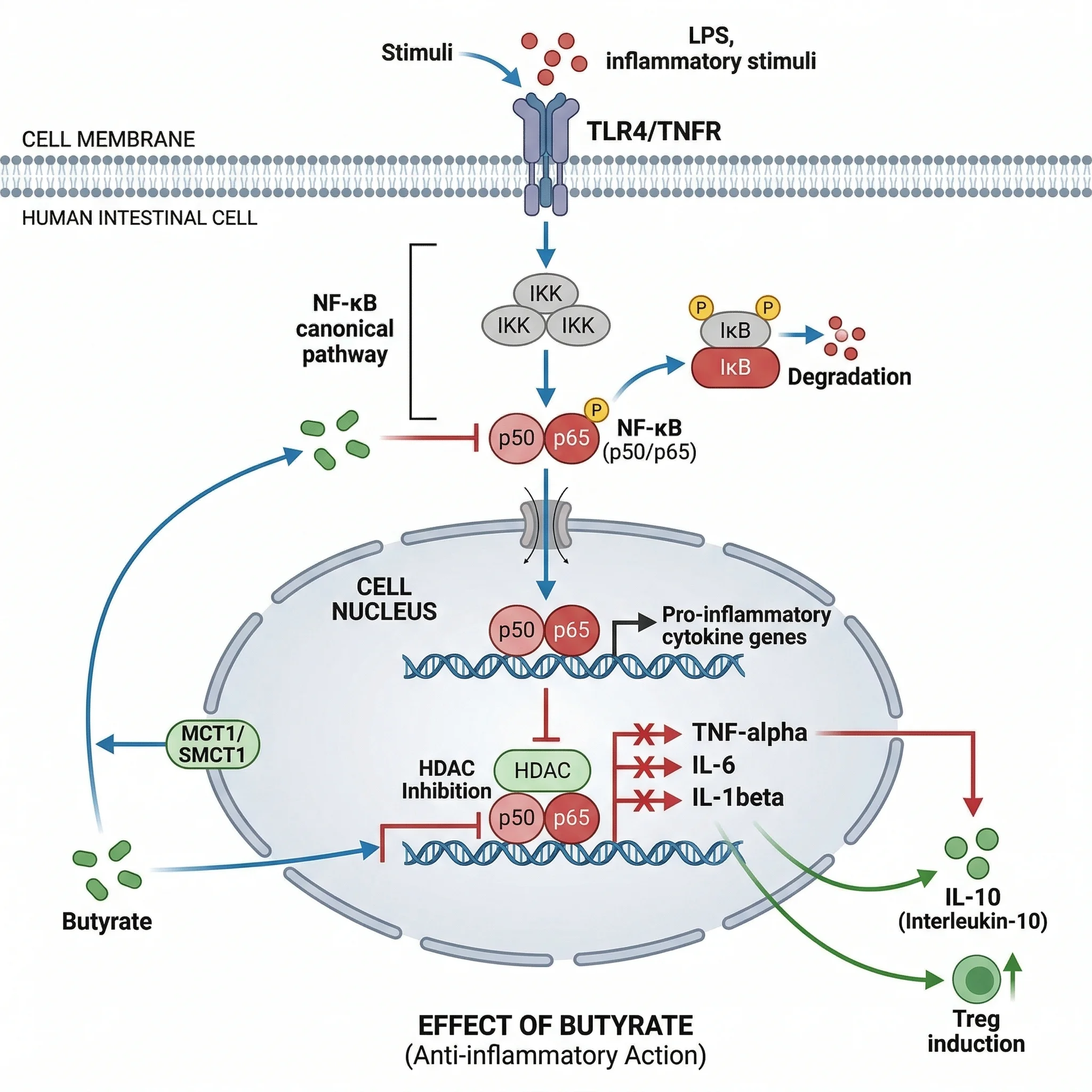
Butyrate also contributes to oxidative stress defense. Research in healthy human volunteers found that locally administered butyrate increased levels of glutathione (GSH) — the body's primary intracellular antioxidant — while decreasing markers of reactive oxygen species production. This antioxidant effect compounds with NF-κB inhibition to create a broadly protective anti-inflammatory environment in the gut.
| Inflammatory Marker | Butyrate's Effect | Pathway |
|---|---|---|
| TNF-α | Decreased | NF-κB inhibition |
| IL-1β | Decreased | NF-κB inhibition |
| IL-6 | Decreased | NF-κB inhibition |
| IL-8 | Decreased | NF-κB inhibition |
| COX-2 | Decreased | NF-κB inhibition |
| IL-10 | Increased | PPAR-γ upregulation |
| TGF-β | Increased | Regulatory T cell differentiation |
| Glutathione (GSH) | Increased | Antioxidant defense |
The connection between gut inflammation and systemic health extends further than many people realize. Chronic intestinal inflammation can increase intestinal permeability, allowing bacterial products like lipopolysaccharide (LPS) to enter the bloodstream and trigger immune responses throughout the body — including the brain. For those working to reduce systemic inflammation, combining butyrate-boosting dietary strategies with other evidence-based approaches like curcumin from turmeric may offer complementary pathways to the same goal.
