Your body already makes TUDCA -- just not very much of it
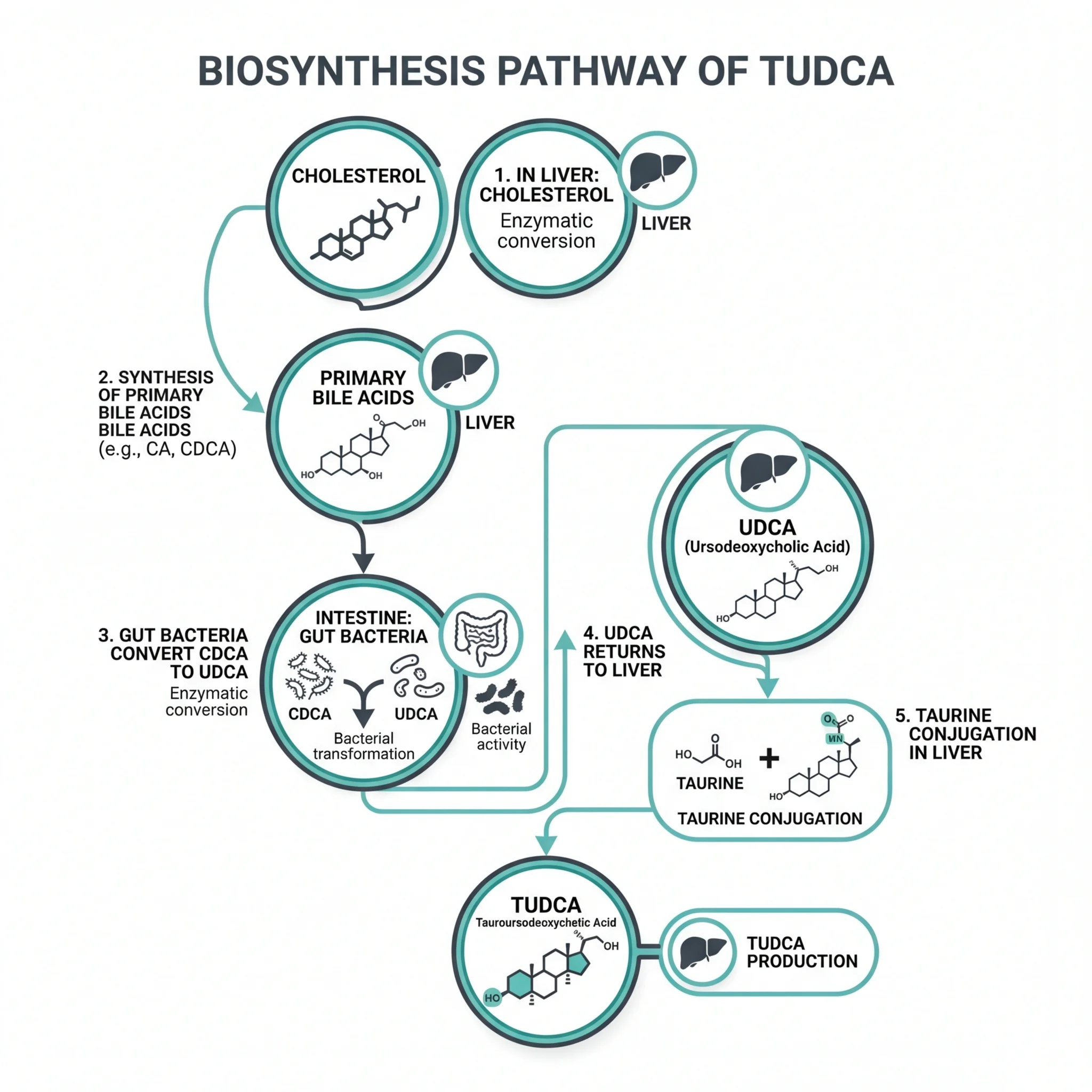
Tauroursodeoxycholic acid -- mercifully shortened to TUDCA -- is a bile acid your body produces in tiny amounts through an indirect, multi-step process. Your liver synthesizes primary bile acids from cholesterol. Those acids travel to your intestines, where gut bacteria convert a small fraction into ursodeoxycholic acid (UDCA). UDCA then returns to the liver through enterohepatic circulation, picks up a taurine molecule, and becomes TUDCA. The whole cycle yields trace quantities. Bears, for comparison, produce TUDCA at concentrations up to 75% of their total bile acid pool, which is why bear bile has been used in traditional Chinese medicine for centuries.
