The peptide most functional medicine clinics quietly recommend for stubborn gut problems is the same one the FDA banned in 2023 — and is now openly reconsidering for ulcerative colitis. The pivot is real, and it is the reason that, on April 16, 2026, the FDA placed BPC-157 on the public docket for its July 23–24, 2026 Pharmacy Compounding Advisory Committee meeting, with a single proposed indication: ulcerative colitis. Functional medicine doctors who have been treating leaky gut, inflammatory bowel disease, and post-NSAID gastric injury with this peptide for years now have a federal advisory committee taking the gut hypothesis seriously.
This article is not the regulatory recap. It is the clinical case file. What does the gut-specific evidence actually show, condition by condition? Why are clinics like Nava Health and physicians like Mark Hyman, MD and Kim Crawford, MD using it as a "Repair" tool inside structured 5R gut protocols? And where does mainstream gastroenterology — which still has the substance on a Category 2 list — say the floor falls out?
What Functional Medicine Sees in BPC-157
Conventional gastroenterology has a tool for almost every gut problem: PPIs to switch off acid, mesalamine to dampen colonic inflammation, biologics to block TNF-α. Each one suppresses something. None of them rebuild anything. That is the gap functional medicine practitioners have been trying to plug for two decades, and BPC-157 is the molecule that finally fits the shape of the hole.
BPC-157 is a 15-amino-acid synthetic peptide derived from a protective fragment of a protein found naturally in human gastric juice, first isolated in the early 1990s by a Croatian research group led by Predrag Sikiric at the University of Zagreb. The functional medicine appeal is not the mechanism in isolation; it is what the mechanism implies. If the body already secretes a fragment of a "body protection compound" into the stomach, then giving a stable, synthetic version is less like introducing a foreign drug and more like restocking a depleted internal supply. That framing is everywhere in the clinic-side literature — Nava Health calls BPC-157 the "architectural blueprint your body needs to rebuild the castle wall," and the model lines up with how functional MDs already think about leaky gut, autoimmunity, and root-cause repair.
Mark Hyman, MD frames peptides in general as "messengers, delivering precise instructions to your cells — like 'heal this tissue,' 'reduce inflammation,' or 'stimulate growth.'" His shop categorizes BPC-157 alongside Leaky Gut Formulas, Reflux Support, Digestive Support, and Microbiome Support — a placement that itself signals where the practitioner community thinks the peptide earns its keep. Kim Crawford, MD is more explicit, calling BPC-157 a "game changer" alongside KPV (an α-MSH-derived tripeptide) and low-dose naltrexone for treating ulcerative colitis and Crohn's disease in the functional setting.
So what does that mean for someone reading this with a gut problem that has not budged? It means the question stops being "is BPC-157 a miracle?" and starts being "is the underlying model right that my gut barrier needs rebuilding rather than just calming?" The answer to the first is unsettled. The answer to the second is increasingly evidence-backed — which is exactly why the FDA conversation is finally happening.
The Ulcerative Colitis Catalyst: Why 2026 Changed Everything
To understand why 2026 is the inflection point, rewind to September 2023. The FDA moved BPC-157 — along with eighteen other peptides — into Category 2 of the 503A bulk substances list, a designation citing "significant safety risks" including immunogenicity and impurity concerns, which effectively prohibited US compounding pharmacies from making it. Functional medicine practitioners did not stop using it; they pushed it offshore, into the gray market, and into the legal exposure of any clinic that wrote a prescription.
The political ground shifted on February 27, 2026, when HHS Secretary Robert F. Kennedy Jr. went on the Joe Rogan podcast and called himself a "big fan" of peptides, charging that the prior administration's restrictions had pushed patients into a black market producing "very, very substandard" ingredients and arguing that legalized compounding would actually improve safety. By April 16, 2026, the FDA had published a Federal Register notice announcing a Pharmacy Compounding Advisory Committee meeting set for July 23–24, 2026, at the agency's White Oak Campus. The agenda for July 23 lists seven peptides for Section 503A consideration. BPC-157 — both free base and acetate forms — appears with a single proposed indication on its line: ulcerative colitis.
