Why BPC-157 Dosing Advice Is So Uncertain
BPC-157 dosing has a strange problem: the internet has a neat answer before medicine has done the neat work. Search results make it look as if there is a standard protocol. The evidence says something more frustrating: BPC-157 is a synthetic peptide with serious research interest, but it does not have an approved human dosing label.
Operation Supplement Safety describes BPC-157 as an unapproved drug, says it is not a dietary ingredient, and says there is little to no reliable scientific evidence supporting its safety or effectiveness in humans. Sport Integrity Australia is similarly blunt, calling it an experimental drug that is not approved by the TGA or any global regulatory authority for human use. A dosage guide is only as solid as the dose-finding trials behind it.
Think of dosing like setting the oven temperature for a cake. If a recipe has been tested hundreds of times, the temperature means something. If people are guessing from a different oven, a different pan, and a different batter, the number may still be useful, but it is not the same kind of knowledge. Most BPC-157 dose ranges are practical conventions built from animal work, clinic experience, and gray-market use, not official human protocols.
The cleanest answer is not "take X." It is "there is no approved BPC-157 dose, and every number should be treated as experimental."
The Therapeutic Goods Administration's scheduling discussion is a good example of why regulators are wary. In its interim decision, the TGA said BPC-157 claims for healing stomach ulcers, tendons, joints, nerves, skin, and the intestinal tract were largely unsubstantiated. The same document said the evidence base included only a small number of peer-reviewed studies that largely came from one laboratory.
The TGA also said the proposal followed 48 referrals for importation of BPC-157 since July 1, 2022. That detail does not prove harm. It shows demand, promotion, and importation were moving faster than the regulator's comfort with the evidence. When a compound becomes popular before dose-finding is settled, the public ends up crowdsourcing details that would normally come from labeling, phase trials, and post-market surveillance.
That does not mean every practical dose people discuss is imaginary. It means the confidence level is different. A clinician-supervised experimental protocol, an animal pharmacokinetic paper, a wellness-clinic blog, and a peptide vendor calculator should not carry the same weight in your decision-making.

Common BPC-157 Dose Ranges and What They Really Mean
Most online protocols orbit the same neighborhood: a few hundred micrograms per day. Swolverine's practical guide says typical daily doses fall between 200 mcg and 1,000 mcg, with 500 mcg often described as a commonly reported dose. Another clinic timing guide says standard protocols often begin at 250-500 mcg daily.
Many guides blur the most important distinction: those are not official dose instructions. BSCG notes that no clinically validated dosing regimen exists for BPC-157, and OPSS says the marketed benefits are mainly based on lab or animal studies rather than comprehensive human trials. The practical range can help you understand what people mean when they say "low dose" or "standard dose," but it should not be mistaken for a prescription.
| Commonly Discussed Range | How It Is Usually Framed | Evidence Confidence |
|---|---|---|
| 200-500 mcg daily | Conservative or common practical range for general repair discussions | Practical guidance, not approved dosing |
| 250-500 mcg once or twice daily | Often discussed for tendon, ligament, and joint use | Clinic/vendor guidance plus animal extrapolation |
| 500-1,000 mcg daily | Often discussed for higher-dose or oral protocols | Lower confidence, especially for systemic claims |
If a source presents a BPC-157 dose with total certainty, distrust the certainty. A better guide tells you where the number came from. For BPC-157, the answer is usually "animal studies, practitioner habits, and anecdotal use," not "large human dose-ranging trial."
That distinction changes how you read every dose chart. A low number does not automatically mean low risk, because purity, sterility, route, injection technique, diagnosis, and medical history all change the risk picture. A higher number does not automatically mean better healing, because the target tissue may not be receiving more useful signal. The dose is one dial on the dashboard. With BPC-157, several other dials are either uncalibrated or missing.
