A Navy Veterinarian, Aging Dolphins, and an Overlooked Fat
The story behind pentadecanoic acid begins not in a nutrition lab, but in the waters of San Diego Bay. Dr. Stephanie Venn-Watson, a veterinary epidemiologist working with the U.S. Navy Marine Mammal Program, noticed something troubling in the early 2000s. The Navy's bottlenose dolphins, some of whom lived past 50 years old (wild dolphins rarely make it past 30), were developing a cluster of conditions that looked eerily human: elevated cholesterol, insulin resistance, fatty liver disease, and even Alzheimer's-like brain changes.
When Venn-Watson combed through bloodwork from the healthiest geriatric dolphins, one nutrient kept surfacing at the top: a 15-carbon saturated fatty acid called C15:0, or pentadecanoic acid. The dolphins eating fish with higher C15:0 content had far better metabolic profiles than those whose diets had shifted toward leaner fish over the years. That single observation launched a decade of research that is now challenging one of the most deeply held assumptions in nutrition: that all saturated fats are harmful.
Worth noting: Before C15:0 entered the conversation, the last fatty acid proposed as essential was alpha-linolenic acid (omega-3), identified back in 1929. If the evidence holds up, C15:0 would be the first new essential fatty acid recognized in nearly a century.
The dolphin connection matters because it frames C15:0 research differently than most supplement stories. This was not a molecule dreamed up in a product lab. It emerged from veterinary epidemiology, then traced backward into human population data showing declining C15:0 blood levels correlating with rising metabolic disease. Whether that correlation reflects causation is the central question.
What Exactly Is Pentadecanoic Acid?
Pentadecanoic acid is a 15-carbon saturated fatty acid classified as an odd-chain fatty acid (OCFA). Most dietary fats are even-chain, containing 12, 14, 16, or 18 carbons. Odd-chain fats like C15:0 and its relative C17:0 (heptadecanoic acid) are comparatively rare in the diet and have been largely ignored by nutritional science until recently.
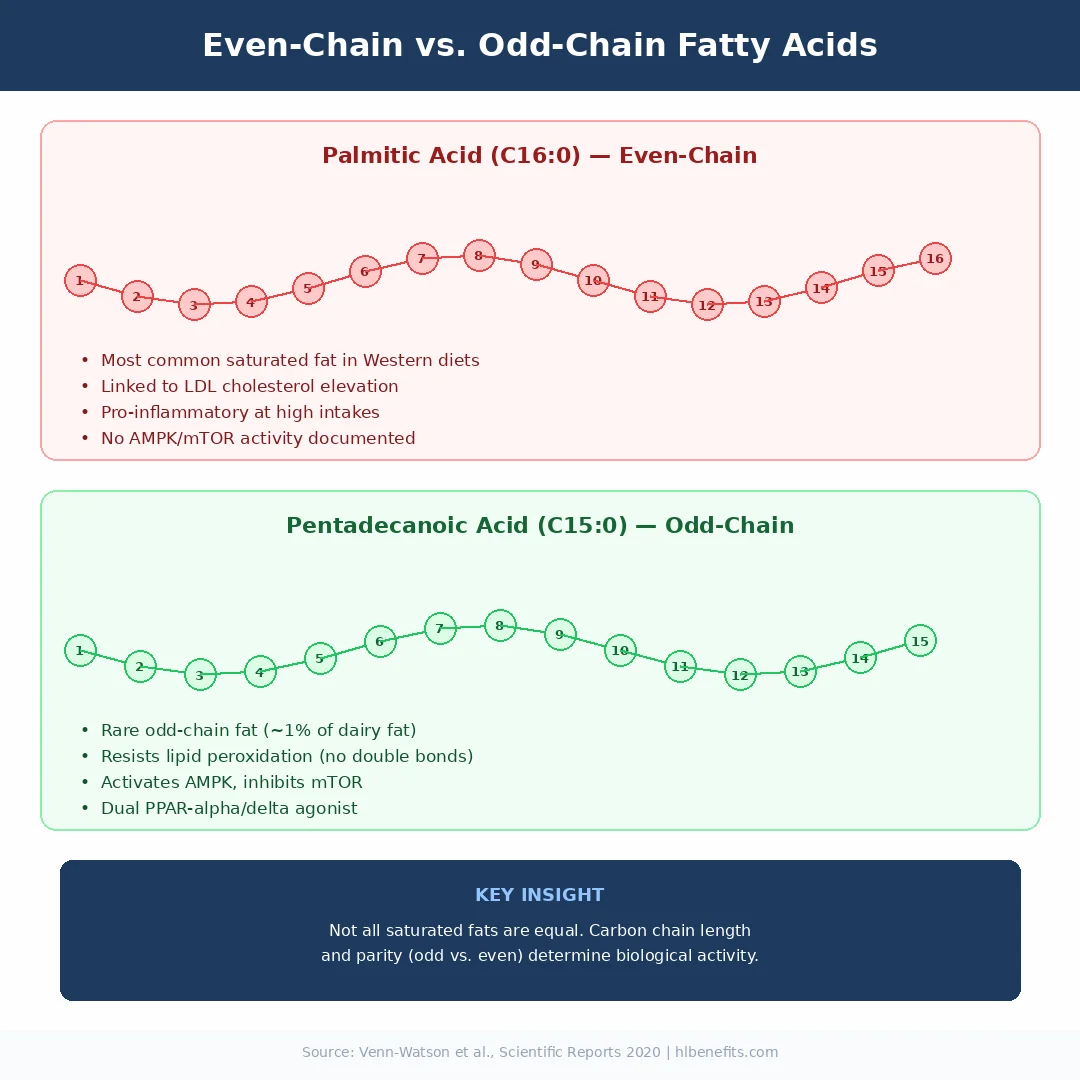