That choice of indication is not arbitrary. It tracks back to a specific human study that has been the workhorse citation in functional medicine practice for years: a multicenter, randomized, double-blind, placebo-controlled Phase II trial by the Ulcerative Colitis Study Group (Ruenzi, Stolte, Veljaca, Oreskovic, Peterson) that randomized 53 patients with mild-to-moderate ulcerative colitis to either an 80 mg BPC-157 enema once daily for two weeks or placebo. The treated group showed a statistically significant decrease in Disease Activity Index, reduced stool frequency, and improved histopathological findings, with no detectable plasma BPC-157 and no difference in adverse events versus placebo. The trial is small (53 patients) and dates from the early 2000s, but it remains the only place where BPC-157's gut data is more than animal modeling.
The other piece of the picture is unpublished but aggressive. A 2024 task-force survey of US 503A compounding pharmacies, cited by clinical reviewers, indicated that over 500,000 BPC-157 prescriptions were filled between 2018 and 2024, with no side effects reported back to the pharmacies. That is not a clinical trial, and it does not capture problems patients did not phone in. But it is real-world signal, and it is now part of the docket the PCAC will weigh.

What the Gut Mechanism Research Actually Shows
The mechanism research is mostly preclinical, but it is unusually specific to the gut — and that is what functional medicine clinicians lean on when they extrapolate. There are five mechanisms worth understanding, because each one maps to a distinct gut problem.
Tight junction repair. Picture the intestinal lining as a wall of single cells held together by molecular Velcro: claudin proteins, occludin, and zonula occludens-1 (ZO-1). When NSAIDs like indomethacin damage the gut, that Velcro tears and undigested food fragments and bacterial molecules slip into circulation — the mechanistic basis of "leaky gut." In rodent models of NSAID-induced permeability, BPC-157 administration prevented the increase in intestinal permeability and preserved ZO-1 and occludin expression at levels comparable to healthy controls. Tight-junction repair is the mechanism functional clinicians cite first when the conversation broadens to autoimmunity, and the indomethacin model is the cleanest experimental version of "leaky gut" in print.
IBD inflammatory cascade modulation. Two animal models dominate IBD research: TNBS (trinitrobenzenesulfonic acid) colitis, which simulates Crohn's, and DSS (dextran sodium sulphate) colitis, which simulates ulcerative colitis. In TNBS models, BPC-157 reduced macroscopic damage scores, histological inflammatory infiltrate, and mucosal ulceration, and lowered TNF-α, IL-1β, and IL-6 versus untreated controls. In DSS models, BPC-157 reduced colonic damage, preserved crypt architecture, and significantly cut myeloperoxidase activity — the standard biochemical marker of neutrophil infiltration into colonic tissue.
Angiogenesis and ulcer healing. Damaged mucosa cannot rebuild without blood supply, and BPC-157's most documented action is upregulating VEGF (vascular endothelial growth factor) and the nitric oxide system to drive new vessel formation at the wound bed. Sikiric's group has documented accelerated healing across cysteamine-induced duodenal ulcers, acetic acid ulcers, aspirin-induced gastric injury, and stress ulcers — a breadth of injury models you don't typically see for a single agent.
Vagal and gut-brain axis modulation. The vagus nerve is the wire between the gut and the brain, and Sikiric's lab has reported that vagotomy — surgical cutting of the vagus nerve — attenuates many of BPC-157's systemic effects, indicating the peptide depends on intact gut-vagal signaling. The same body of work documents interactions with dopamine and serotonin systems, which is why functional medicine practitioners who treat IBS-with-anxiety reach for it.
Microbiome and mucosal defense. Increased prostaglandin E2 production, restored mucus layer integrity, and increased mucosal blood flow round out the cytoprotective picture Sikiric calls "organoprotection." This is also where the practical story lives: clinicians do not use BPC-157 alone — they use it during the "Repair" phase of a multi-step protocol, after the gut has been cleared of dysbiotic flora and inflammatory triggers.