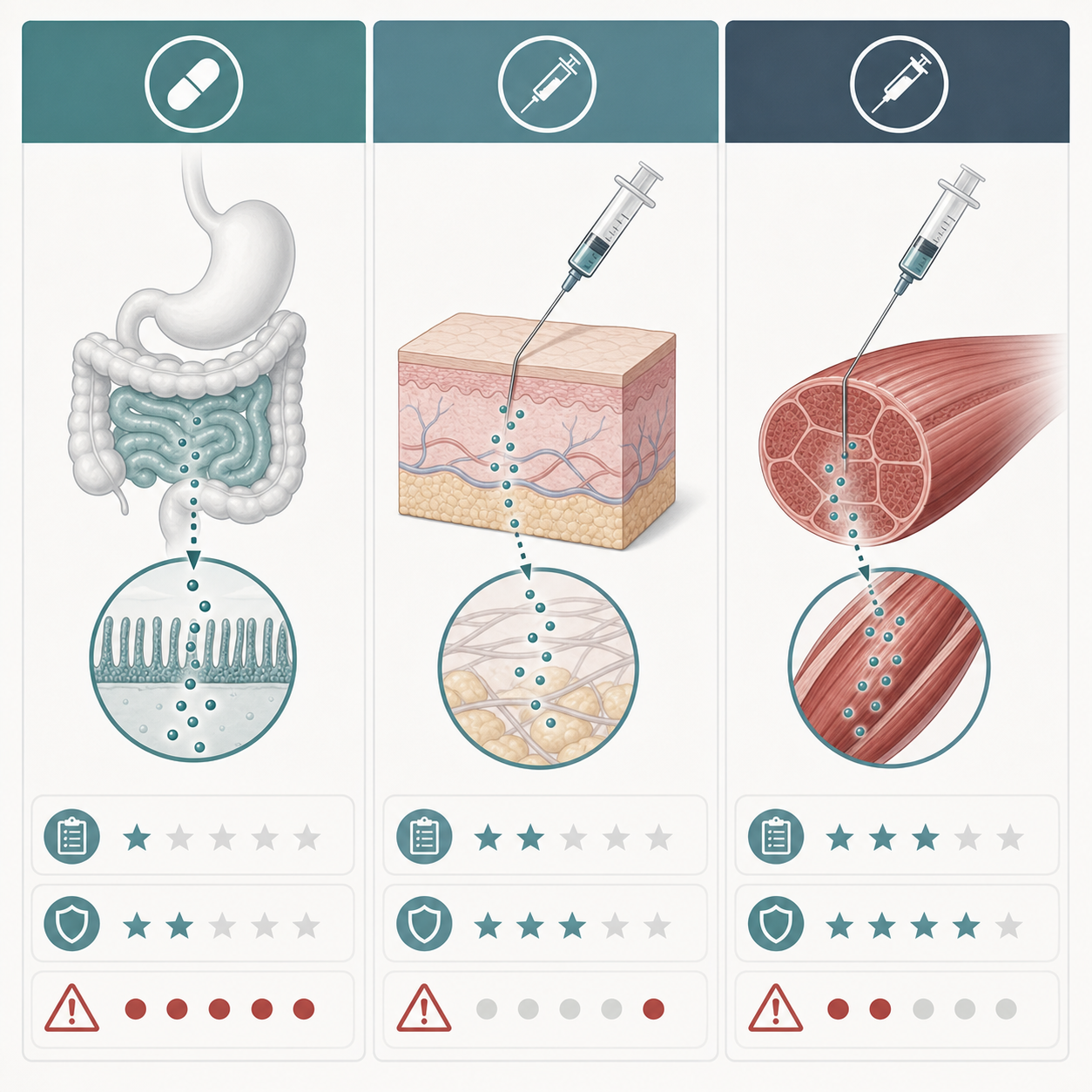
Oral vs. Injectable BPC-157: Timing and Practical Differences
Route changes the whole conversation. Injectable BPC-157 is usually discussed for musculoskeletal problems because it bypasses digestion and can be placed near the injured area. Oral BPC-157 is usually discussed for gut-focused goals because it is marketed as more local to the digestive tract.
Rupa Health says injections are typically subcutaneous or intramuscular and are used near the site of injury or inflammation, while oral forms may have lower bioavailability but may be more relevant to gastrointestinal tissues. Swolverine's route comparison similarly describes injectable forms as higher-bioavailability and oral forms as easier but less consistent outside the gut.