What makes TUDCA unusual among bile acids is its water solubility. Most bile acids are hydrophobic -- they act as biological detergents, dissolving fats but also potentially damaging cell membranes when they accumulate. TUDCA sits at the opposite end of the hydrophobicity spectrum. Instead of dissolving membranes, it stabilizes them. Most of what TUDCA does in the body traces back to this basic physical characteristic.
TUDCA vs. UDCA at a glance: UDCA (the parent compound, sold as the prescription drug Ursodiol) is FDA-approved for primary biliary cholangitis. TUDCA is the taurine-conjugated form with better oral absorption, greater water solubility, and potentially broader biological activity. TUDCA is sold as a dietary supplement in the United States.
The taurine conjugation is more than a minor chemical tweak. Research published in the journal Cells shows that TUDCA is fully ionized and water-soluble across a wide range of pH values, which means it absorbs better from the intestine and reaches higher concentrations in bile compared to unconjugated UDCA. If you are already familiar with taurine's role in heart and brain health, TUDCA adds another dimension to why this amino acid keeps showing up in longevity research.
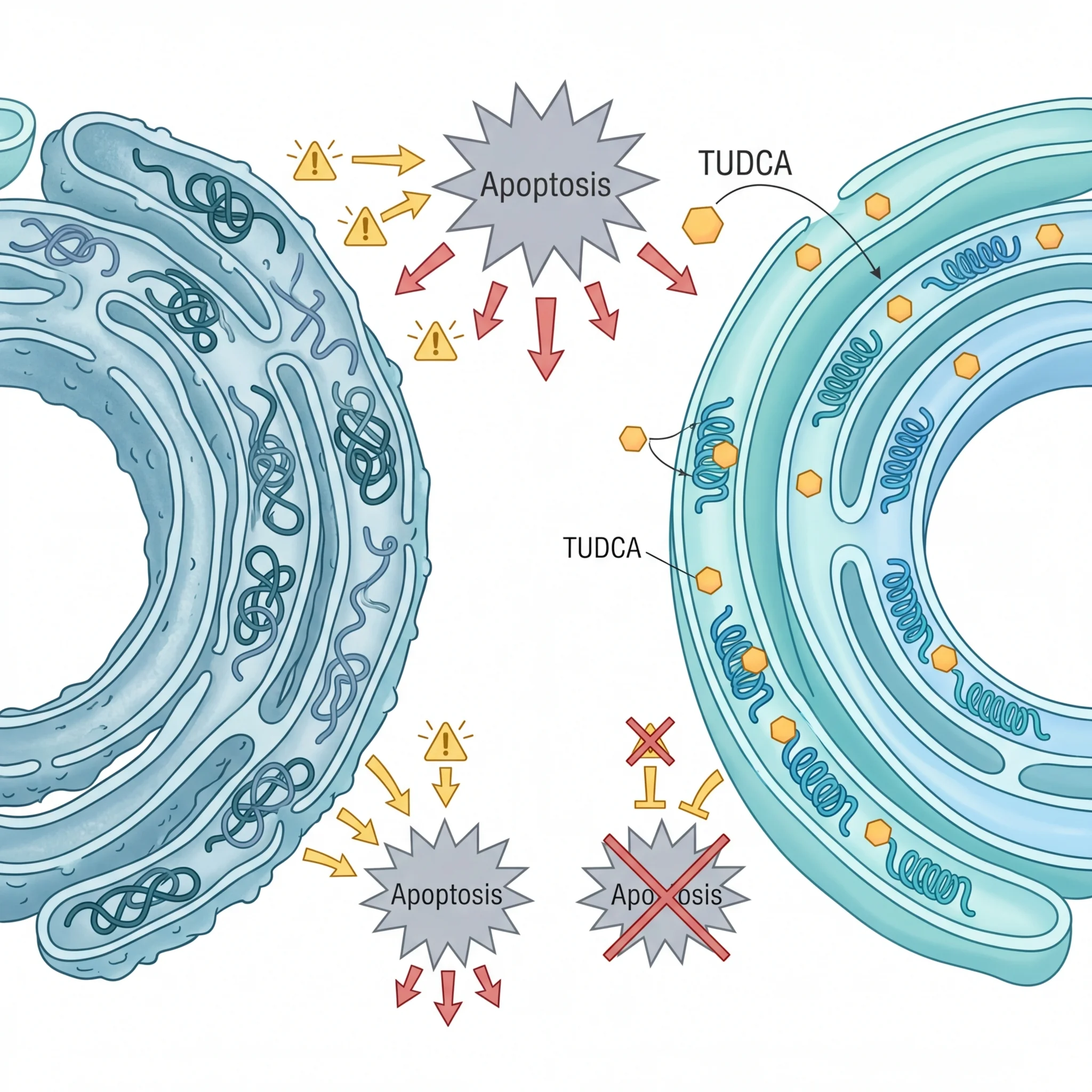
TUDCA acts as a molecular bodyguard inside liver cells
The liver is where TUDCA does its most established work. When bile flow slows or stops -- a condition called cholestasis -- toxic hydrophobic bile acids accumulate inside liver cells and start destroying them from within. Their detergent-like properties punch holes in cell membranes and trigger apoptosis. TUDCA counteracts this in two ways: it competes with toxic bile salts for binding sites on hepatocytes, and it shifts the overall bile acid composition toward less damaging, more hydrophilic forms.
Supplementing with TUDCA at doses of 500 to 1500 mg per day has been shown to increase the hydrophilic fraction of bile from under 3% to as high as 41.6% in patients with primary biliary cirrhosis. That shift alone dramatically reduces the damage bile acids inflict on liver tissue.