The honest summary: every mechanism above is well-replicated in animals. None of them, except tight-junction preservation in the indomethacin model, has been independently validated in a Phase II or III human trial. The functional medicine bet is that the mechanisms are conserved across species — a bet that is sometimes right and sometimes catastrophically wrong, which is what makes the human data gap matter.
Conditions Where the Evidence Is Strongest (and Weakest)
Not all gut indications are equal. The map below, drawn from the peer-reviewed peptide literature and the clinic-side reviews, separates conditions by how much human data exists versus how much is animal extrapolation.
Read it from the top: ulcerative colitis is the only gut indication with a real, if small, randomized human trial behind it, which is why the FDA picked it as the proposed compounding indication. NSAID-induced gastric injury and classical peptic ulcer healing rest on decades of animal data plus the cytoprotection literature from Sikiric's group, with no large human RCT. Crohn's and leaky-gut sit on TNBS and indomethacin models respectively. Anastomotic healing has rodent surgical data. IBS, SIBO, and GERD — three of the most common reasons patients walk into a functional medicine clinic asking about BPC-157 — have essentially zero human data; clinicians use the peptide for them by extrapolating from the tight-junction and angiogenesis story.
The practical implication: if a functional medicine clinician proposes BPC-157 for ulcerative colitis, they are standing on the firmest available ground. If they propose it for IBS or SIBO, they are reasoning from mechanism to outcome — which can be defensible, but you should know that is what is happening.
Oral vs Injectable for Gut Healing: The Bioavailability Question
For most peptides, oral administration is a non-starter — stomach acid eats them. BPC-157 is the unusual case. Because it was originally isolated from gastric juice itself, it is genuinely acid-stable, which is the entire pharmacological argument for an oral capsule for gut conditions. Whether that translates into systemic absorption is a separate question, and the honest answer is: nobody fully knows.
Clinics dosing oral BPC-157 typically use 500 to 1000 micrograms twice daily, which is roughly twice the injection dose, on the assumption that "oral bioavailability is presumably lower than injectable." Note the language — "presumably." There is no published human pharmacokinetic study that quantifies how much oral BPC-157 makes it past the gut wall and into circulation. A Phase I rectal-administration trial by Veljaca and colleagues found that plasma concentrations were mostly below detection limits, suggesting limited mucosal-to-systemic absorption.
For gut indications, the limited systemic absorption may not matter — and may actually be the point. If you are trying to heal the colonic mucosa in ulcerative colitis, you want the peptide to reach the lumen and dwell there. That is why the only positive human trial (Ruenzi et al.) used an enema, not an injection. For inflammatory bowel disease confined to the colon, topical delivery via enema or oral capsule that survives to the small intestine is mechanistically sensible. For systemic effects — say, joint pain riding alongside Crohn's — the case for subcutaneous injection is stronger, because you do need plasma levels for distal tissue effects.
The functional medicine community has split the difference. Many clinics offer both oral capsules and subcutaneous injection and pick based on the indication: oral or sublingual for primarily luminal gut disease; subcutaneous (a small insulin-needle injection near the affected joint or tendon) for musculoskeletal targets; combination protocols for patients with both. None of this is on a label, because there is no label. It is clinic practice extrapolated from animal pharmacology.
What Functional Medicine Protocols Look Like in Practice
BPC-157 is almost never used solo in a functional clinic. It is the "Repair" tool inside a structured protocol — most often a variant of the 5R framework: Remove, Replace, Reinoculate, Repair, Rebalance. Nava Health's published version places BPC-157 in the Repair phase alongside L-glutamine and zinc carnosine, after stool testing has identified dysbiotic organisms that need clearing and an elimination diet has removed the inflammatory food triggers (most often gluten and dairy).
