Not just another adaptogen
Ashwagandha (Withania somnifera) has been part of Ayurvedic medicine for roughly 3,000 years, but Western science has only recently started taking it seriously. A joint taskforce from the World Federation of Societies of Biological Psychiatry (WFSBP) and the Canadian Network for Mood and Anxiety Treatments (CANMAT) now provisionally recommends specific ashwagandha doses for generalized anxiety disorder. For a botanical supplement, getting that kind of nod from psychiatric organizations is unusual.
The plant's roots contain steroidal lactones called withanolides, which are thought to drive most of its effects on the hypothalamic-pituitary-adrenal (HPA) axis, the body's central stress-response system. Some research also points to non-withanolide compounds, including alkaloids and saponins, as contributors. What that means in practice: ashwagandha is not a single-molecule drug. It's a complex botanical whose effects shift depending on which part of the plant is used, how it's extracted, and how much you take.
Two people buying "ashwagandha" from different brands may end up with different products. Below, I'll walk through what the clinical evidence actually supports, which extract formulations have been tested, and where the data gets thin.
KSM-66 vs Sensoril: two extracts, different chemistry
The supplement aisle treats ashwagandha as one thing. Researchers don't. Most clinical trials use one of two branded extracts, and they are not interchangeable.

| Feature | KSM-66 | Sensoril / Shoden |
|---|---|---|
| Plant part | Root only | Root and leaf |
| Withanolide concentration | ≥5% (HPLC-measured) | ≥10% (Sensoril) / ≥35% (Shoden) |
| Extraction method | "Green chemistry" — no alcohol or synthetic solvents | Hydroalcoholic or aqueous extraction |
| Typical study dose | 300-600 mg/day | 120-240 mg/day |
| Key clinical focus | Stress, cortisol, testosterone, sleep | Stress, anxiety, sleep |
| Notable trial | Chandrasekhar 2012 (cortisol -27.9%) | Lopresti 2019 (cortisol P<.001) |
KSM-66, made by Ixoreal Biomed in Hyderabad, is a full-spectrum root extract. Its extraction process avoids alcohol and synthetic solvents, and it is standardized to at least 5% withanolides by HPLC. The landmark Chandrasekhar et al. 2012 trial used KSM-66 at 600 mg/day (300 mg twice daily) in 64 adults with chronic stress and measured a 27.9% reduction in serum cortisol after 60 days, compared to 7.9% in the placebo group. KSM-66 has also been studied for testosterone, sleep, and athletic performance.
Sensoril (from Natreon Inc.) and the related Shoden extract use both root and leaf material, resulting in a different withanolide profile and a higher withanolide concentration per milligram. Because the concentration is higher, effective doses tend to be lower. The Lopresti et al. 2019 trial used Shoden at just 240 mg/day and still achieved statistically significant reductions in morning cortisol (P < .001), DHEA-S (P = .004), and Hamilton Anxiety Rating Scale scores (P = .040) over 60 days.
The practical difference matters when you're reading a supplement label. A product listing "600 mg ashwagandha" with KSM-66 and one listing "240 mg ashwagandha" with Shoden could deliver comparable withanolide loads. The dose number alone tells you almost nothing without knowing the extract type.
Quick takeaway: KSM-66 is root-only at lower withanolide concentration (higher dose needed). Sensoril and Shoden include leaf material at higher withanolide concentration (lower dose needed). Both have clinical evidence for stress and cortisol reduction. Neither has been proven superior to the other in a head-to-head trial.
The cortisol question: what 15 clinical trials found
Cortisol is the headline metric in ashwagandha research, and the data here is more consistent than for most herbal supplements. A 2025 systematic review and meta-analysis pooled 15 randomized controlled trials with a combined 873 participants and found statistically significant reductions in serum cortisol at 8 weeks of supplementation (P < 0.0001). The same meta-analysis showed significant reductions in perceived stress via the Perceived Stress Scale (P = 0.0013) and anxiety via the Hamilton Anxiety Rating Scale (P = 0.0007).
