A 73-year-old rancher ordered a beer on Ozempic. He couldn't finish it.
J. Paul Grayson started taking Ozempic for weight loss after the COVID pandemic left him with high blood pressure, prediabetes, and 40 extra pounds on his 6-foot-3 frame. The appetite suppression was expected. What happened next was not. "I ordered a beer, took a sip, and I couldn't finish it," Grayson told NPR. Before the medication, he could work through a bottle of wine and a bag of chocolates in an evening without much effort. On semaglutide, even one drink felt like enough.
His experience is far from unique. Across clinics, social media threads, and research interviews, people taking GLP-1 receptor agonists like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro, Zepbound) have reported something striking: their interest in alcohol, cigarettes, and other substances fades alongside their food cravings. Carolina Haass-Koffler, an associate professor of psychiatry at Brown University who studies the neurobiology of addiction, said clinicians have reported that semaglutide "obliterated" many patients' cravings for substances they had struggled with for years.
Patients on GLP-1 drugs often describe a phenomenon called "food noise" going silent — the persistent mental chatter about eating that used to dominate their thoughts. But Ziyad Al-Aly, a clinical epidemiologist at Washington University School of Medicine who led one of the largest studies on this topic, thinks what patients are describing goes further. He calls it "drug noise" — the same relentless craving mechanism, just aimed at different targets. "GLP-1 drugs may quiet what I call 'drug noise,' the relentless craving that drives addiction across substances," Al-Aly said.
The people reporting these changes were not trying to quit anything. In the first randomized clinical trial of semaglutide for alcohol use disorder, led by Dr. Christian Hendershot at the University of Southern California, participants were specifically non-treatment-seeking — they had not enrolled with any intention to reduce their drinking. The medication appeared to do it on its own.
