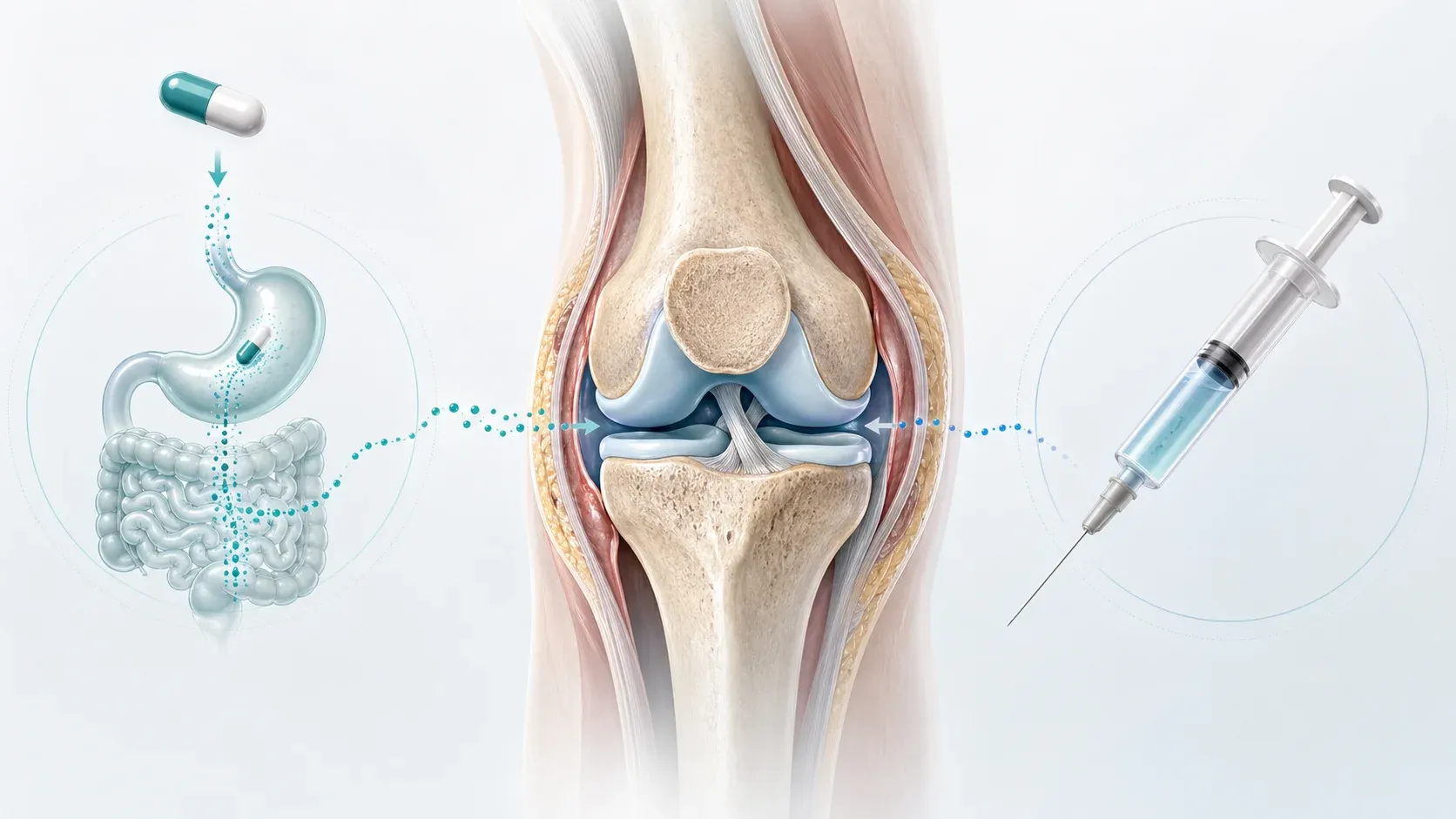
From Injection to Tablet: How Oral GLP-1 Therapy Became Reality
GLP-1 receptor agonists have transformed the management of type 2 diabetes and obesity over the past two decades. Drugs like liraglutide, dulaglutide, and semaglutide delivered remarkable improvements in blood sugar control and body weight reduction. But they all shared one limitation: every dose required a needle.