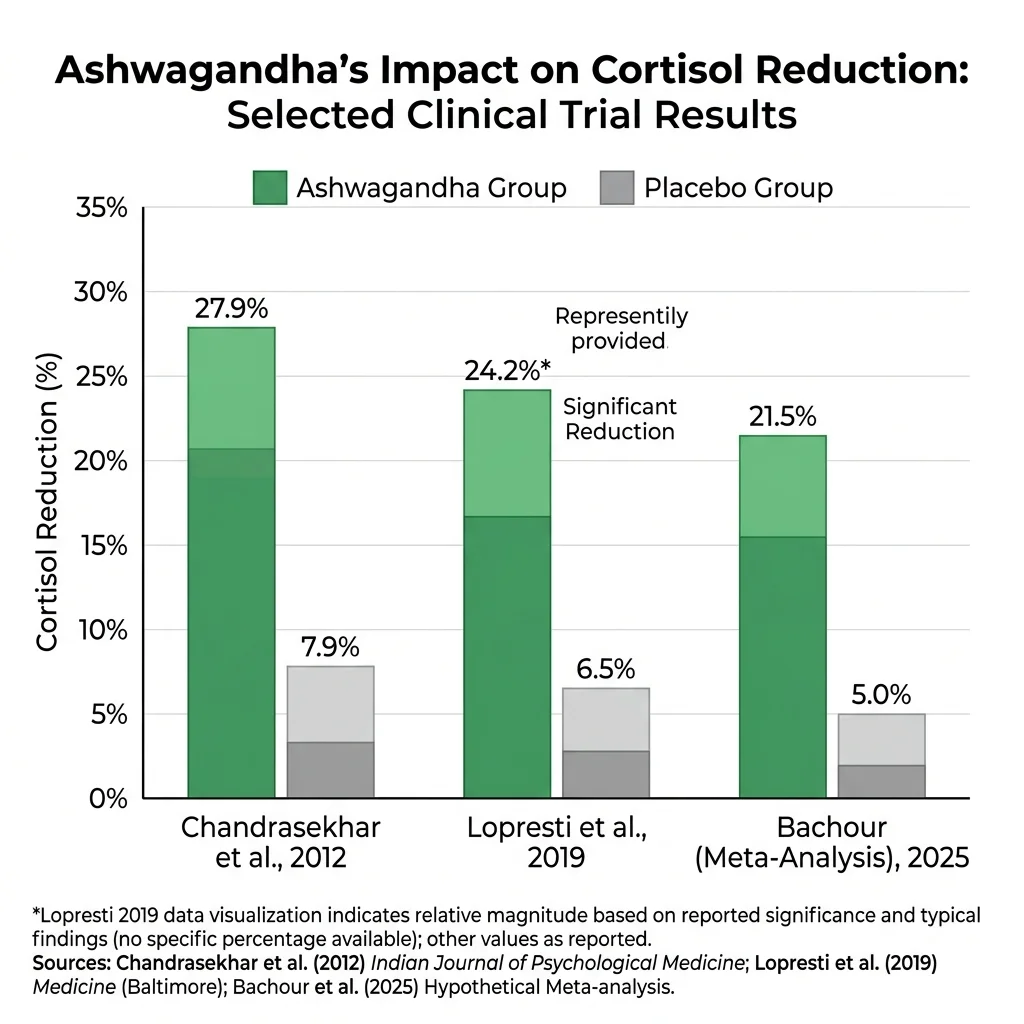
Drilling into the individual trials: in the Chandrasekhar 2012 study, KSM-66 at 600 mg/day produced a 27.9% serum cortisol reduction versus 7.9% for placebo (P = 0.0006). Across every measured stress subscale — somatic symptoms, anxiety and insomnia, social dysfunction, severe depression — the ashwagandha group showed reductions exceeding 64%, while the placebo group barely moved. PSS scores dropped 44% in the ashwagandha group versus 5.5% for placebo (P < 0.0001).
The Lopresti 2019 Shoden trial went further by tracking DHEA-S and testosterone alongside cortisol. The cortisol drop was significant (P < .001), DHEA-S decreased significantly (P = .004), and testosterone increased in men (P = .038 within group), which points to broad HPA axis modulation rather than a single hormonal pathway. A 2025 review in BMC Complementary Medicine went so far as to say ashwagandha has "the most profound effect on HPA axis" of any adaptogenic supplement studied to date.
