What are "Ozempic babies" and why the sudden attention?
A woman with polycystic ovary syndrome spends years being told she may never conceive naturally. She starts semaglutide for weight management. Three months later, she's looking at a positive pregnancy test she never expected to see. This is happening often enough that "Ozempic baby" has become its own search term and hashtag.
The numbers are real. A recent poll found that 1 in 8 American adults have now taken a GLP-1 receptor agonist -- drugs sold under names like Ozempic, Wegovy, Mounjaro, and Zepbound. Originally approved for type 2 diabetes and obesity, these medications are producing a side effect nobody put on the label: restored fertility.
The scale of exposure is growing fast. Analysis of the US MarketScan database found that periconception exposure to GLP-1 receptor agonists quadrupled between 2012 and 2021, rising from roughly 50 to 200 per 100,000 pregnancies among commercially insured women with type 2 diabetes. That was before Wegovy's weight-loss approval sent prescriptions into the millions.
These surprise pregnancies aren't caused by one thing. Weight loss, hormonal shifts, improved insulin sensitivity, and possible drug interactions with birth control all play a role. For women who've struggled with infertility, an unplanned pregnancy on a weight-loss drug is disorienting in the best possible way. But it also raises a question: if these drugs are restoring fertility that women didn't know they'd lost, how many of those pregnancies are happening without contraception, folic acid, or any preconception planning?
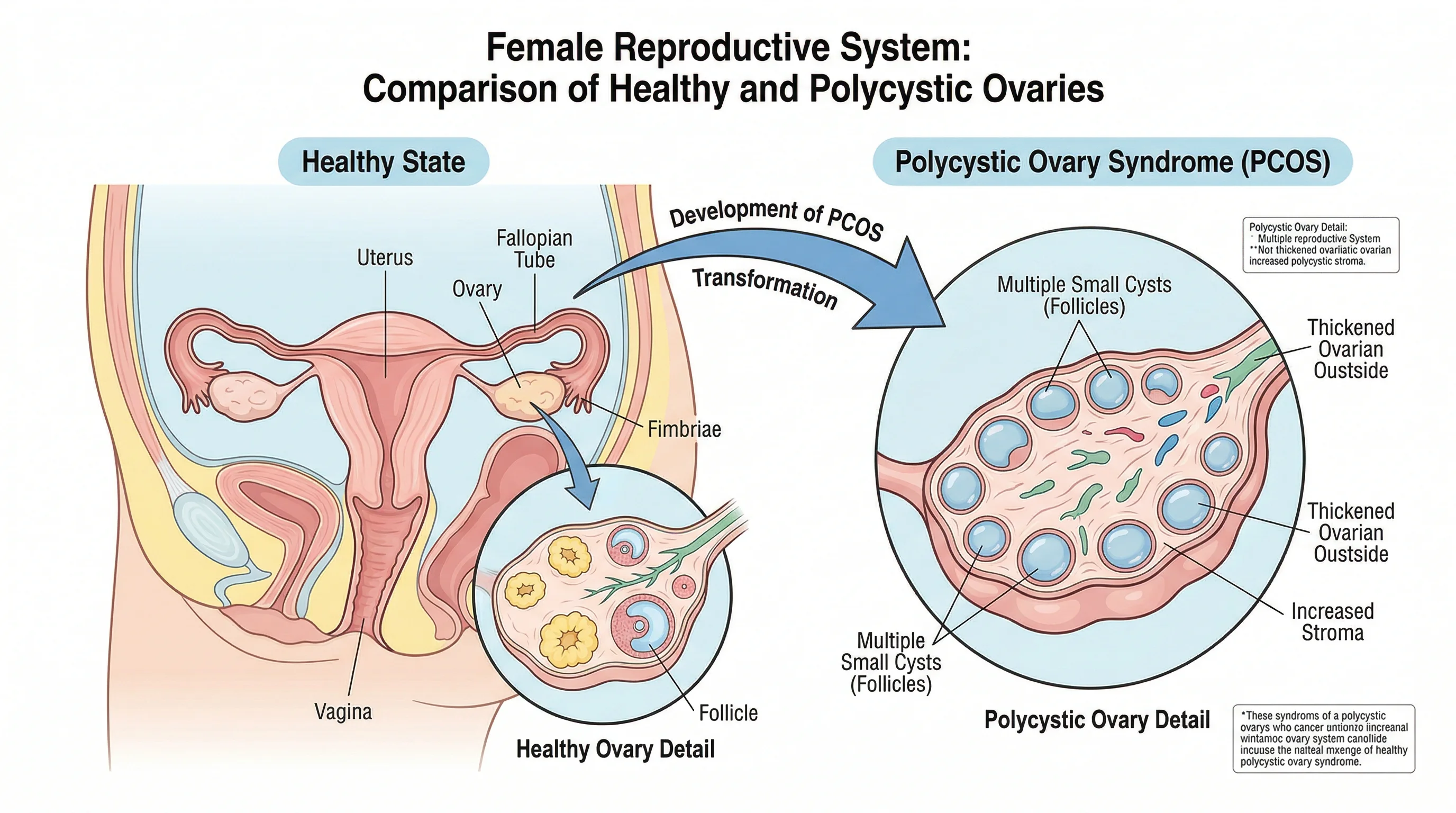
PCOS, weight, and how fertility gets blocked
To understand why weight-loss drugs are accidentally causing pregnancies, you need to understand the hormonal disruption that obesity creates in the reproductive system. Ovulation depends on precisely timed signaling between the brain, the pituitary gland, and the ovaries. Excess weight throws off the timing at every step.
Polycystic ovary syndrome is the biggest piece of the puzzle. Between 7% and 10% of women of reproductive age in the United States have PCOS, making it one of the most common endocrine conditions in women and a leading cause of infertility. The condition involves irregular or absent ovulation, elevated androgens like testosterone, and -- in many cases -- insulin resistance that makes weight gain almost inevitable and weight loss extremely difficult.
The relationship between body weight and fertility is measurable. Women with a BMI above 27 are three times more likely than women in the normal weight range to be unable to conceive, primarily because they stop ovulating regularly.