The journey toward an oral GLP-1 formulation began in earnest when Novo Nordisk partnered with Emisphere Technologies in 2007 to license the Eligen drug delivery platform. Emisphere had developed a compound called SNAC (sodium N-[8-(2-hydroxybenzoyl)amino] caprylate) that could enhance the absorption of large molecules through the stomach lining. By 2020, Novo Nordisk acquired Emisphere outright for $1.8 billion, reflecting the strategic importance of this technology.
The first milestone arrived in September 2019, when the FDA approved Rybelsus (oral semaglutide) for type 2 diabetes. It became the first oral GLP-1 receptor agonist ever brought to market and the first oral peptide drug co-formulated with SNAC, according to a comprehensive review published in Therapeutic Advances in Endocrinology and Metabolism. The European Medicines Agency followed with approval in March 2020.
More recently, the FDA approved oral Wegovy (semaglutide 25 mg) specifically for weight management, making it the first oral GLP-1 for weight loss in adults. Novo Nordisk launched the product at approximately $149 per month without insurance. For those exploring the broader landscape of GLP-1 weight loss drugs, their safety profiles, and alternatives, these oral formulations represent a significant shift in how patients can access these therapies.
Key milestone: Oral semaglutide (Rybelsus) became the first FDA-approved oral GLP-1 receptor agonist in September 2019, ending the injectable-only era for this drug class.
Why Delivering Peptides by Mouth Remains a Pharmacological Challenge
The human gastrointestinal tract evolved to break down proteins efficiently, which is precisely the problem for oral peptide drugs. GLP-1 receptor agonists are peptides — chains of amino acids that the body treats much like dietary protein. Three fundamental barriers stand between an oral GLP-1 tablet and therapeutic blood levels.
| Barrier | Mechanism | Impact on Oral GLP-1 |
|---|---|---|
| Enzymatic degradation | Pepsin, trypsin, chymotrypsin, and DPP-IV break down peptide bonds | Most of the oral dose is destroyed before reaching the bloodstream |
| Low membrane permeability | GLP-1 analogs (~4 kDa) are too large for passive diffusion across GI epithelium | Even intact peptide molecules struggle to cross the gut lining |
| Gastric acid instability | Stomach pH activates pepsinogen to pepsin, accelerating peptide breakdown | The acidic environment compounds enzymatic degradation |
Research published in Scientific Reports details how these barriers collectively reduce oral peptide bioavailability to a fraction of what injectable delivery achieves. For semaglutide, the numbers are striking: oral bioavailability sits at roughly 0.8%, compared to approximately 89% for the subcutaneous formulation, according to clinical pharmacokinetic data. That roughly 100-fold difference means oral doses must be dramatically higher to achieve equivalent plasma concentrations — 14 mg daily by mouth versus 1 mg weekly by injection in the original formulations.
Adding further complexity, within-subject variability for oral semaglutide absorption reaches approximately 137% for single doses. Daily dosing helps smooth this out, reducing steady-state variability to around 33%, though total variability still reaches up to 85%. This pharmacokinetic unpredictability is inherent to oral peptide delivery and shapes everything from dose titration schedules to the strict fasting requirements that patients must follow.
How SNAC Technology Made Oral Semaglutide Possible
SNAC does not work through a single mechanism. According to a detailed analysis in Clinical Diabetes and Endocrinology, the absorption enhancer operates through at least four complementary pathways that work together to get semaglutide through the stomach wall.
First, SNAC buffers the local pH around the dissolving tablet, creating a microenvironment that reduces the conversion of pepsinogen to pepsin. This slows enzymatic degradation of semaglutide in the immediate vicinity of the tablet. Second, SNAC interacts with and fluidizes the lipid membranes of gastric epithelial cells, increasing permeability for transcellular transport — notably, without disrupting tight junctions between cells. Third, SNAC reduces oligomerization of semaglutide molecules, improving their accessibility for absorption. Fourth, and critically, these effects are transient: membrane permeability returns to baseline within approximately 30 minutes of SNAC exposure.