Only about 2% of people with alcohol use disorder receive any medication for it, and fewer than 10% seek treatment in a given year — one of the largest treatment gaps in all of medicine, according to the Hendershot trial data. Meanwhile, an estimated 178,000 Americans die from alcohol-related causes each year.

The brain's volume knob for wanting — and how GLP-1 drugs turn it down
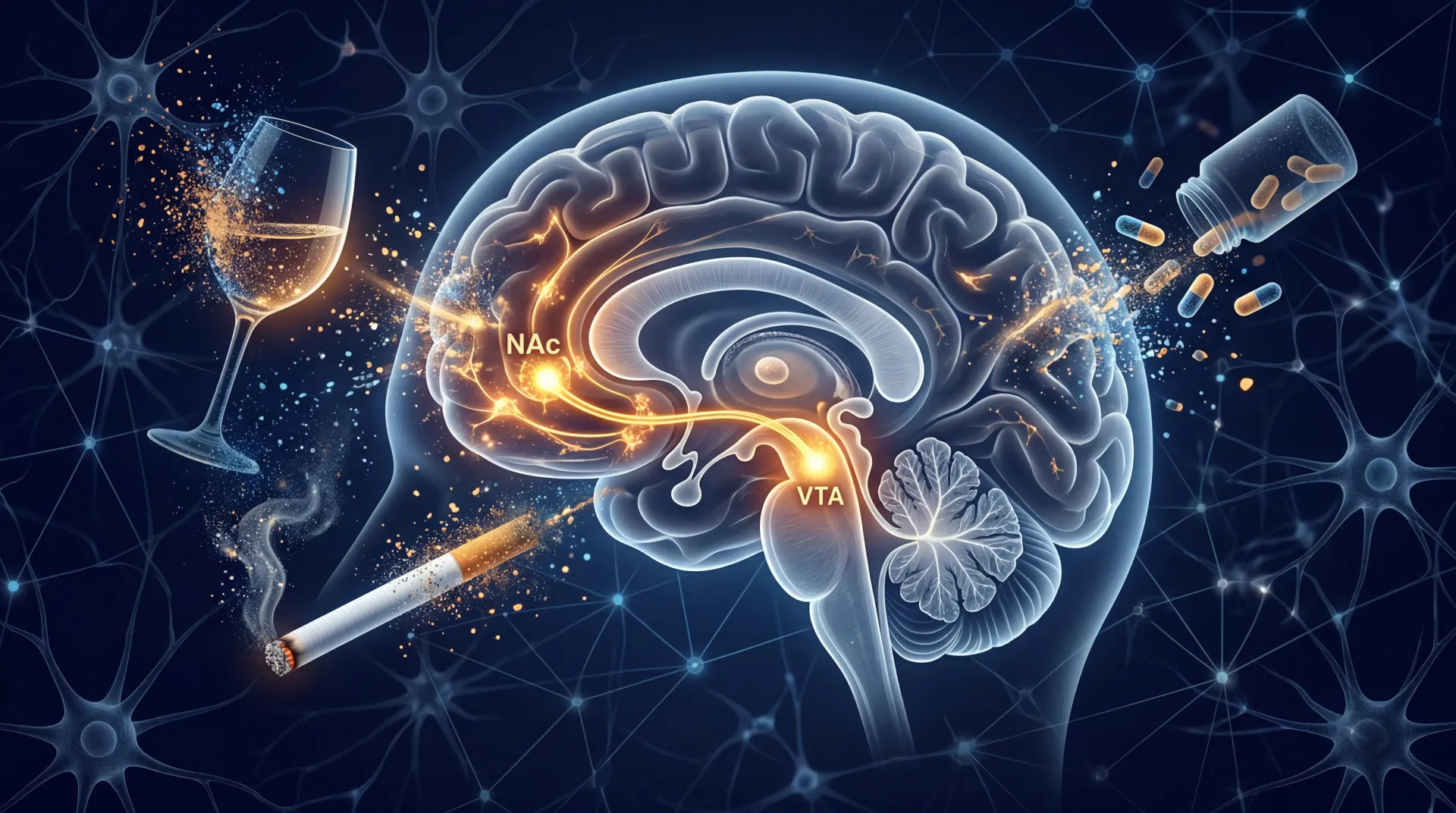
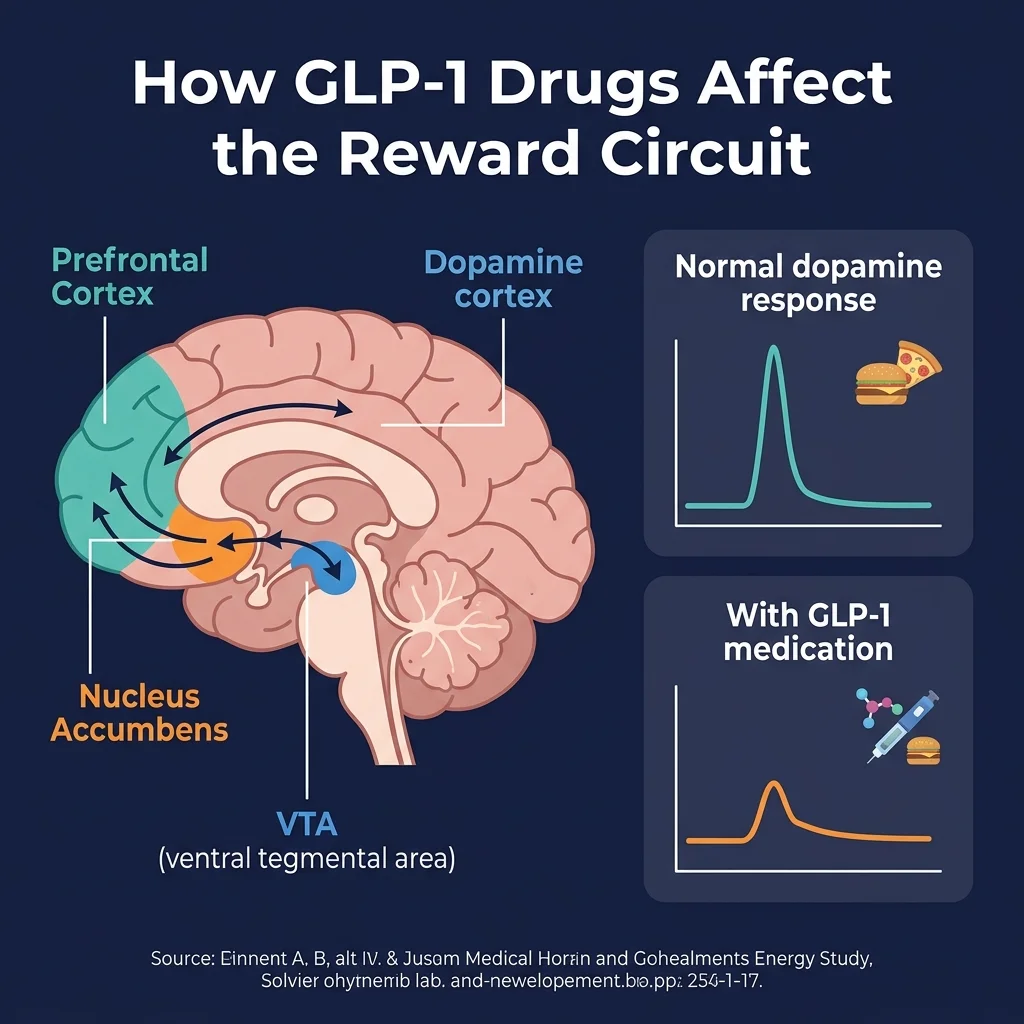
Think of dopamine as your brain's "do that again" signal. When you bite into a cookie, take a sip of cold beer on a hot day, or inhale a cigarette, dopamine surges in a region called the striatum — the brain's motivation headquarters. That spike tells you the experience was worth repeating. It is the same basic mechanism whether the trigger is food, alcohol, nicotine, or opioids. "Dopamine in the striatum is the motivation and learning signal for everything," said Alexandra DiFeliceantonio, a neuroscientist at Virginia Tech. "All addictive drugs increase dopamine there."
GLP-1 receptor agonists appear to reduce that dopamine surge. The hormone GLP-1, which these drugs mimic, is not only produced in the gut after eating — it is also active in the brain, where its receptors cluster in areas governing reward, motivation, and stress. According to Haass-Koffler at Brown, the drugs bind to receptors in three areas that form the core of the reward circuit: the ventral tegmental area (VTA), the nucleus accumbens, and the prefrontal cortex. By activating GLP-1 receptors in these regions, the drugs blunt dopamine release and reduce reward signaling. The result: people feel less compelled to chase whatever was triggering the craving.
