Why Your Birthday Candles Might Be Lying to You
Two people born on the same day in the same hospital can arrive at age fifty in wildly different states. One runs half-marathons and sleeps soundly through the night. The other battles high blood pressure, stiff joints, and brain fog that makes reading feel like translating a foreign language. Same number of candles on the cake. Completely different bodies underneath.
That gap between calendar age and physical reality is what scientists call the difference between chronological age and biological age. Chronological age simply counts the years since birth. Biological age reflects something far more meaningful: the accumulation of cellular damage, declining organ function, and diminishing physiological reserves that determine how much vitality you actually have left.
Why does this matter? Research now indicates that genetic factors account for only 15 to 25 percent of the aging process. The remaining 75 to 85 percent comes from lifestyle, environmental exposures, and accumulated wear and tear on cells. That means most of what drives how quickly you age is, at least in theory, within your control.
The field of biological age testing has grown from a niche academic pursuit into a consumer market where anyone can mail in a blood or saliva sample and receive a number representing how old their cells appear to be. But the science behind these tests is more nuanced than a single number can convey, and knowing what that number actually means requires understanding the molecular processes that produce it.
Key takeaway: Your chronological age tells you how long you have been alive. Your biological age estimates how much wear your cells have accumulated. The two can diverge by a decade or more, and that gap predicts disease risk better than the calendar alone.
The Molecular Machinery Behind Biological Age Testing
Most biological age tests trace back to one molecular process: DNA methylation. This is the addition of small chemical tags, called methyl groups, to specific locations on your DNA. These tags do not change the genetic code itself. Instead, they act like dimmer switches, dialing gene activity up or down without altering the underlying instructions.
Your body contains roughly 28 million CpG dinucleotides where methylation can occur, and the pattern of which sites are methylated shifts in a remarkably consistent way as you age. Some locations gain methyl groups over the decades while others lose them. This is not random drift. The changes follow specific trajectories that can be tracked and measured.
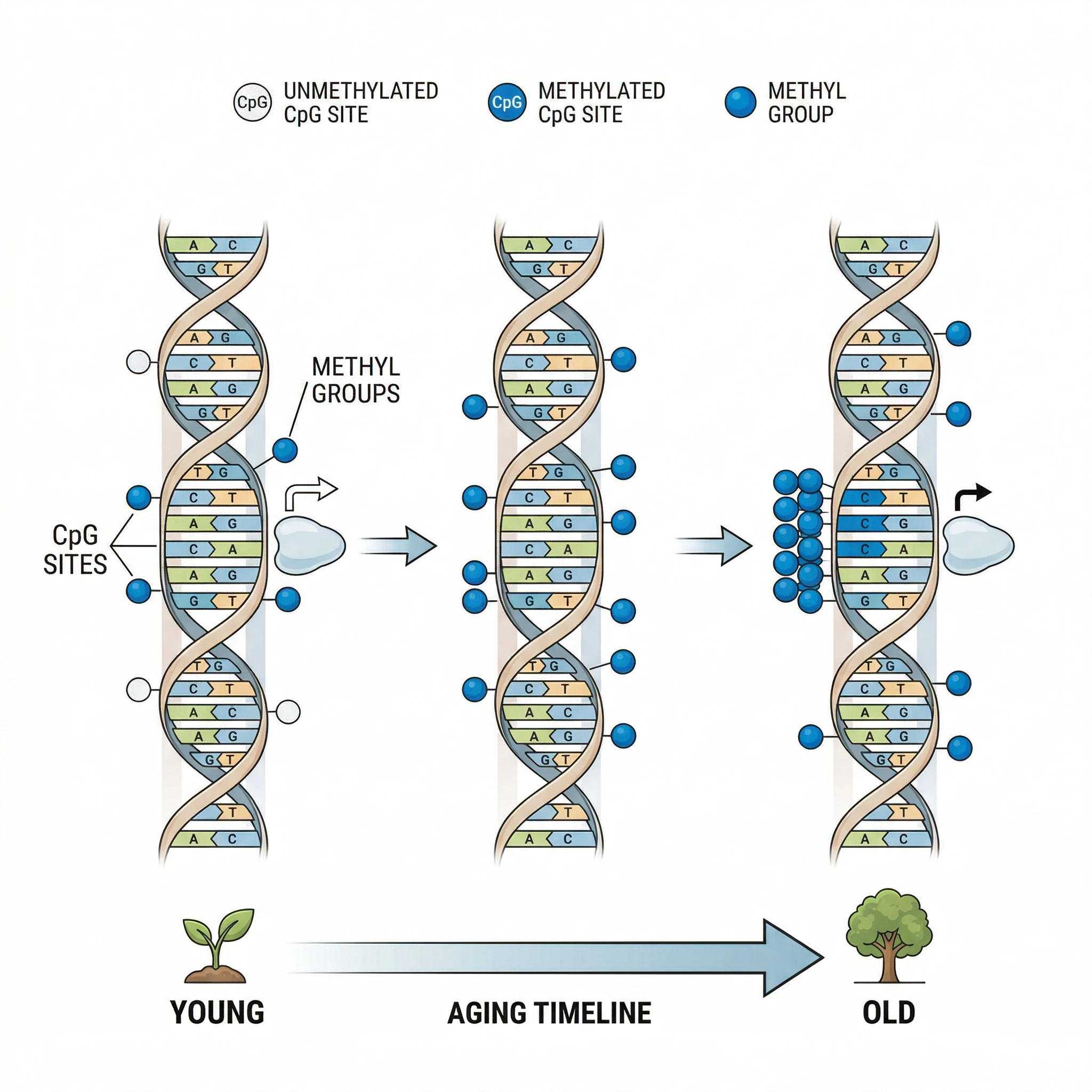
The reason this works so well as an aging biomarker is that DNA methylation reflects a dynamic balance between two enzymatic forces. DNA methyltransferases add methyl groups while TET (ten-eleven translocation) enzymes remove them. In young, healthy cells, these opposing forces maintain a stable equilibrium. As cells accumulate damage from oxidative stress, chronic inflammation, and environmental exposures, that balance shifts, and the methylation landscape drifts in predictable ways.