Fat tissue is not passive storage. It is an active endocrine organ that produces estrogen. When fat cells produce excess estrogen on top of what the ovaries already make, the body's feedback system reads those elevated levels as a signal that pregnancy has already occurred and shuts down ovulation. The body thinks it's already pregnant, so it stops releasing eggs.
Dr. Sharron Manuel, a reproductive endocrinologist at HRC Fertility, describes the specific disruption pathways. Increased body weight inhibits ovulation through "disruption of the hypothalamic-pituitary-ovarian axis, insulin resistance, altered sex steroid metabolism, and direct effects on oocyte and endometrial quality," she explains. These pathways compound each other: insulin resistance drives up androgen production, androgens interfere with follicle development, and poor endometrial quality reduces the chance that a fertilized egg will implant even when ovulation does occur.
For women caught in this cycle, the condition that causes weight gain also punishes them reproductively. Many have been told to "just lose weight" before their doctors will even evaluate their fertility, advice that ignores how aggressively PCOS-driven insulin resistance fights against caloric restriction. GLP-1 drugs change the equation because they attack the exact metabolic dysfunction that was blocking reproduction.
How GLP-1 drugs restart ovulation
GLP-1 receptor agonists work by mimicking a gut hormone called glucagon-like peptide-1. When you eat, your body naturally releases GLP-1 to signal the pancreas to produce insulin, tell your brain you're full, and slow down how quickly food leaves your stomach. The drugs amplify all three of those signals. The result is reduced appetite, better blood sugar control, and for many people, significant weight loss.
That weight loss is the primary driver of restored fertility, and the threshold is lower than most people expect. Dr. Adam Evans, a reproductive endocrinologist at University Hospitals, notes that losing just 5% to 10% of body weight can help women who aren't ovulating regularly achieve normal cycles and ovulation. For a 220-pound woman, that's as little as 11 pounds.
GLP-1 drugs blow past that threshold routinely. In the SELECT trial, 44% of patients using semaglutide lost more than 10% of their body weight within two years, and 11% lost more than 20%. Nearly half of all patients hit the fertility-restoration zone, and about one in nine went well beyond it.
For women with PCOS, the fertility benefit may be even larger than for the general population. GLP-1 drugs simultaneously improve insulin sensitivity and reduce body weight, the two factors most tightly linked to PCOS-driven anovulation.