| Trial | Extract | Dose | Duration | Cortisol result | Stress/anxiety result |
|---|---|---|---|---|---|
| Chandrasekhar 2012 | KSM-66 | 600 mg/day | 60 days | -27.9% vs -7.9% placebo | PSS -44% (P<0.0001) |
| Lopresti 2019 | Shoden | 240 mg/day | 60 days | P<.001 vs placebo | HAM-A P=.040 |
| Bachour 2025 (meta) | Various | Various | 8 weeks | P<0.0001 | PSS P=0.0013, HAM-A P=0.0007 |
The evidence is not uniformly positive, though. A 2023 trial of 120 overweight adults experiencing fatigue used the Witholytin extract (200 mg twice daily for 12 weeks) and found no significant reduction in perceived stress versus placebo, though it did reduce fatigue. This suggests the cortisol-lowering effect may depend on the specific extract formulation, participant stress levels at baseline, and dose. People already under moderate-to-high stress seem to respond more consistently than those whose primary complaint is general fatigue.
If you struggle with recognizing anxiety symptoms, the overlap between stress, anxiety, and cortisol can feel confusing. The clinical trials distinguish between them. Cortisol is a biomarker; perceived stress is self-reported; clinical anxiety is measured by instruments like the HAM-A. Ashwagandha appears to move all three, but the effect on cortisol specifically is the most reproducible finding across trials.
Ashwagandha and thyroid function
The same HPA axis modulation that lowers cortisol can also shift thyroid hormone levels. Whether that shift is helpful or harmful depends on where your thyroid function sits right now.
The most cited thyroid-specific trial is Sharma et al. 2018, which randomized 50 adults with subclinical hypothyroidism to receive either 600 mg/day of ashwagandha root extract or placebo for 8 weeks. The ashwagandha group showed significant improvements: TSH decreased, while both T3 and T4 increased compared to placebo. For people with borderline-low thyroid function who don't yet meet the threshold for medication, this is intriguing data.
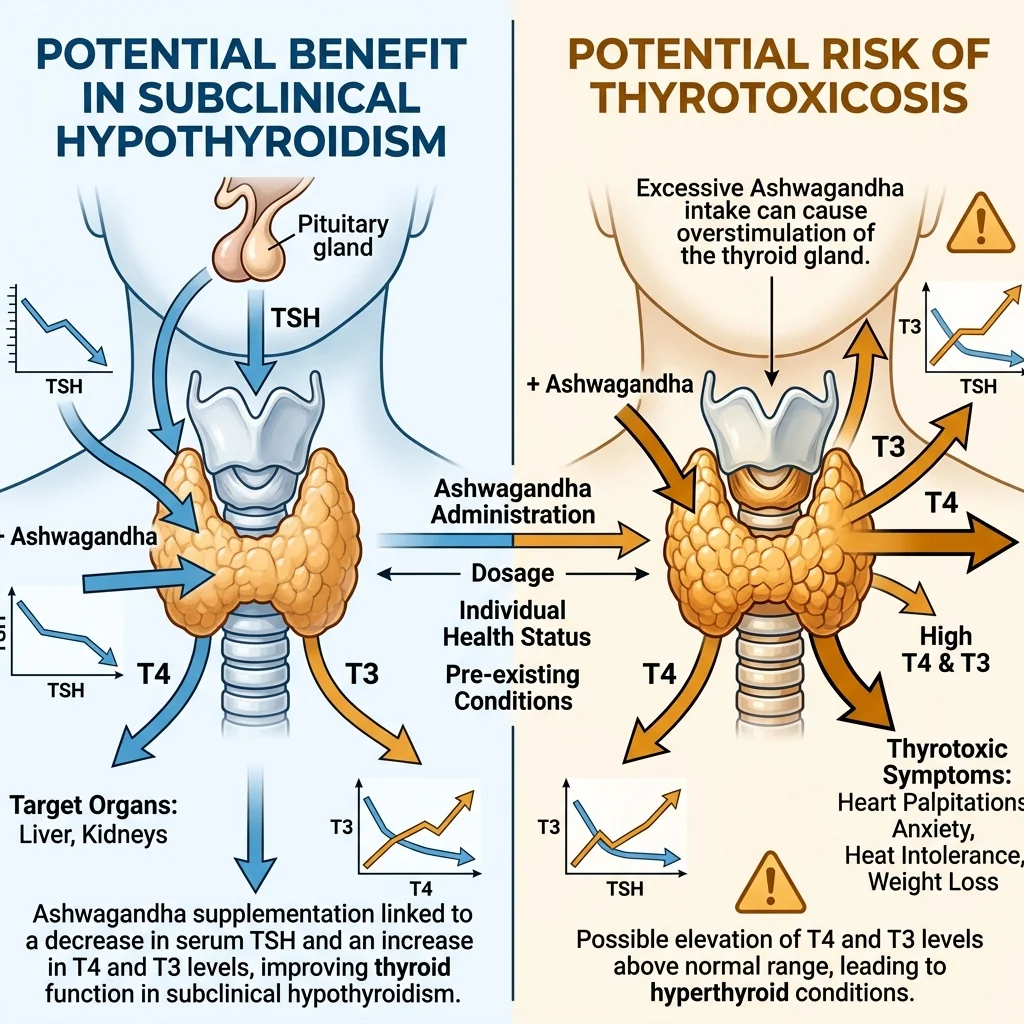
On the other side, three case reports documented thyrotoxicosis — excess thyroid hormone — in women taking ashwagandha extract. Two reports did not specify the dose; the third involved an unusually high 1,950 mg/day for over two months. All three cases resolved after the women stopped taking the supplement. The NIH's Office of Dietary Supplements notes these cases alongside the positive subclinical hypothyroidism data, underscoring that the thyroid effects go in both directions depending on the individual.