C15:0 is found primarily in the milkfat of ruminant animals. When cows, goats, and sheep digest fiber, bacteria in their rumen produce propionate as a fermentation byproduct. That propionate feeds into a metabolic pathway that builds odd-chain fatty acids, which then end up in the animal's milk and meat. Bovine milk fat contains roughly 1 to 2 percent odd-chain fatty acids by weight, with C15:0 accounting for about 1 percent and C17:0 about 0.5 percent.
This is part of why C15:0 levels have been declining in Western populations. Since the 1980s, dietary guidelines have pushed consumers away from full-fat dairy toward skim and low-fat alternatives. Whole milk consumption has dropped roughly fourfold in many countries over the past 40 years. As people followed that advice, their intake of odd-chain fatty acids fell in lockstep, and researchers are now asking whether that nutritional gap has consequences.
| Property | C15:0 (Pentadecanoic Acid) | Omega-3 (EPA/DHA) |
|---|---|---|
| Carbon chain | 15 carbons, saturated (no double bonds) | 20-22 carbons, polyunsaturated (multiple double bonds) |
| Oxidation stability | Highly stable (resists lipid peroxidation) | Prone to oxidation (requires cold storage) |
| Primary dietary source | Grass-fed dairy fat, some ruminant meats | Fatty fish, fish oil, algae |
| Cell membrane role | Strengthens and stabilizes membranes | Increases membrane fluidity |
| Discovery as essential | Proposed 2020 | Recognized 1929 |
| Current dietary trend | Declining (low-fat dairy shift) | Increasing (supplement awareness) |
One biochemical property that sets C15:0 apart from polyunsaturated fats like omega-3 is its stability. Because C15:0 has no double bonds in its carbon chain, it resists lipid peroxidation, the oxidative damage that degrades cell membranes and contributes to aging. Polyunsaturated fats, including the omega-3s that nutrition science has championed for decades, are inherently vulnerable to peroxidation precisely because of their double bonds. This does not make omega-3s less valuable, but it does highlight that membrane stability requires more than just fluidity.
There is also a question about whether C15:0 merely comes from diet or whether the body can make its own. A 2015 analysis in the American Journal of Clinical Nutrition raised this point, noting that C15:0 appears in non-dairy foods including beef, lamb, chicken, certain fish, and even vegetables like cabbage and cucumber. Some researchers suspect limited endogenous synthesis may occur, which would complicate the argument for true essentiality. The debate remains open.