The clinical data on PCOS is building. A 2024 meta-analysis of four randomized controlled trials involving 176 women with PCOS found that GLP-1 receptor agonists significantly reduced waist circumference by 5.16 cm, BMI by 2.42 points, and total testosterone levels compared to placebo. Testosterone suppression matters because excess androgens are one of the direct barriers to follicle maturation and ovulation in PCOS.
A separate meta-analysis of eight randomized controlled trials found that GLP-1 receptor agonists were more effective than metformin -- the traditional first-line drug for PCOS-related insulin resistance -- at improving insulin sensitivity and reducing BMI. That comparison matters because about 20% of women with PCOS who take metformin see improved ovulation, according to Dr. Christina Boots, a reproductive endocrinologist at Northwestern University. If GLP-1 drugs outperform metformin on the metabolic markers that drive anovulation, the ovulation improvements are likely at least as large.
There may also be more going on than weight loss alone. Preclinical data suggest that GLP-1 receptor agonists may directly stimulate luteinizing hormone secretion and support ovarian morphology and endometrial receptivity through mechanisms that appear independent of weight loss, according to Dr. Manuel. These findings are still in animal models, and researchers haven't confirmed them in humans. But they raise the possibility that GLP-1 drugs affect the reproductive system through pathways that go beyond the scale.
What this means in practice: if you're a woman with PCOS or obesity-related anovulation and you start a GLP-1 drug, your body may begin ovulating again within weeks or months, potentially before you notice that your irregular periods have become regular. Women who have spent years assuming they can't conceive may suddenly be fertile again without realizing it.
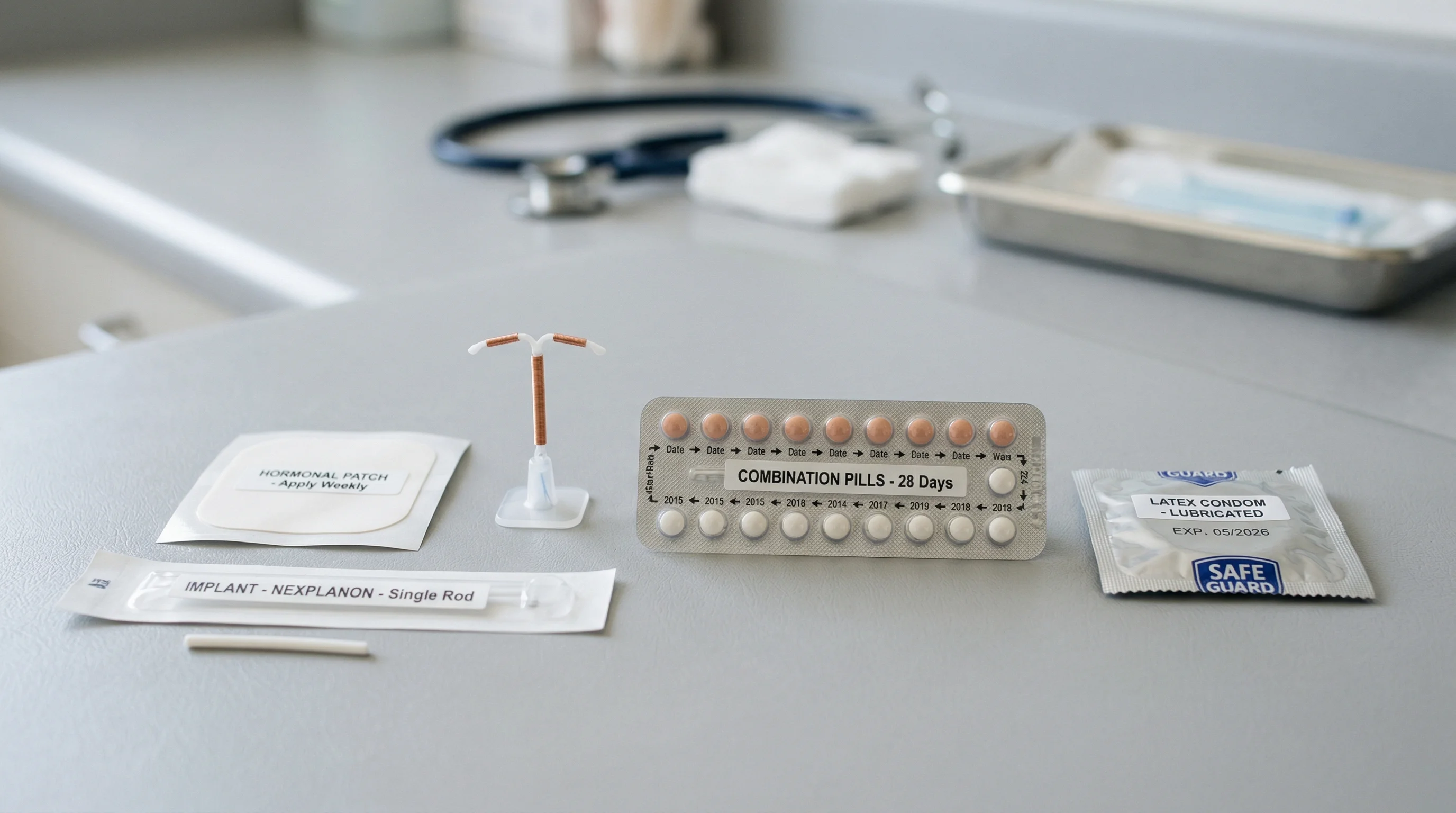
Birth control and semaglutide: the absorption question
The fertility restoration story has a second chapter that gets less attention: what happens to oral birth control when you're taking a drug that fundamentally changes how your digestive system processes pills.
GLP-1 drugs slow gastric emptying. That is how they reduce appetite and help with weight loss. But your stomach doesn't distinguish between food and medication. When a birth control pill sits in your stomach longer than expected, the hormones it contains get absorbed on a different timeline. The question is whether that delay matters enough to cause contraceptive failure.
The answer depends on which GLP-1 drug you're taking, and the differences are large.