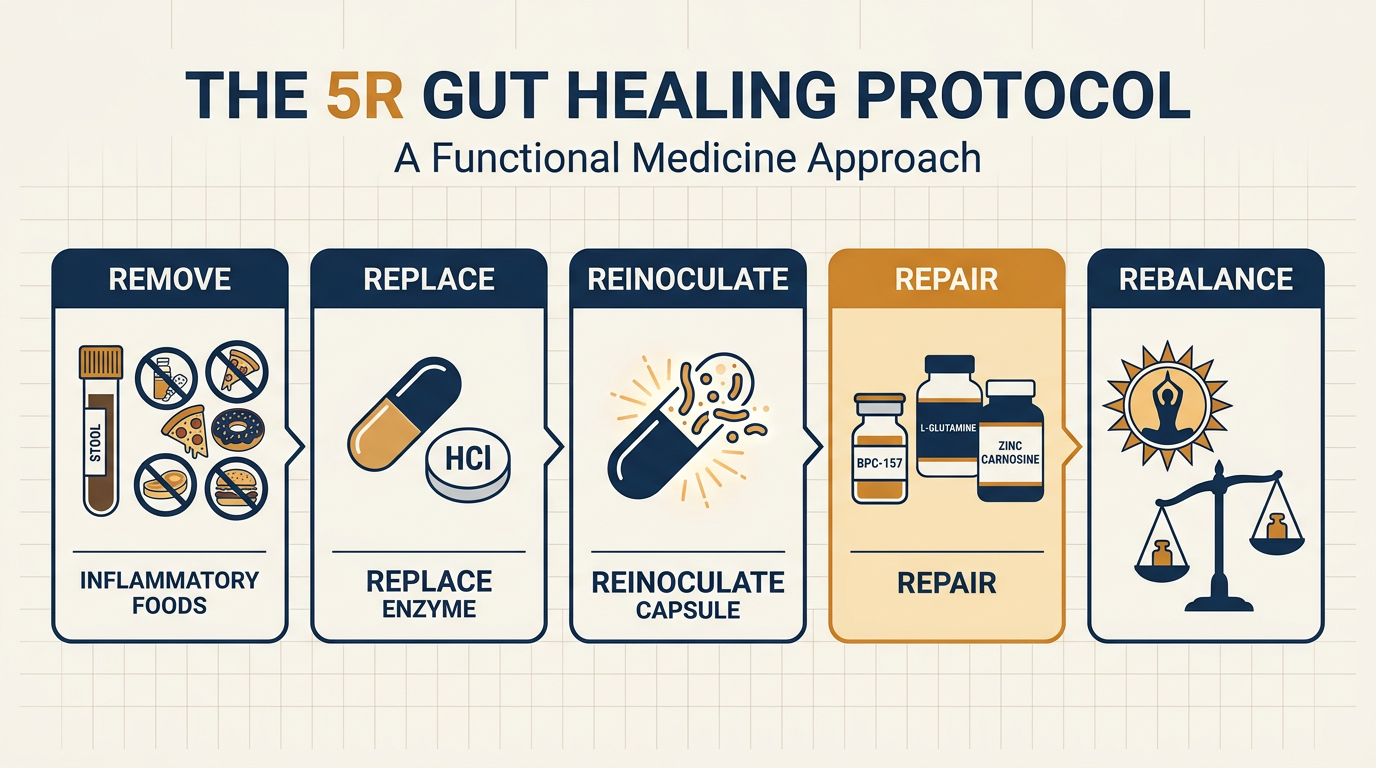
Kim Crawford, MD's published IBD protocol is a useful concrete example. She combines BPC-157 with KPV — the lysine-proline-valine tripeptide derived from α-melanocyte-stimulating hormone, which has its own anti-inflammatory action on colonic epithelial and immune cells — and, in treatment-resistant cases, low-dose naltrexone. She references a clinical study of LDN in IBD enrolling 28 Crohn's and 19 ulcerative colitis patients, in which 35 (75%) responded with reduced disease activity sustained at least one month, and six achieved full clinical remission. The peptide stack is paired with a sporulating probiotic regimen (typically Bacillus subtilis and Bacillus coagulans, starting at 5 billion CFU and titrating up), targeted prebiotic fiber, normalized vitamin D, and an autoimmune-protocol elimination diet.