How C15:0 Activates the Same Pathways as Leading Longevity Drugs
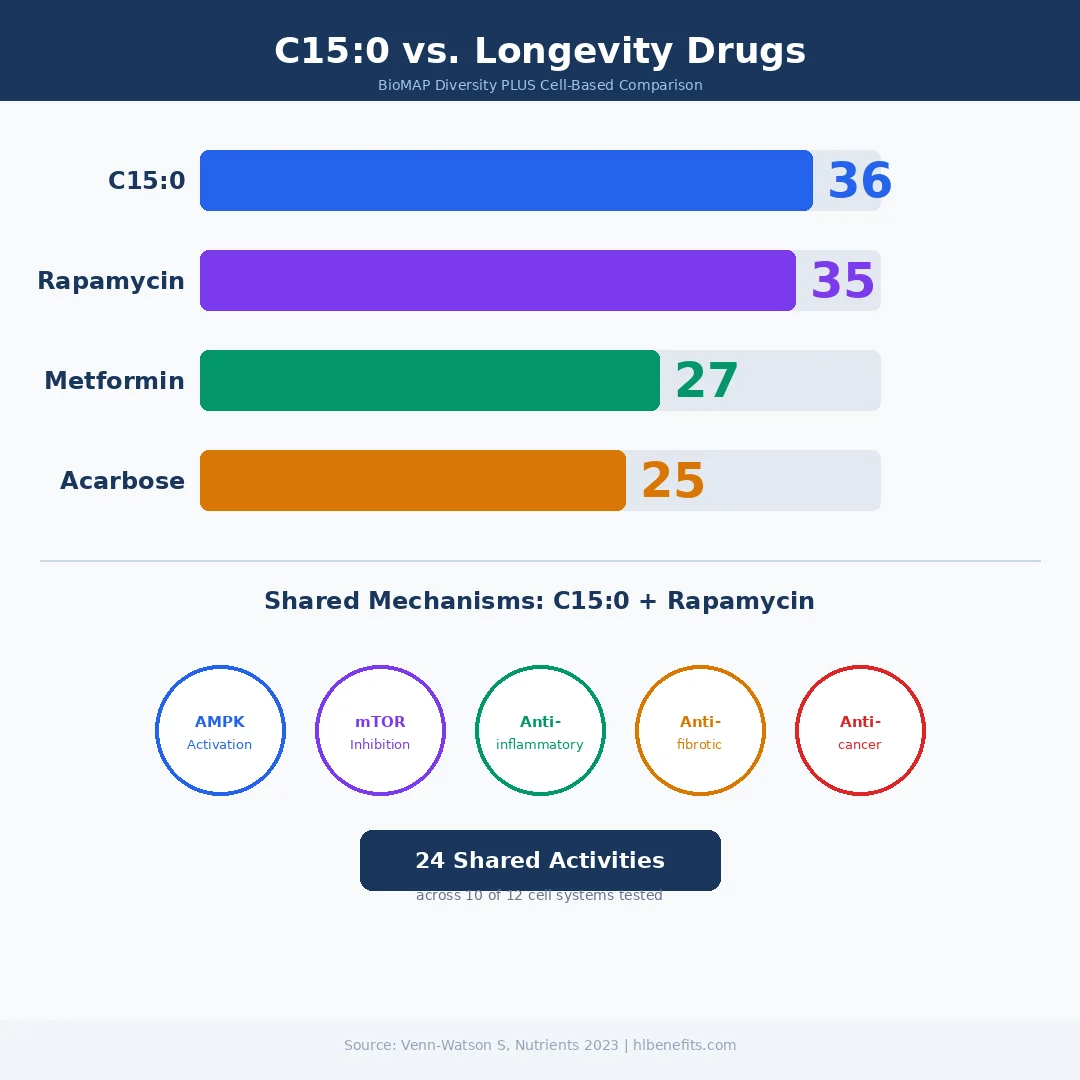
The most striking piece of C15:0 research to emerge in recent years involves a direct comparison with three of the most studied longevity compounds in geroscience: rapamycin, metformin, and acarbose. Published in Nutrients in 2023, the study used a BioMAP Diversity PLUS system, a human primary cell-based platform that measures how compounds affect inflammation, fibrosis, immune function, and tissue remodeling across 12 different cell systems simultaneously.
The results were unexpected. C15:0 demonstrated 36 clinically relevant biomarker activities across 10 of the 12 cell systems tested. Rapamycin, the gold standard geroprotective drug, showed 35 activities. Metformin had 27. Acarbose trailed at 25. What stood out more: C15:0 and rapamycin shared 24 overlapping activities across the same 10 systems, meaning the fatty acid was hitting many of the same anti-aging targets as a prescription immunosuppressant, but without the immune-suppressing side effects.