The optimal formulation uses 300 mg of SNAC paired with each semaglutide tablet. However, this engineered absorption comes with strict conditions. Patients must take oral semaglutide on an empty stomach with no more than 120 mL of water, then wait at least 30 minutes before eating, drinking, or taking other medications. A pharmacological overview published in Drug Design, Development and Therapy found that absorption is limited or absent in the fed state — in one study, 14 of 25 participants had no detectable semaglutide exposure when dosed with food.
| Parameter | Oral Semaglutide | Subcutaneous Semaglutide |
|---|---|---|
| Bioavailability | ~0.8% | ~89% |
| Terminal half-life | ~1 week | ~1 week |
| Time to steady state | 4-5 weeks (daily dosing) | 4-5 weeks (weekly dosing) |
| Primary absorption site | Stomach | Subcutaneous tissue |
| Dose proportionality | Linear | Linear |
| Food requirement | Fasting mandatory | None |
For Rybelsus (type 2 diabetes), dosing starts at 3 mg daily for 30 days, escalates to 7 mg, and optionally increases to 14 mg. Oral Wegovy for weight management escalates through 1.5 mg, 4 mg, and 9 mg up to a maintenance dose of 25 mg. Understanding the full side effect profile of semaglutide, whether oral or injectable, remains important for informed treatment decisions — our guide on Ozempic safety and semaglutide side effects covers this in detail.
What PIONEER and OASIS Trials Reveal About Oral Semaglutide
The clinical evidence for oral semaglutide rests on two major trial programs. The PIONEER program (eight trials) established efficacy for type 2 diabetes, while the OASIS program tested higher doses for weight management.
PIONEER Program Highlights
| Trial | Population | HbA1c Change | Weight Change | Comparator |
|---|---|---|---|---|
| PIONEER 1 | Monotherapy | -0.6% to -1.1% | -2.3 kg (14 mg) | Placebo |
| PIONEER 2 | + metformin | -1.3% (14 mg) | Superior to empagliflozin | Empagliflozin |
| PIONEER 4 | + metformin/SGLT2i | Non-inferior to liraglutide | -1.2 kg advantage | Liraglutide 1.8 mg |
| PIONEER 5 | Moderate renal impairment | -0.8% vs placebo | -2.5 kg vs placebo | Placebo |
| PIONEER 6 | High cardiovascular risk | -1.0% | -4.2 kg | Placebo (MACE HR 0.79) |
Across the PIONEER program, oral semaglutide at 14 mg demonstrated HbA1c reductions of approximately 1.0-1.3% and modest weight reductions. For people managing diabetes through medication and lifestyle strategies, these results established oral semaglutide as a viable alternative to injectable therapies.
OASIS Program: Higher Doses for Weight Loss
The OASIS trials pushed oral semaglutide to higher doses and transformed the weight loss story. OASIS 1 tested a 50 mg dose over 68 weeks and achieved 15.1% body weight reduction versus 2.4% with placebo. The pivotal OASIS 4 trial, published in the New England Journal of Medicine, tested 25 mg over 64 weeks and showed 13.6% weight loss by intention-to-treat analysis. Among participants who adhered to treatment, weight loss reached 16.6%, and one-third achieved 20% or greater body weight reduction.
These results are remarkable because they close the efficacy gap that existed between oral and injectable semaglutide at their original approved doses. At 25-50 mg, oral semaglutide achieves weight loss comparable to injectable Wegovy (semaglutide 2.4 mg), which showed 14.9% weight loss in the landmark STEP 1 trial.
Orforglipron and the Small Molecule Revolution
While Novo Nordisk engineered a way to push a peptide through the stomach wall, Eli Lilly took a fundamentally different approach: design a molecule small enough that it does not need an absorption enhancer at all.
Orforglipron is not a peptide. It is a small molecule that activates the GLP-1 receptor with high selectivity but does not face the enzymatic degradation, permeability barriers, or absorption variability that plague oral peptides. There is no SNAC. No fasting requirement. No water volume restriction. Patients can take it with or without food.
Phase 3 results have been strong. The ATTAIN-1 trial, published in the New England Journal of Medicine, randomized 3,127 adults with obesity across four dose groups over 72 weeks. Weight loss was dose-dependent: 7.8% at 6 mg, 9.3% at 12 mg, and 12.4% at 36 mg, compared to 0.9% with placebo. At the highest dose, 77% of participants achieved at least 5% weight loss and roughly 40% exceeded 15%.