An analogy that helps: imagine your brain has a volume knob for "wanting." Normally, a glass of wine or a drag on a cigarette cranks it up. GLP-1 drugs seem to hold that knob closer to baseline, so the signal still arrives but without the same intensity. Lorenzo Leggio, clinical director of the National Institute on Drug Abuse (NIDA), put it this way: "The mechanism in the brain that regulates overeating overlaps with those responsible for the development and maintenance of addiction."
Some of the most detailed mechanistic work comes from Paul Kenny's lab at Mount Sinai's Friedman Brain Institute. Kenny's team discovered that a brain structure called the interpeduncular nucleus (IPN) contains some of the highest concentrations of GLP-1 receptors anywhere in the brain. The IPN also carries receptors for opioids, nicotine, and cannabinoids — a convergence that suggests it may serve as a crossroads for multiple types of craving. In a study published in Nature Neuroscience, Kenny's team found that stimulating GLP-1 receptors in the IPN completely abolished the rewarding effects of nicotine in animal models.
There is an origin story worth knowing here. The first GLP-1 drug, exenatide, was derived from the saliva of the Gila monster, a venomous lizard native to the American Southwest. Approved in 2005 for type 2 diabetes, it required twice-daily injections and never gained widespread popularity. Semaglutide, approved in 2017, is administered once weekly with far greater potency — and it is the version that has generated the avalanche of addiction-related observations.
Animal research has been building for over a decade. Elisabet Jerlhag and her colleagues at the University of Gothenburg in Sweden have published nearly a dozen studies showing GLP-1 drugs stop binge drinking in rodents, prevent relapse in "addicted" animals, and reduce alcohol consumption by over 50%. When researchers gave semaglutide to green vervet monkeys — primates that voluntarily drink alcohol much like humans — the animals drank less without showing signs of nausea or changes in water intake, suggesting the drug lowered the reward value of alcohol rather than simply making them feel sick.