If you already take thyroid medication for Hashimoto's or hypothyroidism, ashwagandha could alter how much medication you need. This is not theoretical — the pharmacological interaction is direct enough that Denmark banned ashwagandha supplements in 2023, citing thyroid and hormonal concerns. France's ANSES agency followed in 2024 with a recommendation against ashwagandha use in people with endocrine disorders.
| Thyroid scenario | What the evidence suggests | Caution level |
|---|---|---|
| Subclinical hypothyroidism | May lower TSH, raise T3/T4 (Sharma 2018) | Discuss with endocrinologist |
| Hashimoto's on thyroid medication | Could alter medication requirements | Monitor thyroid labs closely |
| Normal thyroid function | Small T4 increases observed (Gannon 2014) | Low risk, but periodic monitoring advisable |
| Hyperthyroidism or Graves' disease | Case reports of thyrotoxicosis | Avoid |
Bottom line: Ashwagandha is not thyroid-neutral. If you have any thyroid condition — diagnosed or suspected — get thyroid labs checked before and during supplementation.
Dosing, timing, and reading the label
Clinical trials have used daily doses ranging from 120 mg to 12,000 mg, but the effective range for stress and cortisol reduction falls between 250 and 600 mg of a standardized root extract, taken for at least 4 to 8 weeks. The WFSBP/CANMAT taskforce recommendation for generalized anxiety disorder specifies 300-600 mg/day of root extract standardized to 5% withanolides.
The Mayo Clinic keeps it simple: doses up to 1,000 mg per day appear safe for at least three months, and your dose should contain at least 6 mg of withanolides. That 6 mg minimum is easy to overlook. Many budget supplements list ashwagandha on the label without specifying withanolide content or standardization percentage, which means you have no way to know what you're getting.
Other supplements that affect similar pathways, like phosphatidylserine for cortisol management, work through different mechanisms and can potentially be combined. But stacking multiple cortisol-lowering compounds without guidance is risky, particularly if you also take medications that affect the HPA axis.
Timing matters less than consistency. Most trials administered ashwagandha once or twice daily with meals. Morning dosing makes sense if your goal is cortisol reduction (morning cortisol is when the HPA axis is most active). Evening dosing may be preferable if sleep is the primary concern, given ashwagandha's GABAergic activity and documented effects on sleep latency and quality.
What to look for on the label:
- Named extract (KSM-66, Sensoril, Shoden, or equivalent branded extract)
- Withanolide percentage (minimum 1.5%, ideally 5%+ for root extracts)
- Third-party testing badge (USP, NSF, or ConsumerLab)
- Root-only vs. root-and-leaf — this determines the withanolide profile
- No proprietary blends that hide the actual ashwagandha dose
Safety profile and drug interactions
Ashwagandha is generally well tolerated in clinical trials lasting up to 12 weeks. Common side effects are mild: stomach upset, loose stools, nausea, and drowsiness. A 90-day toxicity study in rats established a no-observed-adverse-effect level (NOAEL) at 2,000 mg/kg body weight per day for KSM-66, which provides a large safety margin above typical human doses.