One counterintuitive finding deserves mention: butyrate's relationship with cancer cells. Normal colonocytes metabolize butyrate through mitochondrial beta-oxidation, using it as fuel. Cancer cells, however, have shifted their metabolism toward glycolysis (the Warburg effect) and cannot efficiently oxidize butyrate. The result is that unused butyrate accumulates in cancer cell nuclei and inhibits HDAC enzymes, triggering cell cycle arrest and apoptosis. Researchers call this the "butyrate paradox" — the same molecule that nourishes healthy colon cells selectively kills cancerous ones. It upregulates pro-apoptotic BAK while downregulating anti-apoptotic Bcl-xL, and it reactivates the tumor-suppressor receptor GPR109a that is silenced by DNA methylation in cancer cells.
Foods That Fuel Your Butyrate Production
Since the vast majority of butyrate in your colon is produced by bacterial fermentation of dietary fiber, the most reliable way to increase butyrate levels is to feed those bacteria what they need. Not all fibers are equal in this regard — the type of carbohydrate determines how much butyrate gets produced.
Resistant starch stands out as the most efficient butyrate precursor. Research comparing different fermentable carbohydrates found that resistant starch yielded 0.55% butyric acid — considerably higher than oat bran (0.38%), lactulose (0.36%), or cellulose (0.33%). Resistant starch forms when starchy foods are cooked and then cooled: cold potatoes, overnight oats, cooled rice, and green (unripe) bananas are all practical sources. Reheating does not fully reverse the resistant starch formation, so leftover potato salad or day-old rice remain good options.
| Food Category | Top Sources | Why It Works |
|---|---|---|
| Resistant starch | Cooled potatoes, green bananas, overnight oats, cooled rice | Highest butyrate yield of all fiber types |
| Allium vegetables | Garlic, onions, leeks | Rich in inulin and fructooligosaccharides (prebiotic fibers) |
| Legumes | Chickpeas, lentils, green peas, black beans | High in both resistant starch and soluble fiber |
| Cruciferous vegetables | Broccoli, artichokes, asparagus | Mixed fiber types that support diverse SCFA production |
| Whole grains | Oat bran, barley, rye | Beta-glucans and arabinoxylan fermentation |
| Fruits | Apples, pears, kiwis, raspberries | Pectin and cellulose support gradual fermentation |
The USDA recommends 25 grams of fiber daily for women and 35 grams for men, but most Americans fall well short of these targets. Building fiber intake gradually matters — adding too much too quickly can cause bloating and gas as bacterial populations adjust. Increasing by about 5 grams per week while maintaining adequate hydration gives your microbiome time to adapt. Proper electrolyte and hydration balance becomes especially important during fiber increases, as fermentation draws water into the colon.
Butter is sometimes mentioned as a direct dietary source of butyrate, and it does contain 3-4% butyric acid as tributyrin. However, this butyrate is absorbed in the upper digestive tract and largely metabolized by the liver — very little reaches the colon where it is most needed. Fiber fermentation remains the only reliable way to deliver butyrate to colonocytes at meaningful concentrations.
One important finding from recent research: high-fat diets decrease butyrate production and increase inflammation, while fiber supplementation reverses these effects. This means that what you eat alongside fiber matters — a high-fiber diet paired with excessive saturated fat may not produce the butyrate benefits you would expect.

Supplements vs. Fiber: Which Butyrate Strategy Works?
Butyrate supplements — typically sold as sodium butyrate, calcium-magnesium butyrate, or tributyrin capsules — offer a direct dose of the molecule without relying on bacterial fermentation. But oral butyrate delivery is harder than it sounds.
The core challenge with oral butyrate supplements is delivery. Butyrate absorbed in the upper GI tract gets metabolized by the liver through first-pass metabolism, and very little reaches the colon. Enteric-coated formulations attempt to solve this by delaying release until the capsule reaches the lower intestine, but absorption kinetics vary widely between products and individuals. Tributyrin (a prodrug form found in butter) shows somewhat better colonic delivery — one study found it raised portal vein butyrate concentrations to 2.4 mM within one hour in rat models — but human data on colonic delivery from oral supplements remains limited.