50% fewer substance-related deaths: what the clinical data shows
The most comprehensive evidence to date comes from a study published in The BMJ in March 2025, led by Al-Aly at Washington University. His team analyzed the electronic health records of 606,434 U.S. veterans with type 2 diabetes, comparing those who started GLP-1 medications to those prescribed a different class of diabetes drug (SGLT-2 inhibitors) over three years.
The risk reduction showed up across every substance category. Among veterans with no prior substance use disorder, those taking GLP-1 drugs had an 18% lower risk of developing alcohol use disorder, 25% lower risk of opioid use disorder, and roughly 20% lower risk of both cocaine and nicotine dependence. That translated to about seven fewer new substance use disorder diagnoses per 1,000 people taking GLP-1 medications over three years.
Among veterans who already had a substance use disorder, the numbers were more dramatic. GLP-1 users experienced 50% fewer substance-related deaths, 39% fewer overdoses, and 26% fewer drug-related hospitalizations compared to the SGLT-2 group. Over three years, that amounted to roughly 12 fewer serious events — including 2 fewer deaths — per 1,000 people.
| Outcome (pre-existing SUD group) | Risk reduction with GLP-1 | Hazard ratio (95% CI) |
|---|---|---|
| Substance-related deaths | 50% | 0.50 (0.32-0.79) |
| Drug overdose | 39% | 0.61 (0.42-0.88) |
| SUD-related ED visits | 31% | 0.69 (0.61-0.78) |
| SUD-related hospitalizations | 26% | 0.74 (0.65-0.85) |
| Suicidal ideation or attempt | 25% | 0.75 (0.67-0.83) |
A Swedish nationwide study of 227,000 people with alcohol use disorder found a parallel pattern: GLP-1 users had a 36% lower risk of alcohol-related hospitalizations — more than double the 14% reduction seen with naltrexone, which is currently the best-performing FDA-approved medication for alcohol use disorder in that analysis.
These are observational studies, not randomized controlled trials, so they cannot prove GLP-1 drugs directly caused the improvements. But the first randomized trial provided its own signal. Dr. Hendershot's Phase 2 trial at UNC-Chapel Hill randomized 48 adults with alcohol use disorder to receive either low-dose semaglutide or placebo for nine weeks. In a laboratory setting where participants could freely drink their preferred alcoholic beverages, those on semaglutide consumed significantly less alcohol (P=.01) and achieved lower peak blood alcohol concentrations (P=.03) compared to placebo, with medium-to-large effect sizes.
The weekly tracking data told a similar story. Semaglutide significantly reduced alcohol craving (P=.01) and drinks per drinking day (P=.04), and participants on the drug had greater reductions in heavy drinking days over the study period (P=.04). The effect sizes grew with the dose: they were small during weeks 1-4 at the 0.25mg dose but reached the large range (d>0.80) by weeks 5-8 at 0.5mg for heavy drinking days and drinks per drinking day. For comparison, naltrexone — the standard pharmacotherapy for alcohol use disorder — typically shows only small effect sizes (d~0.20) in similar trials.
A secondary finding deserves attention: among the 13 participants who smoked cigarettes at baseline, those receiving semaglutide had significantly greater reductions in cigarettes per day (P=.005) compared to the placebo group. No medication is currently approved for both alcohol reduction and smoking cessation, which makes a dual-action drug especially interesting from a public health perspective.

At least a dozen trials are testing GLP-1 drugs for addiction right now
The Hendershot trial used semaglutide at doses far below what patients typically receive for weight loss (maxing out at 1.0mg versus the standard 2.4mg). The question of whether higher doses produce stronger anti-craving effects is one of many that active clinical trials are designed to answer.
The highest-profile study currently recruiting is the STAR trial (Semaglutide Therapy for Alcohol Reduction), run by the National Institute on Drug Abuse in Baltimore under the direction of Dr. Lorenzo Leggio. This Phase 2a proof-of-concept study is testing semaglutide at the full 2.4mg weekly dose — or the maximum tolerated dose — in adults with alcohol use disorder over approximately 20 weeks. What sets it apart from earlier work is the inclusion of functional MRI brain imaging during alcohol and food cue exposure, which could provide direct evidence of how semaglutide changes reward processing in the human brain.