But TUDCA's liver benefits go beyond cholestasis. Randomized controlled trials have demonstrated that TUDCA treatment lowers serum markers of liver inflammation -- ALT, AST, and ALP -- in patients with liver cirrhosis, chronic hepatitis, and drug-induced cholestasis. These are not marginal reductions. TUDCA is used as a reference drug for cholestatic conditions, meaning other treatments are compared against it.
| Liver condition | Study type | TUDCA dose | Key finding |
|---|---|---|---|
| Liver cirrhosis | RCT | 750 mg/day | Significant reduction in ALT, AST, ALP |
| HCV-related hepatitis | RCT | 750 mg/day | Improved liver function markers |
| Cholestasis | RCT | 500-1500 mg/day | Bile composition shift to hydrophilic acids |
| Chronic liver disease | Clinical trial | 10-13 mg/day | Enhanced hepatocyte regeneration rate |
For people interested in liver support supplements, TUDCA occupies a different niche than milk thistle or NAC. Where those compounds primarily work through antioxidant mechanisms, TUDCA directly modifies bile chemistry and protects hepatocytes at the membrane level. Some people stack TUDCA with NAC for that reason, though no clinical trial has tested the combination specifically.
The ER stress connection links TUDCA to nearly every organ system
The endoplasmic reticulum (ER) is the cell's protein-folding factory. When that factory gets overwhelmed -- by inflammation, metabolic stress, toxin exposure, or just high metabolic demand -- proteins start misfolding. The cell launches an emergency response called the unfolded protein response (UPR). If the UPR cannot fix the problem, it triggers the cell to self-destruct.
TUDCA interrupts this cascade. It functions as a "chemical chaperone," stabilizing protein structures and helping the ER recover its folding capacity without activating the cell death program. A comprehensive review in Cells describes how TUDCA assists in the transfer of mutant proteins and improves protein folding through activation of the ATF6 pathway, one of the three main ER stress sensors.
This mechanism matters because ER stress is not a liver-specific problem. It contributes to insulin resistance, neurodegeneration, inflammatory bowel conditions, and cardiovascular disease. When you read claims that TUDCA helps with everything from diabetes to Parkinson's, the ER stress pathway is usually the common thread.