The pattern across clinics is consistent: BPC-157 is a piece, not the centerpiece. The peptide gets the headline, but the protocol gets the result. That is also why the response data is so hard to pin down — when a patient improves on a protocol that simultaneously removes gluten, kills off SIBO, repopulates with sporulating probiotics, and adds BPC-157, the peptide is one variable in a five-variable change. The clinical anecdotal signal is strong; the variable-isolation evidence is weak. Both things are true at once.
What Mainstream Gastroenterology Says (and Doesn't)
Pull up the position statements from the American College of Gastroenterology, the American Gastroenterological Association, or the Crohn's & Colitis Foundation, and search for "BPC-157." You will find nothing. That silence is itself a position. Mainstream gastroenterology is not anti-BPC-157 in print; it has chosen not to engage with a substance that lacks Phase III human data. The criticism comes from individual academics rather than society guidelines.
The most cited skeptic in print is Patricia Brubaker, Ph.D., F.R.S.C., Professor Emerita of Physiology and Medicine at the University of Toronto, writing for McGill's Office for Science and Society. Brubaker argues there is "a near-total lack of evidence that BPC 157 is effective or even safe for use in humans," that the foundational animal data is concentrated in a single research group, and that the peptide has been categorized as a banned, unapproved substance by the World Anti-Doping Agency. Her central worry is mechanistic: BPC-157 stimulates angiogenesis and the nitric oxide system, both of which can be hijacked by tumor biology, and the absence of long-term human safety data makes that risk impossible to quantify.
Dr. Victor Prisk, an orthopedic surgeon writing for Prisk Orthopaedics and Wellness, makes the same argument in sharper form: BPC-157 strongly activates the FAK-paxillin signaling pathway and increases VEGFR2 expression, "the same pathways that promote healing can also be hijacked by cancer cells." He notes that a 2015 Phase I human trial enrolling 42 healthy volunteers (NCT02637284) was initiated to assess safety and pharmacokinetics, and that the researchers later cancelled submission of the results in 2016 — a non-publication that, in clinical research, is itself a red flag.
The contamination concern is the one mainstream voices and functional medicine voices actually agree on. A study cited in Drug Testing and Analysis found that 30% of online peptide products contained incorrect amino acid sequences and 65% had endotoxin levels above acceptable thresholds. USADA's 2017 testing of black-market peptide products found over 20% mislabeled or contaminated. The FDA's 2026 reconsideration is structured as a compounding-pharmacy pathway rather than a free-market approval for that exact reason — pulling the substance back under 503A pharmacy oversight makes sterile technique, sequence verification, and endotoxin testing at least theoretically enforceable.
Who Shouldn't Use BPC-157 for Gut Issues
The safety conversation about BPC-157 is unusually bimodal. On one side: a 2024 task-force survey suggesting hundreds of thousands of compounding-pharmacy prescriptions filled with no reported side effects. On the other: the theoretical pathways above and the simple fact that long-term human safety data does not exist. Both can be true at once. The standard exclusions in functional medicine practice are conservative for that reason.

Active cancer or recent cancer history. The firmest contraindication in the practitioner literature. BPC-157's stimulation of angiogenesis, VEGFR2, and FAK-paxillin pathways is the cellular machinery aggressive tumors co-opt to grow new blood supply and invade neighboring tissue. As a 2025 review in Pharmaceuticals put it, BPC-157 "may not be the right choice, especially in situations where we are not aware of the presence of cancer cells in our body." Sikiric's group has rebutted this with a controlled-modulation framing, but the prudent clinical posture is exclusion if there is any active or recent malignancy.