A Novo Nordisk trial of 25 women found that oral semaglutide did not affect the bioavailability of combined oral contraceptives containing ethinylestradiol and levonorgestrel. Both the total exposure (AUC) and peak blood levels (Cmax) remained within the predefined "no effect" interval. The FDA's analysis in the Ozempic prescribing label confirms this, showing no clinically relevant drug-drug interaction with ethinylestradiol or levonorgestrel at the injectable semaglutide doses tested.
Tirzepatide, marketed as Mounjaro and Zepbound, is a different drug with a different problem. Pharmacist Jessica Skelley at Samford University found in a 2024 literature review that tirzepatide reduced plasma oral contraceptive hormone levels by up to 66% and delayed the time to maximum effect by up to 4.5 hours. "It was with the five-milligram dose, which is one of the lower doses of tirzepatide," Skelley noted. "Even that lower dose showed a pretty profound impact."
| GLP-1 drug | Oral contraceptive hormone reduction | Absorption delay | FDA birth control warning |
|---|---|---|---|
| Semaglutide (Ozempic, Wegovy) | None detected | Not clinically significant | No specific warning |
| Tirzepatide (Mounjaro, Zepbound) | Up to 66% | Up to 4.5 hours | Yes: backup contraception for 4 weeks after initiation and dose changes |
| Exenatide, liraglutide, dulaglutide | Up to 45% | Up to 3.5 hours | No specific FDA warning, but caution advised |
The difference comes from tirzepatide's dual mechanism. Unlike pure GLP-1 drugs, tirzepatide also activates the glucose-dependent insulinotropic polypeptide (GIP) receptor. That dual action makes it more potent for weight loss and blood sugar control, but it also causes more pronounced slowing of gastric emptying, which is what disrupts oral medication absorption.
But even for semaglutide, where the pharmacokinetic data looks reassuring, there is a subtler problem. Dr. Evans points out that nausea and vomiting, among the most common GLP-1 side effects, can cause a woman to effectively miss a birth control dose without realizing it. A pill thrown up two hours after swallowing may not have been fully absorbed.
Dr. Claire Meek, a diabetes-in-pregnancy specialist at the University of Cambridge, adds another consideration: a missed birth control dose has different consequences depending on your metabolic state. When you weigh more and have insulin resistance, your body is less likely to ovulate from a single missed pill. After losing 30 or 40 pounds on a GLP-1 with normalized insulin, that same missed pill might be the one that lets ovulation through. "It's quite possible that missing a dose of birth control is more likely to result in pregnancy when you're on a GLP-1 agonist," Meek said.