In practice, researchers can take a blood sample, measure methylation levels at a carefully selected set of CpG sites, and compute an age estimate that often correlates with chronological age at a coefficient above 0.9. When that computed age disagrees with the calendar, the direction of the mismatch carries meaning. Someone whose methylation pattern looks five years older than their birthday suggests may be aging at an accelerated rate, carrying elevated risk for the diseases that typically accompany that biological wear.
Beyond DNA methylation, several other molecular processes change with age and can serve as biomarkers. Telomeres, the protective caps on chromosome ends, shorten with each cell division and provide another window into cellular aging. Proteomic clocks, a newer approach, measure shifts in protein levels across the bloodstream. A study of over 44,000 individuals identified 14 metabolic markers that together predicted all-cause mortality risk. And the most recent proteomic aging clocks can forecast disease risk across diverse populations.
Every Type of Biological Age Test Available Today
Biological age testing goes well beyond a single test type. Each approach measures a different facet of the aging process, and no single test captures the whole picture.
| Test Category | What It Measures | Sample Required | Strengths | Limitations |
|---|---|---|---|---|
| Epigenetic clocks (DNA methylation) | Methyl group patterns at selected CpG sites | Blood or saliva | Highest correlation with aging outcomes; most validated | Expensive genome-wide arrays; tissue-specific calibration needed |
| Telomere length | Length of chromosome protective caps | Blood | Simple concept; widely available | High individual variation; weaker mortality prediction than epigenetic clocks |
| Proteomic clocks | Protein level patterns in blood | Blood | Captures organ-level aging; predicts disease in diverse populations | Newer, less long-term validation data |
| Metabolomic panels | Metabolite levels (lipids, amino acids, etc.) | Blood | Reflects metabolic health; 14-marker panel predicts mortality | Influenced by recent meals, acute illness |
| Clinical composite algorithms | Blood pressure, glucose, cholesterol, BMI, heart rate | Standard clinical data | Uses existing medical data; no special test needed | Less precise at molecular level; broad population averages |
| AI-based imaging | Retinal scans, EKG patterns, facial aging | Non-invasive imaging | Rapid; integrates structural data | Requires specialized equipment; emerging validation |
Among these, epigenetic clocks remain the most extensively studied and validated. However, the Max Planck Institute for Biology of Ageing notes that the most promising approach combines multiple methods, since each captures different aspects of the aging process. A single test reveals only a partial view.
Mayo Clinic researchers have demonstrated that AI-enabled electrocardiograms can detect the gap between chronological and biological heart age, and that gap predicted cardiovascular mortality. Northwestern University's Human Longevity Lab takes a similar integrative approach, combining DNA age tests with AI-computed cardiovascular and retinal health assessments to build a more comprehensive picture.