The STAR trial also uses a creative study design: participants sit in a bar-like room and are exposed to cues designed to trigger alcohol cravings, then enter a virtual reality cafeteria to make food and drink choices. Blood biomarkers (PEth levels) provide an objective measure of alcohol consumption that does not rely solely on self-report.
A search of ClinicalTrials.gov reveals at least eight registered studies focused specifically on semaglutide for alcohol and nicotine-related conditions. These include trials testing semaglutide for smoking cessation (one UNC study with 24 participants has already completed), post-smoking-cessation weight management, and alcohol reduction in various populations. Al-Aly noted in his BMJ paper that more than a dozen additional trials are underway or actively enrolling, with several more planned.
| Trial | Target | Phase | Status | Key feature |
|---|---|---|---|---|
| STAR (NCT06015893) | Alcohol use disorder | Phase 2a | Recruiting | Full 2.4mg dose, fMRI imaging |
| NCT05520775 | Alcohol use disorder | Phase 2 | Completed | First published semaglutide RCT for AUD |
| NCT05530577 | Nicotine/smoking | Phase 2 | Completed | Smoking lapse after overnight abstinence |
| NCT07059377 | Smoking cessation | — | Registered | Semaglutide for smoking cessation |
| NCT06173778 | Post-cessation weight | — | Registered | Weight prevention after quitting smoking |
| NCT05891587 | Alcohol reduction | — | Registered | Additional STAR-related site |
The scope of ongoing research reflects a broader shift in how scientists think about these drugs. As Al-Aly put it: "In addiction medicine, a lot of treatments target just one thing — for example, a nicotine patch helps with smoking, but not alcohol — but there is no medication that works across addictive substances, let alone all of them." The possibility that GLP-1 drugs could target a shared vulnerability underlying multiple forms of addiction, rather than any single substance pathway, is the hypothesis driving most of these trials.
Small samples, low doses, and the question nobody can answer yet
The caveats are substantial. The Hendershot trial — the only published randomized controlled trial of semaglutide for alcohol use disorder — enrolled just 48 people. That is enough to detect a signal, which it did, but far too few to draw confident conclusions about how well this works across the broader population. The trial lasted only nine weeks. Participants had moderate alcohol use disorder, not severe. And the doses were the two lowest available — well below what millions of people take for weight management.
Anna Lembke, a professor of psychiatry at Stanford Medicine and the author of "Dopamine Nation," frames the limitation clearly: "Interindividual variability in response to medications to treat addiction has always been true and will continue to be true with GLP-1s." Some people may respond dramatically, while others see no effect at all — a pattern seen with every existing addiction medication.
There is also the weight loss problem. Participants in the semaglutide group lost an average of 5% of their body weight over the nine-week trial. For most participants — whose BMI averaged 32, in the obese range — that was a welcome benefit. But for someone with alcohol use disorder who is already at normal or low weight, that kind of loss could be medically concerning. The trial excluded anyone with a BMI below 23 for safety reasons, which means we have no data on how the drug performs in leaner populations.
History offers its own cautionary tale. A previous randomized trial tested exenatide — an older, less potent GLP-1 drug — for alcohol use disorder and found no overall benefit. There were hints of efficacy in a subgroup of participants with BMI above 30, but nothing conclusive. The most likely explanation is simply that semaglutide is a more potent drug than exenatide. The point remains: not every GLP-1 agonist at every dose will necessarily work for addiction.
| What we know | What we don't know |
|---|---|
| GLP-1 drugs reduce dopamine signaling in reward centers | Which brain regions matter most for each substance |
| One RCT (n=48) showed reduced craving and heavy drinking | Whether effects hold at full therapeutic doses |
| Large observational data (606K+) shows reduced SUD risk | Whether these drugs work for severe alcohol use disorder |
| Animal models consistently show reduced substance intake | Whether cravings rebound after stopping the medication |
| Side effects are consistent with known GLP-1 profile | Long-term safety in non-obese addiction populations |
The biggest unanswered question may be what happens when patients stop. People who discontinue GLP-1 drugs for weight management typically see their appetite return and regain lost weight. Whether the same rebound occurs with addiction cravings — and what that would mean for someone in recovery suddenly facing the full force of craving again — is completely unknown. There is also an open question about whether prolonged suppression of the brain's reward circuitry could dampen everyday motivational drive in some people — the willingness to work hard, compete, or pursue goals that are also mediated by dopamine.