So even though semaglutide doesn't pharmacologically interfere with the pill, the metabolic changes it produces shrink the margin for any contraceptive lapse. The pill works the same as it always did. Your body just responds differently to gaps in coverage now that it is capable of ovulating again.
What the research actually shows so far
Women who discover they're pregnant while taking semaglutide face immediate anxiety: could the drug have harmed the baby? Based on the data we have, the answer is reassuring. But the data is limited.

The largest population-based study to date, published in JAMA Internal Medicine in 2024 by Cesta et al., examined data from approximately 3.5 million women across the United States, four Nordic countries, and Israel. Of these, 938 had periconception exposure to GLP-1 receptor agonists. The adjusted relative risk for major congenital malformations was 0.95 (95% CI: 0.72-1.26) compared with insulin, meaning no increased risk was detected.
A Danish cohort study by Kolding et al. tracked 32 semaglutide-exposed pregnancies within a population of 104,422 singleton pregnancies. The rate of major malformations among semaglutide-exposed pregnancies was 9.4%, compared to 13.9% in insulin-exposed and 7.7% in unexposed pregnancies -- not statistically different from either group. Semaglutide-exposed pregnancies did show higher rates of preterm birth and large-for-gestational-age babies compared to unexposed pregnancies, though these rates were comparable to the insulin group, suggesting that underlying diabetes and obesity, not the drug itself, likely drove those outcomes.
A separate multicenter prospective study by Dao et al. followed 168 pregnancies exposed to GLP-1 receptor agonists in the first trimester, including 51 on semaglutide. The rate of major birth defects was 2.6% in the GLP-1 group, compared to 2.3% in women with diabetes on non-GLP-1 therapy and 3.9% in overweight/obese women without diabetes. No increased risk of pregnancy loss was found.
One point in favor of safety: ex-vivo and in-vivo studies have shown negligible placental transfer of GLP-1 receptor agonists, meaning the drug molecules likely don't reach the fetus in meaningful quantities. This is consistent with their large molecular size.
The animal data is less comforting. The FDA's prescribing information for Ozempic reports that in animal studies, GLP-1 agonists were associated with embryofetal mortality, structural abnormalities, and growth disturbances including delayed bone development. In female rats, semaglutide caused increased estrus cycle length and a small reduction in corpora lutea (the structures that produce progesterone after ovulation). The FDA label characterizes these effects as "likely an adaptive response" to the drug's effect on food consumption and body weight rather than direct toxicity. Put simply, the animals weren't eating enough to support normal reproduction.
An important caveat on all the human data: the sample sizes are small (32 pregnancies in the Danish study, 168 in the Dao study), the exposure periods varied, and no study was designed to measure semaglutide's effect on fetal development specifically. These are observational snapshots, not definitive safety trials.
Dr. Zaher Merhi, a reproductive endocrinologist at Albert Einstein College of Medicine, is blunt about the gap: "There are zero -- let me repeat that, zero -- studies looking at the impact of GLP-1 receptor agonists and anything related to fertility or menstruation in women without PCOS." Most of the fertility discussion has been extrapolated from PCOS populations and general weight-loss physiology. Rigorous prospective trials are years away.
Both Novo Nordisk and Eli Lilly are now building pregnancy registries to track outcomes in women who conceive while taking their drugs. Until those registries produce results, women and their doctors are working with incomplete information.

Practical guidance for women on GLP-1s
The gap between the fertility effects of GLP-1 drugs and the advice women actually receive is wide. In the Australian case series by Morton and He, none of the 12 women with type 2 diabetes who became pregnant while on semaglutide recalled receiving any preconception counseling. Not one.
That gap matters because the FDA's guidance is specific. The Ozempic prescribing label states that women should discontinue the drug at least two months before a planned pregnancy. The reason is pharmacokinetic: semaglutide has an elimination half-life of approximately one week and remains present in the circulation for about five weeks after the last dose. The two-month window gives the drug time to fully clear.