Gut barrier integrity
Your intestinal lining is a single cell layer thick in places. ER stress in those epithelial cells weakens the tight junctions between them, allowing bacteria and food particles to cross into your bloodstream -- the process behind leaky gut syndrome. TUDCA's ability to reduce ER stress in intestinal cells is one reason it appears in gut healing protocols, though human trials specifically measuring gut permeability with TUDCA supplementation are still limited.
The bile flow angle matters here too. Adequate bile secretion is necessary for fat digestion and for controlling bacterial populations in the small intestine. Poor bile flow is one factor in SIBO development. By supporting healthy bile production and flow, TUDCA indirectly supports the gut environment.
Brain and neuroprotection
TUDCA crosses the blood-brain barrier, which most supplements cannot do. Once in neural tissue, it has shown protective effects in animal models of Alzheimer's, Parkinson's, Huntington's, and ALS. The mechanisms involve reduced apoptosis, lower oxidative stress, and preserved mitochondrial function.
In Alzheimer's mouse models, TUDCA treatment decreased amyloid-beta deposits in the hippocampus and frontal cortex, interfered with amyloid precursor protein processing, and partially rescued synaptic function. In Parkinson's models, it prevented dopaminergic neuron death by modulating JNK signaling and reducing reactive oxygen species production.
There is an important caveat: all the neuroprotection data comes from animal studies. No human trial has demonstrated cognitive benefits from TUDCA supplementation. The Alzheimer's Drug Discovery Foundation rates TUDCA as "promising for neuroprotection" while noting that zero human evidence exists for dementia prevention or cognitive improvement. That gap between animal promise and human evidence is worth keeping in mind before buying TUDCA specifically for brain health.
Anti-inflammatory pathways
Beyond ER stress, TUDCA directly suppresses inflammation through at least two pathways. Research from Romero-Ramirez and colleagues showed that TUDCA inhibits NF-kB activation in both astrocytes and microglial cells under pro-inflammatory conditions, reducing nitric oxide production and microglial migration to injury sites. A follow-up study found that TUDCA also upregulates TGF-beta3 expression, pushing immune cells toward an anti-inflammatory phenotype.
If you have tried curcumin or omega-3s for chronic inflammation, TUDCA works differently. It operates upstream, at the level of cellular stress signaling, rather than blocking specific inflammatory mediators after they have been released.
Where TUDCA delivered -- and where it fell short
The human clinical evidence for TUDCA clusters around three areas: liver disease, metabolic health, and neurodegenerative disease. The strength of evidence is uneven.
Insulin sensitivity: the standout human trial
The most frequently cited clinical study randomized 20 obese men and women with insulin resistance to either TUDCA (1,750 mg/day) or placebo for four weeks. Hepatic and muscle insulin sensitivity increased by approximately 30% in the TUDCA group with no change in the placebo group. Insulin signaling markers (phosphorylated IRS and Akt) improved in muscle tissue.
One puzzling finding: despite TUDCA's reputation as an ER stress reliever, markers of ER stress in muscle and adipose tissue did not change. The insulin sensitizing effect happened through some other mechanism. The researchers concluded that "additional studies are needed to evaluate the target cells and mechanisms responsible."
That was published in 2010. Additional human metabolic studies remain limited.
Liver disease: consistent evidence
Multiple randomized controlled trials support TUDCA for liver conditions. A multicenter RCT showed TUDCA to be as safe and well-tolerated as UDCA for primary biliary cholangitis, with potentially better symptom relief. Its use in cholestasis is well-established enough that it serves as a reference treatment in clinical settings.