In the body, route is like choosing a delivery service. A package dropped at the correct building is different from a package sent through a central warehouse and hoping it reaches the right room. That does not prove injections work for every injury, but it explains why injection-focused protocols dominate musculoskeletal discussions.
| Route | Common Practical Use | Main Caveat |
|---|---|---|
| Subcutaneous injection | Systemic or local injury-focused use | Sterile technique and product quality matter |
| Intramuscular injection | Deep tissue or localized musculoskeletal use | Higher technique burden and discomfort risk |
| Oral or sublingual | Gut-focused use or injection-avoidant protocols | Lower and less predictable systemic exposure |
Animal pharmacokinetic data adds one useful caution. A peer-reviewed review reports that intramuscular BPC-157 bioavailability was about 14-19% in rats and 45-51% in beagle dogs. The same review reports peak concentration after intramuscular dosing at 3 minutes in rats and roughly 6.33-8.67 minutes in beagle dogs. Those numbers show that route affects exposure, but they do not give humans a clean conversion chart.
For a first-time user, "oral or injection?" is too narrow. Better questions come first: what goal am I treating, what evidence supports that route, and who is checking the sterile technique, product identity, and adverse events? Without those answers, route choice becomes a confidence game.
It also helps to separate convenience from evidence. Oral drops and capsules are easier to live with, travel with, and discuss in wellness content. Injectable routes carry a bigger burden: mixing, storage, sterile supplies, local irritation, and the possibility of introducing contaminants below the skin. Neither route solves the core evidence problem. A route can be practical and still experimental.
How Long People Use BPC-157 and When They Stop
Cycle length is where online advice gets especially tidy. You will see short cycles for sprains, medium cycles for post-surgical recovery, and longer cycles for chronic issues. That structure is useful as a vocabulary map, not as a validated medical schedule.
Swolverine's practical dosage guide describes 2-4 weeks for minor injuries and up to 6 weeks for post-operative recovery. Natura Dermatology says some chronic cases may need 8-12 weeks instead of 4-6 weeks. Real Peptides discusses rest-cycle ideas, but BSCG's warning that no validated dosing regimen exists should sit next to every one of those timelines.
| Use Case Discussed Online | Common Cycle Language | How to Interpret It |
|---|---|---|
| Minor soft-tissue injury | 2-4 weeks | A practical convention, not a trial-proven endpoint |
| More severe or post-operative recovery | 4-8 weeks | Requires medical context, especially after surgery |
| Chronic or gut-focused protocols | 8-12 weeks | Higher uncertainty because long-term human data are thin |
The most useful stop rule is not a calendar date. It is a checkpoint. If there is no meaningful improvement, new symptoms appear, the product source is unclear, or a clinician cannot explain why the protocol continues, the cycle has already become too vague.
Some guides talk about a 6-week non-response checkpoint. Natura Dermatology says that after 6 weeks without improvement, dosing, product quality, administration, or the underlying diagnosis should be questioned. That is a reasonable troubleshooting idea, but it is not a validated BPC-157 stopping rule.
Rest periods are even less proven. Real Peptides discusses breaks and time-off logic, while broader safety sources emphasize the absence of controlled human data. Treat a "washout" recommendation like a seatbelt made from assumptions: better than ignoring risk, but not proof that the trip is safe.
The strongest practical stop rule is diagnosis-based. If pain is worsening, swelling is spreading, gut symptoms are bleeding or severe, or post-surgical recovery is not following the expected course, another peptide cycle is the wrong response. Reassess the problem. BPC-157 marketing often talks about repair as if more repair signal is always better, but bodies also need correct loading, nutrition, sleep, infection control, and sometimes imaging or surgery. A peptide cannot compensate for a missed diagnosis.

Safety Checkpoints Before and During Use
The best human safety signal in this research set is not a microgram protocol. It is a tiny IV pilot study. Edwin Lee and Kailynd Burgess reported a PubMed-indexed pilot in which 10 mg was infused on day 1 and 20 mg on day 2 in two adults, with fasting labs and vital signs monitored. The authors reported no measurable effects on heart, liver, kidney, thyroid, or blood glucose biomarkers and no side effects.
That sounds reassuring until you look at the size of the bridge it is being asked to support. The study had two participants, both had prior IV BPC-157 exposure, and the paper says future studies are needed to confirm safety. Two people can tell you that something did not obviously go wrong in that setting. They cannot tell you the rare-event rate, cancer risk, fertility implications, immune reactions, drug interactions, or what happens after months of repeated use.
The exclusion criteria are also practical. The full paper excluded people with chronic kidney disease, diabetes, hypothyroidism, liver failure, pregnancy, or heart conditions. If a safety study leaves those groups out, those groups should not use the study as reassurance.