Three Generations of Epigenetic Clocks Compared
Epigenetic clocks have evolved through three distinct generations, each designed to solve a different problem. Understanding what each generation measures is essential for interpreting any test result you might receive.
| Generation | Key Clocks | Year | What It Predicts | CpG Sites Used |
|---|---|---|---|---|
| First | Horvath, Hannum | 2013 | Chronological age with high accuracy | 353 (Horvath), 71 (Hannum) |
| Second | PhenoAge, GrimAge | 2018-2019 | Mortality risk, disease onset, healthspan | 513 (PhenoAge), 1030 (GrimAge) |
| Third | DunedinPACE | 2022 | Rate of aging (pace, not status) | 173 |
First-generation clocks like Horvath's multi-tissue clock were trained to predict chronological age. Steve Horvath's 2013 clock analyzed methylation at 353 CpG sites across 51 different tissue types and achieved correlation coefficients above 0.9 and average prediction errors of less than five years. These clocks were groundbreaking but had a limitation: because they were calibrated to match the calendar, they measured a blend of aging signal and other age-correlated factors rather than biological aging specifically.
Second-generation clocks like PhenoAge and GrimAge were designed to predict health outcomes rather than just count years. GrimAge, named half-jokingly after the Grim Reaper, incorporates surrogate measures of smoking history, plasma protein levels, and other mortality-linked biomarkers. A study of over 3,500 participants found that these second and third generation clocks were capable of predicting functional limitations of daily living and impaired cognitive function, while first-generation clocks showed weaker associations with these outcomes.
Third-generation clocks represent a conceptual shift. The DunedinPACE test, developed from the Dunedin Study in New Zealand, does not estimate how old you are biologically. Instead, it measures how fast you are aging right now. Built by tracking within-individual decline across 19 indicators of organ-system integrity over two decades, DunedinPACE quantifies the pace at which cardiovascular, metabolic, renal, hepatic, immune, dental, and pulmonary systems are deteriorating. The measurement is scaled so that a score of 1.0 means you are aging at the average rate of one biological year per calendar year. In the study cohort, the slowest ager accumulated just 0.40 biological years per calendar year while the fastest ager racked up 2.44.
Why this matters: Knowing your pace of aging is arguably more useful than knowing your cumulative biological age, because pace reflects your current trajectory. A person with an unfavorable biological age who has recently slowed their pace may be on a better path than someone whose biological age looks fine but whose pace is quietly accelerating.
Reading Your Results Without Losing Your Mind
If you order a direct-to-consumer biological age test, you will receive a number. That number will either match your chronological age, fall below it (suggesting slower aging), or exceed it (suggesting accelerated aging). The impulse to panic or celebrate is natural. Neither reaction is fully warranted.
| Myth | Reality |
|---|---|
| Biological age tells you how long you will live | It estimates a statistical tendency, not a personal expiration date. Many other factors influence longevity. |
| All biological age tests measure the same thing | Different clocks use different CpG sites, different tissues, and different calibration targets. Results vary across tests. |
| A bad result means permanent damage | Epigenetic methylation patterns are dynamic. Lifestyle changes can shift them in favorable directions within weeks to months. |
| A good result means you can relax about health habits | A favorable score reflects current habits. Abandoning them will shift the methylation pattern over time. |
| Consumer tests are clinically validated medical diagnostics | Most have not undergone the rigorous independent evaluation required for clinical diagnostic tools. |
Luigi Ferrucci, the scientific director at the National Institute on Aging, summarizes the current state directly: taking a biological age test should be based on curiosity rather than clinical need. Matt Kaeberlein, founding director of the Healthy Aging and Longevity Research Institute at the University of Washington, puts it even more bluntly: "I don't think they're actionable right now."
That said, for some people, seeing a concrete number serves as a catalyst. If discovering that your body appears five years older than your birth certificate motivates you to start exercising, fix your sleep, or address chronic stress, the test delivered value regardless of its technical limitations. The Cleveland Clinic's Dr. Saadia Hussain acknowledges this: the question to ask yourself before testing is whether the result would actually change how you live your life.
One important caveat: researchers warn against ordering from companies that bundle testing with their own supplement products. As Kaeberlein notes, the idea that "you take the test and then they tell you to buy their supplement" is problematic. No supplements are FDA-approved for anti-aging, and the conflict of interest is obvious.
Another limitation worth understanding: epigenetic clocks trained on blood samples lose precision when applied to other tissues. A blood-based test tells you about your blood cells' epigenetic age. It may not accurately reflect what is happening in your brain, liver, or muscles. Tissue-specific clocks exist for research settings but are not yet available to consumers.

What Actually Slows the Clock, According to Clinical Trials
The best case for biological age testing comes down to one fact: the methylation patterns it measures are not fixed. Unlike your DNA sequence, which stays the same from birth to death, your methylation landscape shifts in response to how you live. And the evidence that specific interventions can reverse epigenetic aging is no longer theoretical.