The more serious concern is liver injury. The Cleveland Clinic notes that ashwagandha may have negative effects on liver function, and the NIH documents multiple case reports. Five individuals taking 450-1,350 mg/day for 1 week to 4 months developed jaundice, pruritus, nausea, and elevated bilirubin. All improved after discontinuation and medical treatment. Some of these products were combination formulas, and the contents were not independently verified in every case, so attributing the injury specifically to ashwagandha versus contaminants or adulterants is difficult.
Known and suspected drug interactions include:
- Thyroid medications — can alter thyroid hormone levels (see thyroid section above)
- Sedatives and benzodiazepines — ashwagandha's GABAergic activity may amplify sedation
- Immunosuppressants — ashwagandha has immunomodulatory effects that could counteract these drugs
- Antidiabetes medications — some evidence of blood sugar effects
- Antihypertensives — potential additive blood pressure lowering
Pregnancy is a clear contraindication. While the American Herbal Pharmacopoeia has pushed back against claims that ashwagandha root is an abortifacient, enough concern exists that both the ANSES in France and regulatory bodies in Denmark advise pregnant women to avoid it entirely. Men with hormone-sensitive prostate cancer should also avoid ashwagandha, given its documented effects on testosterone levels.
If you're currently taking supplements that support liver function like milk thistle or NAC, that doesn't make ashwagandha-related liver risk disappear. Liver-supportive supplements are not antidotes. The safe approach is monitoring: get a baseline metabolic panel before starting ashwagandha and recheck after 8-12 weeks, especially if you take other supplements or medications metabolized by the liver.
Who should avoid ashwagandha: pregnant or breastfeeding women, people with hyperthyroidism or Graves' disease, anyone with active liver disease, men with hormone-sensitive prostate cancer, and people on immunosuppressant therapy.
Frequently Asked Questions
How long does ashwagandha take to work?
Most clinical trials show measurable changes in cortisol and perceived stress within 4-8 weeks of daily supplementation. Some trials have documented partial improvements as early as 2 weeks, but the full effect typically requires at least 6-8 weeks of consistent use. Sleep improvements follow a similar timeline, with benefits more pronounced at the 8-week mark and beyond.
Can I take ashwagandha with other adaptogens like tongkat ali?
There are no clinical trials directly testing ashwagandha combined with tongkat ali or other adaptogens. Both affect the HPA axis and hormonal balance, so stacking them increases the chance of unexpected hormonal shifts. If you want to try both, use them separately first to understand your individual response to each, and inform your healthcare provider.
Is ashwagandha safe to take every day long-term?
Clinical trial data supports safety for up to about 12 weeks of continuous use. Beyond that, there is no published evidence confirming or denying long-term safety. Some practitioners recommend cycling — for example, 8 weeks on, 2-4 weeks off — but this practice is based on precaution rather than data. Long-term use without monitoring is not recommended.
Does the form matter — capsules vs powder vs liquid?
Most clinical evidence comes from capsule-based standardized extracts. Powders and liquids can work, but without standardization to a specific withanolide percentage, the dose you're actually getting is uncertain. If using powder, look for products that specify the withanolide content per serving. Traditional Ayurvedic preparations sometimes combine ashwagandha powder with warm milk, and a few studies used this approach with positive results.
Should I take ashwagandha in the morning or at night?
It depends on your goal. For cortisol and stress management, morning dosing aligns with the body's natural cortisol rhythm. For sleep support, evening dosing takes advantage of ashwagandha's GABAergic activity. In studies using twice-daily dosing (such as 300 mg morning and 300 mg evening), participants got both benefits. Taking it with food reduces the chance of stomach upset.
Related Articles
- Phosphatidylserine for Memory, Cortisol, and Brain Health — Another evidence-based approach to cortisol management through supplementation.
- Tongkat Ali: Testosterone, Energy, and Stress — A different adaptogenic herb with overlapping stress and hormonal benefits.
- Thyroid Health and Hashimoto's: Symptoms, Diet, and Natural Support — Essential reading before combining ashwagandha with any thyroid condition.
- Myo-Inositol: The Supplement Going Viral for Anxiety, Sleep, and Hormonal Health — Covers another compound targeting anxiety and sleep through different mechanisms.
- Glycine: The Amino Acid That Improves Sleep, Collagen, and Longevity — A sleep-focused supplement with complementary evidence to ashwagandha.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.