ALS: the disappointing phase 3
After a small 2016 trial suggested TUDCA might slow ALS progression, a large Phase 3 trial (TUDCA-ALS) enrolled 334 participants across seven European countries. The trial did not meet its primary endpoint. No significant reduction in disease progression was observed over 18 months, and secondary endpoints including survival showed no benefit. This was a well-designed, adequately powered trial, and its negative result carries weight.
| Research area | Evidence level | Key finding |
|---|---|---|
| Cholestasis/liver disease | Multiple RCTs | Consistently positive, reference-level treatment |
| Insulin sensitivity | Single RCT (n=20) | 30% improvement, needs replication |
| Cholesterol reduction | Small clinical trial (n=24) | Dose-dependent decrease in total and HDL cholesterol |
| Neuroprotection (Alzheimer's, Parkinson's) | Animal studies only | Promising in mice, zero human data |
| ALS | Phase 3 RCT (n=334) | Failed primary endpoint |
| Gallstone dissolution | Clinical use, limited trials | Effective only for small, uncalcified cholesterol stones |
Myth vs. reality
| Claim you will see online | What the evidence actually says |
|---|---|
| "TUDCA cures fatty liver" | It improves liver enzymes in diseased livers. No RCT specifically studied NAFLD. |
| "TUDCA prevents Alzheimer's" | Reduced amyloid plaques in mice. No human cognitive trials exist. |
| "TUDCA is a proven weight loss supplement" | One 4-week trial in obese subjects showed no change in body weight or fat mass. |
| "TUDCA is the same as bear bile" | Bear bile contains TUDCA among many other compounds. Synthetic TUDCA is chemically identical to the TUDCA fraction only. |
| "TUDCA protects your liver from alcohol" | Co-administration with alcohol reduced liver cell death in vitro. Pre-loading TUDCA before drinking made damage worse. |

Finding the right dose depends on what you are trying to do
Clinical studies have used TUDCA at wildly different doses depending on the condition being studied. There is no single "correct" dose, and supplement labels typically suggest amounts that may not match the research for your specific goal.
| Purpose | Dose used in research | Duration studied | Notes |
|---|---|---|---|
| General liver support | 250-500 mg/day | Ongoing | Common supplement dose; below most clinical trial doses |
| Cholestasis treatment | 500-1500 mg/day | 2-6 months | Higher doses shifted bile composition more effectively |
| Liver cirrhosis | 750 mg/day | Up to 6 months | Reduced ALT, AST, ALP in RCTs |
| Insulin sensitivity | 1,750 mg/day | 4 weeks | Only dose tested; 30% improvement in one RCT |
| Hepatocyte regeneration | 10-13 mg/day | 3 months | Very low dose; specific to chronic liver disease |
The optimal bile composition shift appears to occur at 15-20 mg per kilogram of body weight per day. For a 70 kg (154 lb) person, that is 1,050 to 1,400 mg daily. Most supplements sell 250 mg or 500 mg capsules, so hitting clinically studied doses means taking multiple capsules.
Timing matters less than consistency. TUDCA absorbs well with or without food, though taking it with meals aligns with natural bile acid secretion patterns. Some practitioners recommend splitting the dose -- half in the morning, half in the evening -- particularly at higher doses, since bile acid cycling follows a circadian rhythm.
When choosing a supplement, look for third-party testing (NSF, USP, or Informed Sport certification). TUDCA is a specific compound, not a plant extract with variable potency, so quality control issues are more about purity and accurate labeling than bioavailability enhancement. No studies have compared different supplement brands head-to-head.
TUDCA's safety profile is good -- with a few caveats worth knowing
Across clinical trials lasting up to one year, TUDCA has consistently been well-tolerated. A Cochrane meta-analysis of seven randomized controlled trials in liver transplant patients found that bile acids including TUDCA were safe, with diarrhea as the only reported adverse event, and even that occurred in only two of the four trials that tracked adverse events.
Other trials in liver cirrhosis (up to 6 months) and ALS (1 year) confirmed the same picture: TUDCA is well-tolerated with diarrhea being the only consistently reported side effect. In obese subjects taking 1,750 mg daily for four weeks, no adverse effects were noted at all.
The PSC warning
One study raises an important red flag. In the Lindor trial, patients with primary sclerosing cholangitis (PSC) received high-dose UDCA (28-30 mg/kg/day) for up to six years. Patients on UDCA were 2.3 times more likely to reach an adverse primary endpoint (cirrhosis, bile duct cancer, liver transplant, or death) than those on placebo. The trial was terminated early. While this used UDCA rather than TUDCA, and at doses well above typical supplement ranges, it is a reminder that bile acids are biologically active compounds and more is not automatically better.