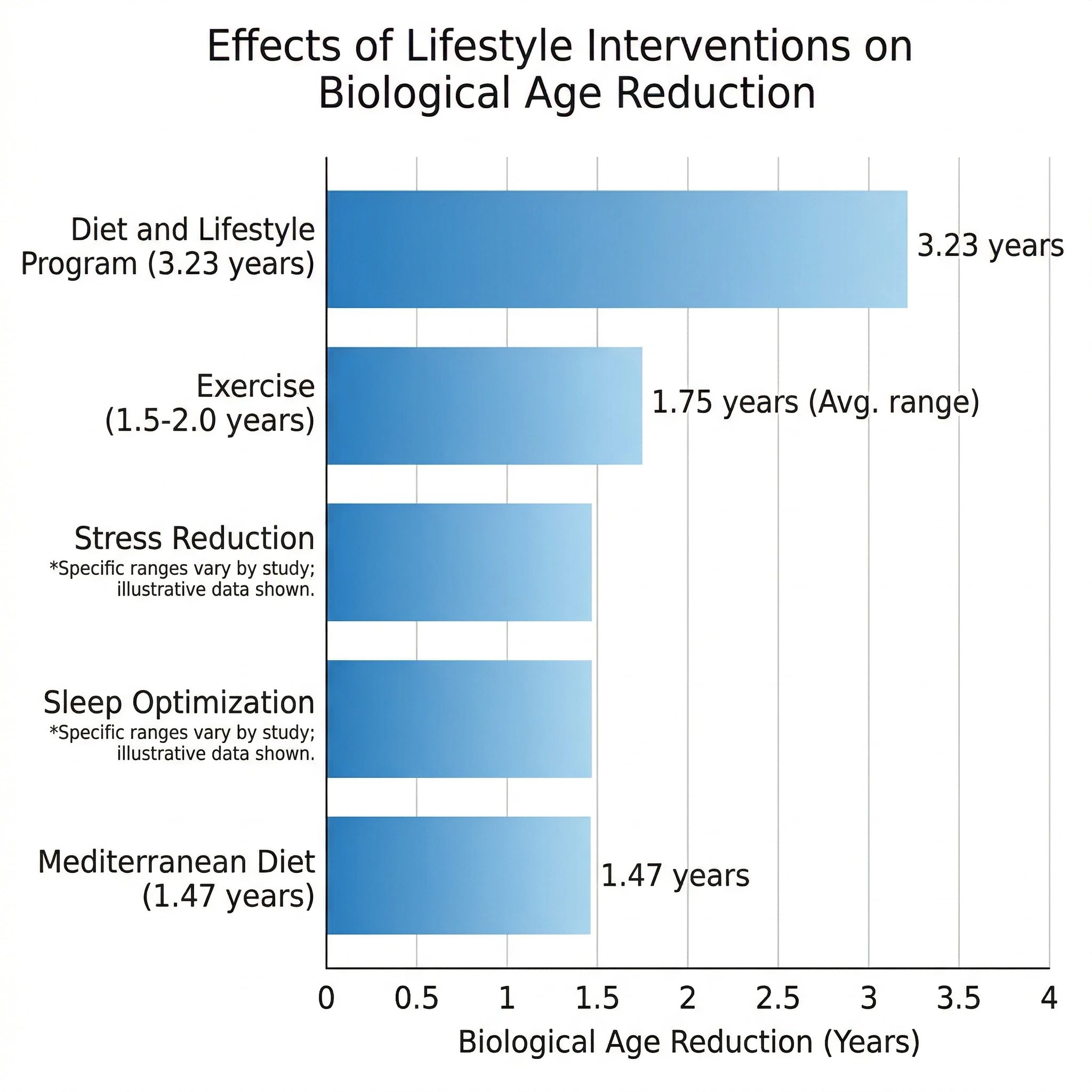
The most concrete clinical evidence comes from a randomized controlled trial led by Dr. Kara Fitzgerald, published in the journal Aging. Forty-three healthy men between 50 and 72 followed an eight-week program combining a plant-centered diet rich in methylation-supporting nutrients, daily exercise, stress-reduction breathing exercises, and optimized sleep. The treatment group's Horvath epigenetic age decreased by an average of 3.23 years compared to controls (p=0.018). Their triglycerides dropped 25 percent and serum folate increased 15 percent. This was the first RCT to demonstrate potential reversal of epigenetic aging through lifestyle alone.