Product quality is part of dosing too. The IV pilot used a compounded product with a certificate of analysis and endotoxin testing. That detail matters because a vial label is only a dose if the vial contains what it says, is sterile enough for the route, and is measured correctly.
A good safety conversation should feel boringly specific. What is the lot number? Who tested identity? Was endotoxin testing done? What sterile supplies are used? Which symptoms trigger stopping? Which labs or vital signs will be checked? Those questions are not bureaucratic clutter. They are the difference between "a dose" and "a mystery amount of a mystery product entering the body by a route that may bypass normal defenses."
- Do not treat a label claim as proof of identity or sterility.
- Do not keep escalating dose when the diagnosis is uncertain.
- Do not use BPC-157 to avoid evaluation of a tendon tear, infection, inflammatory bowel disease flare, or post-surgical complication.
- Do stop and seek medical help for allergic symptoms, fever, spreading redness, severe pain, chest symptoms, neurologic symptoms, or any unexpected systemic reaction.
The half-life detail is useful but easy to misuse. The IV paper's pharmacokinetic discussion says BPC-157 half-life was less than 30 minutes in rat and beagle dog work. A short half-life does not automatically mean repeated dosing is safe. It only means the measured compound clears quickly in those models.

A Safer Decision Framework for First-Time Users
If you are reading a dosage guide because you want permission, this is the wrong compound to ask that from. BPC-157 sits in a gray zone: biologically interesting, heavily marketed, widely used outside approvals, and still missing the human trials that would turn a dosing culture into a dosing standard.
For athletes, the answer is clearer. Sport Integrity Australia lists BPC-157 as S0, non-approved, and prohibited at all times. OPSS says it is also on the DoD prohibited list and warns service members to avoid products containing BPC-157 regardless of whether they are taken by mouth, injection, or nasal spray.
The TGA also shows why "research use only" labels are not a safety plan. Its interim decision said BPC-157 had been identified in unapproved injectable, oral, powder, liquid, and nasal spray formulations. It also cited concern that research-only disclaimers were insufficient to deter consumer misuse.
| Question | Safer Answer | Red Flag |
|---|---|---|
| Is there an approved indication? | No approved human therapeutic use | Vendor claims it is proven for your condition |
| Is the dose official? | No validated human dosing regimen | Exact dose promised without medical evaluation |
| Is the product verified? | Identity, sterility, endotoxin, and lot documentation reviewed | Research chemical label or no certificate trail |
| When should it stop? | Stop for adverse effects, non-response, unclear diagnosis, or clinician concern | Open-ended use because "healing peptides are natural" |
A responsible BPC-157 conversation starts with the problem, not the vial. What injury or condition is being treated? Is there a diagnosis? What approved therapies have been tried? What risk factors are present? What endpoint would prove it is helping? What endpoint would prove it should stop?
That is not as satisfying as a tidy dose chart. It is more honest. With BPC-157, the safest "dosage guide" is the one that makes the uncertainty impossible to miss.
The most reader-friendly answer is also the least viral one: do not start with the highest dose a forum repeats, and do not continue because a calendar says the cycle is not done. Start with the diagnosis, the evidence quality, the route risk, the product documentation, and the exit plan. If any one of those is missing, the dosage question is premature.

Frequently Asked Questions
What is the standard BPC-157 dose?
There is no approved standard BPC-157 dose. Practical guides often discuss 200-1,000 mcg per day, but BSCG notes that no clinically validated dosing regimen exists.
Is oral BPC-157 as effective as injectable BPC-157?
Not for every goal. Practical route guides usually frame injectable BPC-157 as more relevant for musculoskeletal injuries and oral BPC-157 as more GI-focused because oral systemic absorption is less predictable.
How long do people usually take BPC-157?
Common practical guides describe 2-4 week cycles for minor injuries, 4-8 week cycles for more serious recovery, and longer protocols for chronic issues, but these are not validated clinical rules.
When should someone stop taking BPC-157?
Stop and seek medical guidance if side effects appear, the original diagnosis is uncertain, the product source is unclear, or there is no meaningful improvement after a planned checkpoint. People covered by anti-doping rules should avoid it entirely because it is prohibited at all times.
Is BPC-157 safe?
Short-term human reports are small and cannot settle safety. A two-person IV pilot reported no adverse effects after 10 mg and 20 mg infusions, but regulators still warn that BPC-157 lacks enough human safety and efficacy evidence.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.