For type 2 diabetes, the ACHIEVE-1 trial demonstrated HbA1c reductions of 1.24% to 1.48% across dose levels. Perhaps most notable, the ATTAIN-MAINTAIN study showed that patients previously treated with injectable Wegovy or Zepbound maintained their weight loss when switched to oral orforglipron over 52 weeks, suggesting the small molecule can sustain effects initially achieved by injections.
Paradigm shift: Orforglipron represents a move from "making peptides survive the gut" to "designing non-peptide molecules that bypass the problem entirely." No absorption enhancer. No fasting. No water restrictions.
Other Pipeline Candidates
Pfizer's danuglipron, another small molecule GLP-1 agonist, showed 5.0-12.9% weight loss in phase 2b. However, Pfizer discontinued development in April 2025 after a case of potential drug-induced liver injury, combined with the intensely competitive landscape.
Viking Therapeutics is developing VK2735, an oral dual GLP-1/GIP agonist (a peptide). Phase 2 results showed up to 12.2% body weight reduction in just 13 weeks. Phase 3 VANQUISH trials are underway with results expected by 2027. If successful, VK2735 would bring dual-agonist potency — similar to tirzepatide (Zepbound) — into oral form for the first time.
Injectable vs Oral GLP-1: Bioavailability, Efficacy, and Adherence
Efficacy Comparison
Cross-trial comparisons must be interpreted cautiously, but the data provides a useful picture of how oral and injectable semaglutide stack up:
| Formulation | Weight Loss | Duration | Trial |
|---|---|---|---|
| Oral semaglutide 14 mg | ~2.3 kg vs placebo | 26 weeks | PIONEER 1 |
| Oral semaglutide 25 mg | -13.6% (ITT) | 64 weeks | OASIS 4 |
| Oral semaglutide 50 mg | -15.1% | 68 weeks | OASIS 1 |
| Injectable semaglutide 2.4 mg | -14.9% | 68 weeks | STEP 1 |
| Oral orforglipron 36 mg | -12.4% | 72 weeks | ATTAIN-1 |
| Injectable tirzepatide 15 mg | -20.9% | 72 weeks | SURMOUNT 1 |
A real-world retrospective study of 105 patients compared oral semaglutide 14 mg with injectable semaglutide 1 mg at 26 weeks. The oral group showed HbA1c reductions of 2.43% versus 1.70% for the injectable group, while weight loss was 3.70 kg versus 6.24 kg. Neither difference reached statistical significance, suggesting broadly comparable real-world performance at these dose levels.
Patient Preference and Adherence
Initial assumptions that patients would uniformly prefer pills over needles have not held up cleanly. The REVISE study, a cross-sectional survey of 600 patients with type 2 diabetes, found that before education about dosing procedures, 76.5% preferred oral delivery. After watching administration videos, preference dropped to 52.5% oral versus 47.5% injectable — essentially a coin flip. Over a quarter of participants switched their preference away from oral after learning about the 30-minute fasting requirement.
Real-world adherence data presents a mixed picture. Claims analysis shows oral semaglutide achieving 82.4% mean medication adherence at one year for type 2 diabetes, which was superior to all injectable formulations tested. However, for obesity without diabetes, weekly injectables showed higher persistence than oral formulations, with injectable semaglutide reaching 47.1% persistence at one year compared to lower rates for oral semaglutide. The daily dosing burden and fasting restrictions appear to offset the needle-free advantage in some patient populations. This is where non-peptide oral agents like orforglipron — which eliminate fasting requirements entirely — could meaningfully change the adherence calculus.
What These Advancements Mean for Patients
The pharmacokinetic progress in oral GLP-1 therapy has moved through two distinct phases. The first, exemplified by Rybelsus and oral Wegovy, proved that a peptide could be delivered orally with enough therapeutic effect to earn regulatory approval. The trade-off was strict dosing conditions and lower bioavailability that required substantially higher doses. The second phase, led by orforglipron, sidesteps the fundamental challenge of oral peptide delivery by abandoning the peptide altogether.