Based on what reproductive endocrinologists and the available evidence suggest for women of reproductive age on GLP-1 drugs:
| If you are... | Recommendation |
|---|---|
| Not planning pregnancy and on oral birth control | Consider switching to a non-oral method (IUD, implant, patch) or adding barrier contraception, especially during dose changes or if experiencing GI side effects |
| Taking tirzepatide (Mounjaro/Zepbound) specifically | Use backup contraception for 4 weeks after starting and after each dose increase (per FDA label) |
| Planning pregnancy in the future | Discuss a timeline with your doctor. Stop the GLP-1 at least 2 months before trying to conceive. Start prenatal vitamins including folic acid before discontinuing |
| Discovered you're pregnant while on a GLP-1 | Stop the medication immediately and contact your OB/GYN. The limited human data does not show increased malformation risk, but your doctor may recommend maternal-fetal medicine consultation |
| Using GLP-1 for PCOS management | Be aware that ovulation may resume before your periods become noticeably regular. Use contraception if pregnancy is not desired |
Dr. Evans stresses that non-oral contraceptives -- IUDs, hormonal implants, patches, and condoms -- do not appear to be affected by GLP-1 use. For women who want the most reliable protection while on any GLP-1 drug, long-acting reversible contraception eliminates the absorption question entirely.
There is also the weight-regain factor to plan around. "Many people will regain weight when they come off these medications," Dr. Evans notes. Women using GLP-1 drugs as a bridge to fertility treatment should work with their medical team on timing: losing weight before conception may improve pregnancy outcomes, but waiting too long introduces age-related fertility decline. There is no universal formula. It depends on age, metabolic health, and whether assisted reproduction is needed.
Dr. Jenna McCarthy, a reproductive endocrinologist at IVFMD, puts the difficulty plainly: "For someone who is actively undergoing infertility treatment, they have to internalize the idea of not getting pregnant, which can be hard to do. But because we don't have safety data on GLP-1s and pregnancy, patients on them have to be intentional in preventing pregnancy."
GLP-1 drugs are fertility-adjacent medications now, whether anyone intended that or not. Every prescription written for a woman of reproductive age should include a conversation about contraception, pregnancy planning, and the two-month washout window. That conversation isn't happening consistently, and the growing number of women arriving at OB/GYN offices with surprise pregnancies makes that clear.

Frequently Asked Questions
Can Ozempic directly make you more fertile?
Not directly in the way a fertility drug like clomiphene does. The fertility boost comes primarily from weight loss and improved insulin sensitivity, which can restore normal ovulation in women whose cycles were disrupted by obesity or PCOS. Some preclinical evidence suggests GLP-1 receptors may have direct effects on the reproductive system, but this hasn't been confirmed in humans. The practical result is the same: women who weren't ovulating may start ovulating again, sometimes within weeks of starting the medication.
Does semaglutide interfere with birth control pills?
Clinical trials show that semaglutide (Ozempic, Wegovy) does not significantly reduce the blood levels of oral contraceptive hormones. Tirzepatide (Mounjaro, Zepbound) can reduce oral contraceptive hormone levels by up to 66%. The FDA recommends backup contraception when starting or increasing tirzepatide doses. For any GLP-1 drug, nausea and vomiting can cause you to effectively miss a pill dose, and the weight loss itself makes your body more likely to ovulate from any contraceptive lapse.
Should I stop taking Ozempic if I want to get pregnant?
Yes. The FDA recommends discontinuing semaglutide at least two months before a planned pregnancy because the drug stays in your system for approximately five weeks after the last dose. Work with your doctor to time the transition, start prenatal vitamins with folic acid before stopping the medication, and have a plan for managing weight and blood sugar during the washout period and pregnancy.
What if I got pregnant while taking a GLP-1 drug?
Stop the medication immediately and contact your OB/GYN. The human data available so far, from studies tracking hundreds of exposed pregnancies, has not shown an increased rate of birth defects compared to other diabetes medications. These studies are small and observational, not designed as safety trials. Your doctor may recommend a referral to a maternal-fetal medicine specialist for additional monitoring. Animal studies showed potential for harm at high doses, but ex-vivo research suggests the drug transfers minimally across the placenta.
Are "Ozempic babies" healthy?
In the clinical data published so far, babies born to women who were exposed to semaglutide in the first trimester have had malformation rates comparable to those born to women on insulin or no diabetes medication. Some studies noted higher rates of preterm birth and larger birth weights in semaglutide-exposed pregnancies, but these were also common in the insulin-comparison group, suggesting maternal diabetes and obesity, not the drug, were the likely causes. Novo Nordisk and Eli Lilly are both running pregnancy registries to gather more comprehensive long-term data.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.