At the molecular level, C15:0 appears to work through at least three overlapping mechanisms that longevity researchers care about deeply:
| Mechanism | What C15:0 Does | Why Longevity Researchers Care |
|---|---|---|
| AMPK activation | Turns on the cell's energy-sensing master switch | AMPK activation mimics caloric restriction, the most reliable lifespan-extending intervention known |
| mTOR inhibition | Dampens the growth-signaling pathway | mTOR suppression is how rapamycin extends lifespan in mice; C15:0 achieves partial inhibition naturally |
| PPAR-alpha/delta agonism | Activates nuclear receptors controlling fat metabolism and inflammation | PPAR agonists improve insulin sensitivity and reduce systemic inflammation |
The foundational 2020 study in Scientific Reports showed that C15:0 is a dual partial agonist of PPARalpha and PPARdelta, reaching 65.8 percent and 52.8 percent of maximum activity respectively. Partial agonism is actually desirable here because full PPAR activation can cause unwanted side effects (as the diabetes drug rosiglitazone demonstrated). C15:0 sits in a pharmacological sweet spot: enough activation to improve metabolic function, not so much that it creates new problems.
The same study demonstrated that C15:0 repaired mitochondrial function and lowered reactive oxygen species in human cell cultures at concentrations between 10 and 50 micromolar. Mitochondrial dysfunction is one of the nine hallmarks of aging, and compounds that reverse it are among the most sought-after targets in geroscience.
Perspective check: C15:0 matched rapamycin in breadth of anti-aging biomarker activity in cell systems. But rapamycin has decades of animal lifespan data. C15:0 does not yet have a single completed lifespan study in mammals. Promising cell data does not guarantee real-world longevity effects.
The Cardiovascular and Metabolic Evidence
Population-level data on C15:0 and heart health is where the fatty acid makes its strongest epidemiological case. A 2025 narrative review in the World Journal of Cardiology synthesized 115 PubMed-indexed studies and found a consistent pattern: individuals in the highest quintile of blood C15:0 levels had 12 to 25 percent fewer cardiovascular events and approximately 14 percent lower risk of type 2 diabetes compared to those in the lowest quintile.
The proposed mechanisms are metabolically coherent. C15:0 appears to lower LDL cholesterol, reduce circulating IL-6 and TNF-alpha (two major inflammatory cytokines), enhance insulin sensitivity, and repair endothelial function through mitochondrial support. These are not fringe pathways. They overlap substantially with the mechanisms behind established cardiovascular interventions and align with what we know about how metabolic syndrome progresses.
But the review also surfaced important caveats. Data from the UK Biobank, one of the largest prospective cohort studies, showed null findings for some cardiovascular endpoints. And some analyses suggested a U-shaped relationship, where very high C15:0 levels were associated with worse outcomes. This is not unusual in nutritional epidemiology (selenium, vitamin D, and iron all show U-shaped curves), but it does mean that "more is better" is almost certainly the wrong framing.
| Health Outcome | Association with Higher C15:0 Levels | Strength of Evidence |
|---|---|---|
| Cardiovascular events | 12-25% reduction in highest vs. lowest quintile | Strong (multiple cohorts, meta-analyses) |
| Type 2 diabetes risk | ~14% reduction | Moderate-strong (consistent across studies) |
| All-cause mortality | Inverse association in several cohorts | Moderate (observational only) |
| NAFLD | Protective trend, limited data | Weak-moderate (small RCTs) |
| Cancer risk | Antiproliferative in 13.8% of tested cell lines | Preclinical only (no human cancer outcome data) |
Cancer research on C15:0 remains in early stages but shows intriguing signals. A 2025 study in Nutrients tested C15:0 against 94 human cancer cell lines. The fatty acid showed antiproliferative effects in 13 cell lines (13.8 percent), with non-Hodgkin B-cell lymphomas being most responsive. The researchers identified a genetic marker, CCND3 mutation, that appeared in 22 percent of C15:0-responsive cancers versus just 1.6 percent of non-responsive ones. This suggests C15:0 may eventually find a role in precision oncology, but that work is years from clinical application.