If you're already taking a GLP-1 drug, here's what actually matters
GLP-1 receptor agonists have not been approved by the FDA for treating any form of addiction. The evidence, while promising, comes from one small randomized trial and several large observational studies — not the Phase 3 trials that regulators require for a new indication. That means insurance will not cover these drugs when prescribed specifically for addiction, and most addiction specialists are not yet recommending them for this purpose.
One thing that Lembke at Stanford emphasized is reassuring: GLP-1 drugs are not themselves addictive and do not trigger withdrawal when discontinued. That puts them in a different category from some existing addiction treatments that carry their own dependence risks.
If you are already taking a GLP-1 medication for diabetes or weight management and have noticed changes in your desire to drink or smoke, you are experiencing something that researchers are actively studying. The practical question is whether to act on it. Al-Aly framed one reasonable approach: a patient with diabetes who is also trying to quit smoking "might reasonably choose a GLP-1 drug over another glucose-lowering medication" — not because it is approved for smoking cessation, but because it may offer a benefit that other diabetes drugs do not.
However, Leggio at NIDA was more cautious in an earlier interview, stating that using Ozempic off-label for addiction is "too early." The Recovery Answers analysis of the Hendershot trial concluded that for people whose primary goal is addressing alcohol use, existing first-line treatments like cognitive behavioral therapy and FDA-approved medications (naltrexone, acamprosate, disulfiram) remain more appropriate — not because GLP-1 drugs have shown no promise, but because the evidence for established treatments is deeper.
Context matters here. The FDA has approved fewer than 10 medications to treat addiction of any kind, despite the fact that substance use disorders affect more than 48 million Americans. GLP-1 drugs are already prescribed at enormous scale by primary care doctors, which means the delivery infrastructure to reach millions of patients exists. If Phase 3 trials confirm what the early data suggests, GLP-1 drugs could bypass many of the barriers — stigma, specialist access, provider unfamiliarity — that have kept existing addiction medications underused for decades. "In order to adapt to this environment, we need all the tools we can find," Lembke said. "It's the reality of the world we live in now."
Frequently Asked Questions
Can I ask my doctor to prescribe Ozempic specifically for alcohol cravings?
You can discuss it, but GLP-1 drugs are not FDA-approved for addiction treatment. Some doctors are prescribing them off-label for this purpose, but most addiction specialists advise caution until larger trials are completed. If you have diabetes or obesity alongside a drinking problem, a GLP-1 drug may offer benefits on both fronts — discuss the trade-offs with your physician. Currently, FDA-approved medications like naltrexone and acamprosate remain the standard first-line pharmacotherapy for alcohol use disorder.
Does semaglutide reduce cravings for all addictive substances, or just alcohol?
The largest observational study found reduced risk across alcohol, opioids, cocaine, cannabis, and nicotine — suggesting the drug may target a common biological mechanism underlying addiction rather than any single substance pathway. However, randomized trial data currently exists only for alcohol and (in a small subsample) cigarettes. Researchers are also investigating whether the effect extends to behavioral addictions like gambling, but that evidence is purely anecdotal at this stage.
Will the cravings come back if I stop taking the GLP-1 drug?
Nobody knows yet. People who stop GLP-1 drugs for weight management typically regain their appetite and lost weight. Whether the same rebound occurs with substance cravings has not been studied. This is one of the most important unanswered questions in the field, and several of the active clinical trials are designed to track what happens after medication discontinuation.
Are there risks to using GLP-1 drugs for addiction if I'm not overweight?
Potentially. In the Hendershot trial, participants on semaglutide lost an average of 5% of their body weight in nine weeks. For most participants (who were obese), this was safe. But for someone at normal or low weight, that loss could be harmful. Common side effects include nausea, constipation, and decreased appetite. Rarely, more serious complications like pancreatitis can occur. Future trials will need to evaluate safety specifically in leaner populations with substance use disorders.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.