The specific protocol from that trial is worth examining for anyone interested in an evidence-based approach:
- Diet: Plant-centered with high intake of folate, betaine, vitamin C, and polyphenols (curcumin, EGCG, quercetin). Limited carbohydrates, mild intermittent fasting, plus a daily probiotic (Lactobacillus plantarum 299v)
- Exercise: Minimum 30 minutes daily, at least 5 days per week, at 60-80 percent of maximum perceived exertion
- Stress management: Twice-daily breathing exercises designed to elicit the relaxation response
- Sleep: Minimum seven hours nightly
The stress component deserves special attention. The researchers noted that nearly a quarter of the CpG sites in the Horvath clock (85 out of 353) sit within glucocorticoid response elements, regions of DNA that respond directly to stress hormones. Cumulative lifetime stress has been independently associated with accelerated epigenetic aging, and conditions like PTSD show measurable acceleration of the methylation clock.
Exercise alone carries substantial evidence. A 2025 review in the journal Aging synthesized findings across multiple studies and reported that high-fitness individuals showed 1.5 to 2.0 years lower epigenetic age acceleration compared to those with medium-to-low fitness. Cardiorespiratory fitness, measured by VO2 peak, correlated more strongly with lower epigenetic age than grip strength or leg power, suggesting that aerobic capacity matters most. The benefits extended across multiple organs, with animal studies showing lower epigenetic age in skeletal muscle, adipose tissue, cardiac muscle, and liver in the high-fitness group.
Diet-specific evidence also shows promise. The NU-AGE trial found that one year of a Mediterranean diet reduced epigenetic age in elderly participants, with the strongest effect (1.47-year reduction) observed in Polish women, whose baseline diet was furthest from Mediterranean patterns. This suggests that people with the poorest dietary habits stand to gain the most from nutritional improvements.
The mechanisms connecting autophagy, fasting, and exercise to slower aging appear to converge on the same epigenetic pathways that these clocks measure. When cells efficiently clear damaged components and maintain their repair machinery, the methylation landscape stays closer to its youthful configuration.
Frequently Asked Questions
How much does a biological age test cost, and do I need a doctor to order one?
Direct-to-consumer epigenetic age tests typically cost between $200 and $500. Most do not require a physician's order. You collect a blood or saliva sample at home and mail it to the testing company. However, researchers recommend interpreting results with professional guidance, since the tests are not yet classified as clinical diagnostics and results can be confusing without context.
Can biological age testing predict specific diseases?
Biological age tests do not diagnose specific diseases. What they can do is estimate your overall pace of aging, which correlates with risk for multiple age-related conditions including cardiovascular disease, cognitive decline, and multimorbidity. A study of over 3,500 Americans found that second and third generation epigenetic clocks were the strongest predictors of multimorbidity specifically, outperforming even socioeconomic and behavioral factors for that particular outcome.
How often should I retest to track changes?
Most longevity researchers who use these tests suggest retesting no more frequently than every six to twelve months. Meaningful changes in methylation patterns take time to accumulate, and shorter intervals introduce more noise than signal. If you are using the test to track the impact of lifestyle changes, a twelve-month gap between tests gives interventions enough time to produce measurable shifts.
Are some people genetically locked into fast aging regardless of lifestyle?
Genetic conditions like Down syndrome do accelerate epigenetic aging, with affected individuals showing an average epigenetic age discrepancy of about 11 years. However, for the general population, genetics account for only 15 to 25 percent of the aging process. Centenarians, who represent the opposite extreme, show epigenetic ages roughly 6.5 years younger than their chronological age, and their offspring also show slower epigenetic aging. Most people fall between these extremes and have substantial room to influence their trajectory through lifestyle.
Do supplements like NAD+ and NMN or spermidine actually slow biological aging?
Some longevity compounds show promising results in laboratory and animal studies, but clinical evidence in humans measured by validated epigenetic clocks remains limited. No supplements have been FDA-approved for anti-aging use. The strongest evidence for slowing epigenetic aging in humans currently comes from comprehensive lifestyle interventions combining diet, exercise, sleep optimization, and stress management, not from any single supplement.
Related Articles
- Telomere Length and Cellular Aging — How the protective caps on your chromosomes connect to longevity and what you can do to preserve them.
- Autophagy, Fasting, and Exercise — The cellular cleanup process that keeps your body's repair machinery running and its connection to slower aging.
- NAD+ and NMN Supplements for Cellular Repair — What the research says about these popular longevity supplements and their effects on aging pathways.
- Spermidine for Autophagy and Anti-Aging — A naturally occurring compound found in wheat germ and other foods that activates the same cellular renewal pathways targeted by fasting.
- Chronic Inflammation and Anti-Inflammatory Living — How persistent low-grade inflammation accelerates biological aging and evidence-based strategies for reducing it.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.