Cellular Fragility Syndrome: The Deficiency No One Is Testing For
In 2024, a paper in Metabolites introduced the concept of "Cellular Fragility Syndrome," a proposed condition where inadequate C15:0 in cell membranes makes cells structurally vulnerable to a form of death called ferroptosis. The hypothesis argues that cell membranes need at least 0.4 percent C15:0 content to maintain long-term structural integrity, and that levels below 0.2 percent create a danger zone where ferroptosis risk climbs sharply.
Ferroptosis differs from the apoptosis most people have heard of. It is driven by iron accumulation, reactive oxygen species, and peroxidation of polyunsaturated fatty acids in cell membranes. Because C15:0 is saturated, it resists peroxidation and may act as structural reinforcement preventing the cascading lipid damage that triggers ferroptotic death.
The paper estimated roughly one in three people globally may have C15:0 below this threshold, particularly in populations following low-fat dietary patterns. No standard blood panel tests for C15:0, and no dietary guidelines currently recommend its intake.
Independent support for C15:0's physiological importance came from a 2025 study in the Journal of Nutritional Biochemistry. French researchers (not affiliated with the Fatty15 supplement company) fed C15:0 to rats that had been raised on essential fatty acid-deficient diets and found it increased early growth rates and triggered the synthesis of previously undescribed odd-chain polyunsaturated fatty acids. The appearance of these novel downstream metabolites suggests C15:0 may feed into biosynthetic pathways that nutritional science has not yet mapped.
Key distinction: The "Cellular Fragility Syndrome" concept was introduced by the co-founder of the Fatty15 supplement. While the underlying biochemistry of ferroptosis and membrane stability is well-established, the specific syndrome framing has not been validated by independent clinical studies.
Myths vs. Reality: What the Clinical Trials Actually Show
The gap between C15:0's impressive cell-based and epidemiological data and its clinical trial results is the elephant in the room. Only two randomized controlled trials of C15:0 supplementation in humans have been published, and their results should give anyone considering a supplement some pause.
The first trial enrolled 88 women with non-alcoholic fatty liver disease and compared C15:0 supplementation to a Mediterranean diet alone. The C15:0 group showed no advantage over Mediterranean diet alone on most outcomes, with only a modest additional reduction in LDL cholesterol. Given that Mediterranean diets already have strong evidence for metabolic improvement, this was a disappointing result for C15:0 advocates.
The second trial was smaller: 30 young, healthy adults taking Fatty15 (100 mg C15:0) versus placebo. After the supplementation period, there were no significant differences in body weight, waist circumference, cholesterol, blood sugar, or inflammatory markers between the two groups. For a compound compared to rapamycin in cell studies, these human results are underwhelming.
| Claim | Reality |
|---|---|
| "C15:0 is as powerful as rapamycin" | They share overlapping biomarker activities in cell-based assays, but C15:0 has zero animal lifespan data and limited human trial results |
| "C15:0 is the first essential fatty acid in 90 years" | The National Academies of Sciences does not currently recognize C15:0 as essential; only one independent group has explored essentiality criteria |
| "Saturated fats are all bad" | Odd-chain saturated fats behave differently from even-chain ones like palmitic acid; population studies consistently show positive associations with C15:0 |
| "You need a supplement to get C15:0" | Two servings of full-fat dairy daily can provide roughly 200 mg of C15:0, within the proposed therapeutic range |
| "Higher C15:0 is always better" | Some data suggests a U-shaped curve; very high levels may not be beneficial |
There is also the conflict-of-interest question. Most published C15:0 research comes from Dr. Venn-Watson and colleagues affiliated with Seraphina Therapeutics, the Fatty15 manufacturer. This does not invalidate peer-reviewed findings, but the evidence base is narrower than it first appears.
The Center for Science in the Public Interest reviewed the evidence in 2024 and concluded the supplement is not worth the cost given current evidence. Their core argument: the 100 mg dose in Fatty15 has never been specifically tested in a published trial, and cell-based results have not translated into meaningful clinical benefits in the two completed human studies.
Food Sources, Supplements, and Practical Dosing
If you are interested in raising your C15:0 levels, the most straightforward approach is dietary. Grass-fed dairy products are the richest available source. The table below shows approximate C15:0 content in common foods.