Concentration is also a concern. Research has identified a clear dose-response boundary: butyrate supports intestinal barrier function at concentrations of 2 mM or below, but may actually disrupt barrier integrity and induce cell death at concentrations of 5-8 mM. With fiber fermentation, butyrate production ramps up gradually as bacteria process fiber throughout the day, maintaining physiological concentrations. Supplement boluses risk exceeding that threshold locally, especially if enteric coating releases the full dose in a concentrated area.
Myth vs. Fact: "Butyrate supplements are a shortcut to the same benefits as high-fiber eating." In reality, fiber fermentation provides steady-state butyrate delivery at safe concentrations, feeds diverse bacterial populations, produces other beneficial SCFAs (acetate, propionate), and maintains the oxygen gradients that butyrate-producing bacteria need to thrive. Supplements deliver the molecule but miss the ecosystem.
The fiber advantage extends beyond butyrate delivery itself. When you eat resistant starch or other fermentable fibers, you support the growth of butyrate-producing bacteria — which then continue producing butyrate between meals. Research from the University of Tennessee confirms that the prebiotic effect of high-fiber diets depends on functional host cell metabolism of butyrate, creating a virtuous cycle where eating fiber grows the bacteria that make more butyrate, which keeps colonocytes healthy enough to continue supporting those bacteria.
Supplements do have a place in specific clinical contexts. Butyrate enemas have shown benefit in ulcerative colitis patients, and clinicians sometimes use rectal butyrate at concentrations around 100 mM — comparable to physiological levels in the colon after a high-fiber meal. For irritable bowel syndrome, studies have shown that sodium butyrate at physiological concentrations blocks visceral pain through AMPK and PPAR-γ dependent pathways. These are supervised clinical applications, not general wellness supplementation.
For most people, the practical recommendation is straightforward: prioritize diverse fiber-rich foods over butyrate supplements. If you do supplement, look for enteric-coated or tributyrin formulations, start with lower doses, and use them as additions to — not replacements for — a high-fiber diet. Avoid butyric acid supplements during pregnancy or breastfeeding, as safety data for these populations is limited.
Frequently Asked Questions
How long does it take to increase butyrate levels through diet?
Measurable changes in fecal butyrate concentration can occur within days of increasing fermentable fiber intake. A clinical trial found that adding 60 grams of oat bran daily produced significant butyrate increases and symptom improvement in ulcerative colitis patients within four weeks. However, building a diverse butyrate-producing bacterial population takes longer — expect gradual improvements over 2-3 months of consistent high-fiber eating.
Can you get enough butyrate from butter alone?
Butter contains 3-4% butyric acid, but this is absorbed in the upper digestive tract and metabolized by the liver before reaching the colon. The butyrate your colonocytes need comes almost entirely from bacterial fermentation of dietary fiber in the large intestine. Butter is not a meaningful source of colonic butyrate.
Is butyrate safe for everyone?
Butyrate produced naturally through fiber fermentation is safe for the general population at normal dietary fiber intakes. Supplemental butyrate requires more caution — concentrated doses above 5 mM can disrupt intestinal barrier function rather than support it. People with existing gut conditions should consult their healthcare provider before starting butyrate supplements, and supplementation is not recommended during pregnancy or breastfeeding.
Does butyrate actually reach the brain?
Research shows that less than 0.006% of colonic butyrate enters the brain directly. However, butyrate influences brain function through multiple indirect pathways: it activates vagal afferent neurons that signal to the central nervous system, it modulates systemic inflammatory markers that cross the blood-brain barrier, and it supports BBB integrity itself. The small amount that does cross the BBB can still inhibit HDAC enzymes and alter gene expression in brain tissue.
What is the "butyrate paradox" in cancer research?
Healthy colonocytes metabolize butyrate through mitochondrial beta-oxidation, using it as their primary energy source. Cancer cells have shifted to glycolysis and cannot efficiently process butyrate. Unmetabolized butyrate accumulates in cancer cell nuclei, where it inhibits HDAC enzymes and triggers cell cycle arrest and apoptosis. The same molecule that feeds healthy cells selectively kills cancerous ones.
Related Articles
- Gut-Brain Axis: How Your Microbiome Affects Mood and Cognition — A deeper look at the communication highway between your gut bacteria and your brain.
- Leaky Gut Syndrome: Symptoms, Causes, and Evidence-Based Healing — Understanding intestinal permeability and the role of tight junction proteins in barrier function.
- Bone Broth Benefits for Gut Healing, Joints, and Skin — Another approach to supporting intestinal lining repair and gut health.
- Health Benefits of Turmeric: The Complete Evidence-Based Guide — Curcumin's anti-inflammatory pathways complement butyrate's NF-κB inhibition.
- Electrolytes Explained: Sodium, Potassium, Magnesium for Hydration — Why electrolyte balance matters when increasing dietary fiber intake.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.