Drug interactions and timing considerations
Formal drug interaction data for TUDCA as a supplement is limited. The Mayo Clinic's drug interaction list for taurursodiol (the prescription form combined with sodium phenylbutyrate in Relyvrio) includes bile acid sequestrants like cholestyramine and colestipol, which would reduce TUDCA absorption. Cyclosporine and certain kinase inhibitors are also flagged.
The alcohol timing issue deserves repeating: in human liver cell culture, TUDCA reduced alcohol-induced cell death when administered simultaneously with ethanol. But when given before alcohol exposure, it actually increased liver cell damage. The mechanisms behind this reversal are unclear, but until they are sorted out, taking TUDCA as a "pre-game" liver protector is not supported by the evidence.
Who should avoid TUDCA
No clinical trials have studied TUDCA in pregnant or breastfeeding women. People with moderate to severe kidney disease or liver disease should consult their physician, as TUDCA can affect bile composition in ways that may be unpredictable in advanced disease states. Anyone on prescription bile acid therapy or immunosuppressants should discuss TUDCA with their prescriber.
There is also no clinical data on TUDCA supplementation lasting longer than one year. If you plan to use it long-term, periodic liver function testing is a reasonable precaution -- not because problems have been documented, but because they simply have not been studied.
Frequently asked questions
Can I take TUDCA and UDCA together?
There is no published clinical data on combining TUDCA with prescription UDCA (Ursodiol). Since TUDCA converts to UDCA in the body, combining them could result in higher-than-studied bile acid concentrations. If you are on prescription UDCA for a liver condition, ask your gastroenterologist before adding TUDCA supplements.
How long does it take for TUDCA to work?
Bile composition changes are measurable within two months of daily supplementation at 750 mg. The insulin sensitivity trial showed effects within four weeks at 1,750 mg/day. Liver enzyme improvements in clinical trials typically appeared over two to six months. There is no evidence for immediate or single-dose effects.
Is TUDCA safe for people without liver problems?
The only trial in people without liver disease (obese but otherwise healthy subjects) used 1,750 mg/day for four weeks with no adverse effects. The Cochrane meta-analysis and other clinical data also show a clean safety profile. That said, TUDCA specifically modifies bile acid composition, so the benefit-to-risk calculation in someone with normal liver function and no metabolic issues is less clear than in someone with an identified condition.
Does TUDCA help with gut health?
TUDCA supports bile flow, which is necessary for fat digestion and maintaining healthy bacterial populations in the small intestine. It also reduces ER stress in intestinal epithelial cells, which theoretically supports gut barrier integrity. However, no human trial has measured intestinal permeability, microbiome composition, or digestive symptoms as primary outcomes of TUDCA supplementation. The gut health claims are mechanistically plausible but clinically unproven.
Should I take TUDCA with food or on an empty stomach?
TUDCA absorbs well regardless of food intake. Taking it with meals matches the natural timing of bile secretion and may reduce the chance of mild GI discomfort at higher doses. Splitting a large daily dose into two servings (morning and evening meals) is a common practice, though no study has compared split versus single dosing.
Related Articles
- Liver Support: Milk Thistle, NAC, and the Best Evidence-Based Detox Supplements -- a companion guide to the other major liver-support supplements and how they compare to bile acid therapies.
- Taurine: Heart, Brain, Longevity, and Why This Amino Acid Is Having a Moment -- TUDCA is a taurine conjugate, and understanding taurine's independent effects adds context to how TUDCA works.
- Leaky Gut Syndrome: Symptoms, Causes, and Evidence-Based Healing -- TUDCA's gut barrier support connects directly to the intestinal permeability discussion.
- Inflammation, Chronic Disease, and Anti-Inflammatory Living -- for a broader view of the inflammatory pathways TUDCA modulates.
- The Gut-Brain Axis: How Your Microbiome Affects Mood and Cognition -- TUDCA's production depends on gut bacteria, linking it to the microbiome conversation.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.