| Food Source | Approximate C15:0 Content | Notes |
|---|---|---|
| Whole milk (1 cup, grass-fed) | ~50-90 mg | Pasture-raised has higher OCFA content than grain-fed |
| Full-fat yogurt (1 cup) | ~40-70 mg | Greek yogurt retains more milkfat per serving |
| Butter (1 tablespoon) | ~50-80 mg | Grass-fed/pasture butter has up to 30% more OCFAs |
| Aged cheese (1 oz) | ~30-50 mg | Higher-fat cheeses contain proportionally more C15:0 |
| Heavy cream (1 tablespoon) | ~25-40 mg | Dense calorie source; small amounts add up |
| Lamb/mutton (3 oz) | ~15-30 mg | Ruminant meats contain modest amounts |
Based on the cell-based research, the proposed therapeutic range is 100 to 300 mg of C15:0 daily, which would produce circulating blood levels between 10 and 30 micromolar. Two servings of full-fat dairy from grass-fed sources can realistically deliver 150 to 250 mg, putting most people within range without supplementation.
The Fatty15 supplement provides 100 mg of purified C15:0 (as FA15, a vegan form). At roughly $1.50 to $2.00 per day, it targets people who avoid dairy or prefer a standardized dose. Whether that dose is sufficient remains uncertain given the clinical trial results discussed above.
For those choosing the dietary route, a few practical considerations apply. Grass-fed dairy consistently contains higher concentrations of odd-chain fatty acids than conventional dairy because pasture-raised animals produce more rumen propionate from fiber digestion. The quality of your fat sources matters, not just the quantity. Full-fat products are non-negotiable here, as skim and low-fat versions have had the very milkfat that carries C15:0 removed.
If you are currently following a carnivore or animal-based diet, you are likely already consuming meaningful amounts of C15:0 through whole dairy and ruminant meats without any supplementation. Conversely, if you follow a strict vegan diet with no dairy, your C15:0 intake is probably near zero unless you supplement.
Frequently Asked Questions
Is C15:0 officially recognized as an essential fatty acid?
Not yet. The National Academies of Sciences has not added C15:0 to its list of essential nutrients. A 2020 study in Scientific Reports argued C15:0 meets traditional essentiality criteria, but whether humans can synthesize it endogenously remains unresolved, and independent validation is limited.
Can I get enough C15:0 from diet alone without taking a supplement?
For most people who consume full-fat dairy, yes. Two daily servings of grass-fed dairy products (for example, a cup of whole milk and an ounce of aged cheese) can supply 100 to 200 mg of C15:0, which falls within the proposed beneficial range. The key is choosing full-fat versions from pasture-raised animals, as low-fat dairy and conventional grain-fed products contain substantially less C15:0.
Is C15:0 safe, and are there any side effects?
C15:0 has shown a favorable safety profile in published research. The 2020 Scientific Reports study found it was non-cytotoxic across all 12 human primary cell systems tested, and a head-to-head comparison found it was safer than EPA (omega-3), which showed toxicity to some cell systems at higher concentrations. No serious adverse events have been reported in the two published human trials. However, long-term safety data from large clinical trials is not yet available.
How does C15:0 compare to omega-3 fatty acids?
They serve complementary roles. Omega-3s reduce inflammation and improve membrane fluidity with decades of clinical evidence. C15:0 stabilizes membranes against oxidative damage and activates AMPK and PPAR pathways. A 2022 cell-based study found C15:0 had broader biomarker activity than EPA and was non-toxic at all concentrations, while EPA showed cytotoxicity at higher doses. They address different aspects of cellular health.
Should I stop taking omega-3 supplements and switch to C15:0?
No. Omega-3s have solid evidence from hundreds of clinical trials. C15:0 research is promising but far more limited, with only two small human trials completed. View C15:0 as a potential addition to, not replacement for, proven interventions. Increasing full-fat dairy is a low-risk way to boost levels while evidence develops.
Related Articles
- NAD+ and NMN Supplements for Aging and Cellular Repair -- Another longevity-focused compound targeting mitochondrial function and cellular energy.
- Rapamycin: The Most Studied Longevity Drug -- The prescription geroprotector that C15:0 was compared against in cell-based assays.
- Spermidine, Autophagy, and Anti-Aging: From Everyday Foods to Longevity -- A dietary compound that activates autophagy, another core aging pathway.
- Are Seed Oils Toxic? What the Evidence Actually Says -- Context on the broader debate about dietary fats and their health effects.
- Chronic Inflammation and Disease: The Anti-Inflammatory Living Guide -- How systemic inflammation drives the conditions C15:0 research is targeting.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.