Pregnancy and breastfeeding. Mark Hyman's peptide guidance is explicit: "Pregnant or breastfeeding individuals should avoid certain peptides, and anyone with cancer or a history of cancer should consult a healthcare provider before starting peptide therapy." There is no human pregnancy safety data for BPC-157. Treat it as a hard exclusion.
Significant cardiovascular or clotting risk factors. Because BPC-157 modulates the nitric oxide system and vascular endothelial signaling, clinicians who use it in cardiovascular patients do so cautiously and not without specialty input.
Competitive athletes. The World Anti-Doping Agency classifies BPC-157 as an S0 "Unapproved Substance," which means it is banned in competition and out of competition for any WADA-tested sport. The US Department of Defense also prohibits it. If you compete at any sanctioned level — including masters-level events — assume any peptide compounded outside FDA-approved channels is disqualifying.
Patients sourcing from unverified suppliers. This may be the largest practical risk for the average reader. Until and unless the PCAC vote in July 2026 results in BPC-157 being removed from Category 2 and added to the 503A bulks list, the supply chain remains fragmented between gray-market online vendors and a small number of functional clinics still working with cooperative compounding pharmacies. Buying "research-only" peptides online, mixing them with bacteriostatic water at the kitchen table, and self-injecting is the worst version of this scenario — and it is exactly what the 30%-incorrect-sequence and 65%-high-endotoxin numbers from the Drug Testing and Analysis paper describe.
So what is the practical bottom line? If you are a candidate — no cancer history, not pregnant, no major clotting risk, not an active competitor — and you have a well-defined gut problem (especially ulcerative colitis or NSAID-induced gastric injury), the path through a board-certified functional medicine practitioner who works with a 503A pharmacy is the highest-evidence, lowest-risk version of trying BPC-157. If any of those exclusions apply, or you are tempted by the online research-chemical route, the math is not in your favor — wait for the July 2026 PCAC outcome and the regulated supply that follows.
Frequently Asked Questions
Is BPC-157 FDA-approved for ulcerative colitis or any gut condition?
No. As of April 2026, BPC-157 is on the FDA's Category 2 bulk substances list and is not approved for any indication. The July 23–24, 2026 Pharmacy Compounding Advisory Committee meeting will consider adding it to the Section 503A bulks list with a proposed indication of ulcerative colitis. Even if approved, that pathway permits compounding-pharmacy production for prescriptions, not full pharmaceutical approval.
Does the human ulcerative colitis trial actually support oral or capsule use?
The Ruenzi et al. Phase II trial used an 80 mg BPC-157 enema, not an oral capsule. So the strongest human gut-healing evidence for BPC-157 is for rectal/topical delivery in colitis specifically. Oral capsule use is supported by mechanism (acid stability) and clinical practice but not by an equivalent human RCT.
How is BPC-157 different from glutamine, zinc carnosine, or slippery elm for gut healing?
Glutamine fuels enterocyte metabolism, zinc carnosine stabilizes the gastric mucosa, and slippery elm provides a mucilage barrier. BPC-157's distinct contribution is angiogenic — it signals new blood vessel formation at the wound bed and preserves tight-junction proteins (ZO-1, occludin) directly. Functional medicine protocols typically stack BPC-157 with these other agents rather than substitute it.
Is the cancer-promotion concern theoretical or proven?
It is theoretical but biologically plausible. BPC-157 upregulates VEGF, VEGFR2, and FAK-paxillin signaling, which are pathways tumors hijack for angiogenesis and invasion. No human study has shown BPC-157 accelerating tumor growth, but no adequately powered long-term human safety study exists either. The standard clinical posture is exclusion if there is active or recent malignancy.
Should I wait for the July 2026 FDA PCAC outcome before trying it?
If you are not in active gut crisis, waiting is the more conservative choice — the PCAC outcome will determine whether US compounding pharmacies can legally supply BPC-157 again, which dramatically improves the supply-chain quality story. If you have active ulcerative colitis or another defined gut condition and are working with an experienced functional medicine practitioner, that conversation is appropriate now, but stay away from online research-chemical sources regardless of the timing.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.