For people managing type 2 diabetes or obesity, these developments expand treatment options in concrete ways. Oral semaglutide provides a needle-free option for those who respond well to semaglutide but resist injections. The higher-dose oral formulations (25-50 mg) now deliver weight loss comparable to injectable Wegovy. And next-generation small molecules promise to remove the dosing complexity that limits adherence with current oral peptide formulations.
Cost remains a factor. Annual U.S. costs for oral GLP-1 agents range from $6,000 to $14,000, though oral Wegovy launched at approximately $149 per month. For those looking to support metabolic health through complementary approaches alongside pharmacotherapy, understanding dietary strategies that help prevent and manage diabetes and the role of evidence-based natural appetite suppressants can provide a more complete picture.
Bottom line: Oral GLP-1 therapy has evolved from a pharmacological curiosity to a clinically proven treatment class. With small molecule agents on the horizon, the next generation of oral formulations may finally deliver injectable-level efficacy without the needles or the fasting restrictions.
Frequently Asked Questions
What is the bioavailability of oral semaglutide compared to the injectable form?
Oral semaglutide has a bioavailability of approximately 0.8%, compared to roughly 89% for subcutaneous semaglutide. This means oral doses must be significantly higher to achieve therapeutic plasma levels — for example, 14 mg daily by mouth versus 1 mg weekly by injection. Despite this gap, the oral formulation reaches clinically effective blood concentrations through daily dosing with SNAC absorption enhancement.
Why do you have to take Rybelsus on an empty stomach?
The SNAC absorption enhancer in Rybelsus works by creating a protective microenvironment around the tablet as it dissolves in the stomach. Food in the stomach disrupts this process by diluting SNAC concentration, altering gastric pH, and physically interfering with tablet dissolution at the stomach wall. Clinical studies showed that many patients had no detectable semaglutide in their blood when dosed with food. The 30-minute post-dose fast ensures SNAC can do its job.
How does orforglipron differ from oral semaglutide?
Orforglipron is a small molecule, not a peptide, so it does not face the enzymatic degradation and absorption barriers that oral semaglutide must overcome with SNAC technology. This means orforglipron can be taken with or without food, has no water volume restrictions, and does not require a fasting window. It represents a fundamentally different pharmacological approach to oral GLP-1 receptor activation.
Can oral GLP-1 medications achieve the same weight loss as injections?
At higher doses, yes. The OASIS 4 trial showed oral semaglutide 25 mg achieving 13.6% weight loss (intention-to-treat) over 64 weeks, which is comparable to the 14.9% achieved by injectable semaglutide 2.4 mg (Wegovy) in the STEP 1 trial. The oral 50 mg dose in OASIS 1 reached 15.1% weight loss. Next-generation oral agents like orforglipron (12.4% at 36 mg) are also approaching injectable-level efficacy.
Are there oral dual or triple agonists in development?
Viking Therapeutics is developing VK2735, an oral dual GLP-1/GIP receptor agonist. Phase 2 results showed up to 12.2% body weight reduction in just 13 weeks. Phase 3 trials are underway with results expected by 2027. There is no oral triple agonist (GLP-1/GIP/glucagon) currently in clinical trials, though the injectable triple agonist retatrutide from Eli Lilly has shown up to 28.7% weight loss in phase 3 studies.
Related Articles
- GLP-1 Weight Loss Drugs: Safety, Side Effects, and Natural Alternatives — A broader look at GLP-1 receptor agonists, covering safety profiles, common side effects, and complementary natural approaches.
- Ozempic Safety and Side Effects: What Semaglutide Does to Your Body — Detailed breakdown of semaglutide-specific side effects and safety considerations for both oral and injectable forms.
- Diabetes: Causes, Symptoms, and Treatments — The Ultimate Guide — Comprehensive guide to understanding diabetes, its risk factors, and the full range of current treatment options.
- Diets That Help Prevent and Manage Diabetes: Ultimate Guide — Evidence-based dietary strategies that complement pharmacological therapy for blood sugar management.
- Natural Appetite Suppressants: The Ultimate Evidence-Based Guide — Research-backed natural compounds that support appetite regulation alongside or independent of GLP-1 therapy.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.