Why the Safety Answer Is Messier Than the Hype Suggests
The honest answer is not "BPC-157 is safe" or "BPC-157 is dangerous." It is more uncomfortable: the strongest claims are running ahead of the human evidence. BPC-157 is a synthetic 15-amino-acid peptide connected to research on a protective compound found in gastric juice, but that origin story does not make a self-injected online vial medically proven in people.
Think of it like seeing a building design that performs beautifully in a wind tunnel. Useful? Yes. Enough to let families move in before inspectors test the finished structure? No. Flynn McGuire, MD, and colleagues wrote in a 2025 narrative review that BPC-157 human data is "exceedingly sparse," even though online communities treat the peptide as a near-routine recovery tool for injuries.
The regulatory picture adds another layer. The Department of Defense's Operation Supplement Safety describes BPC-157 as an unapproved drug found in some health and wellness products, and it is not FDA-approved for any medical use in humans. Australian health educators make the same practical point: the drug is discussed online as if it were a wellness supplement, but it sits outside ordinary approved-medicine pathways in major markets.
Bottom line: BPC-157 may have interesting biology, but first-time users should treat it as an experimental compound with thin human safety data, not as a proven recovery shortcut.

What Human Data Can and Cannot Tell Us
The human evidence is not empty. It is just too small to carry the confidence people put on it. ClinicalTrials.gov lists NCT02637284, a safety and pharmacokinetics trial planned for 42 healthy volunteers, but the results submission was canceled before quality-control review on the public record. That does not prove harm. It does mean readers cannot inspect the results of what should have been one of the cleanest human safety datasets.
The published IV safety paper is much narrower. Edwin Lee, MD, and Kailynd Burgess reported a pilot study in 2 healthy adults who received 10 mg and then 20 mg of IV BPC-157, with no reported side effects and no clinically relevant changes in heart, liver, kidney, thyroid, or glucose markers over the brief observation window. That is useful, but it is not a green light for everyone. The study excluded people with chronic kidney disease, diabetes, hypothyroidism, liver failure, pregnancy, or heart disease from participation.
Other small reports sound more exciting than they should. The same IV paper summarizes a knee-pain report in which 14 of 16 patients said pain improved after one intra-articular BPC-157 injection, and an interstitial-cystitis report in which 12 women received 10 mg into the bladder wall, with 10 reporting complete improvement and 2 reporting 80% improvement without reported side effects. Those outcomes are interesting enough to justify better trials. They are not enough to predict what happens to a stranger ordering powder online.
| Human evidence | What it suggests | Why it is not enough |
|---|---|---|
| 42-person registered trial NCT02637284 | Designed to test safety and pharmacokinetics | Results submission was canceled before public review |
| 2-person IV pilot Lee and Burgess | No short-term side effects reported after 10 mg and 20 mg IV doses | Tiny sample, short timeframe, selected healthier adults |
| 16-patient knee report summarized by Lee and Burgess | 14 of 16 reported pain relief | Not a large blinded placebo-controlled trial |
| 12-woman bladder-pain report summarized by Lee and Burgess | 10 complete responses and 2 partial responses were reported | Small, condition-specific, not enough for broad safety claims |
The practical read is simple: small no-harm reports are signals, not proof. If you would not accept a 2-person trial as proof that a blood pressure medication is safe, do not accept it as proof that an unapproved peptide is safe for broad use.
Known and Plausible Side Effects to Watch For
Because the human dataset is small, the published side-effect list is short. That sounds comforting until you remember how rare problems are found. A rare side effect will not reliably show up in a tiny pilot, bladder report, or knee report by design.
The basic injection risks are not unique to BPC-157. Any injected substance can cause local pain, redness, swelling, infection, or allergic reaction, especially when the product source or technique is poor as Healthy Male notes. Think of injection as a locked side door into the body. Used in a clinic, that door is opened with sterile procedure. Used casually at home with a gray-market vial, it is still a door.
The IV pilot specifically asked participants about symptoms including nausea, diarrhea, vomiting, swelling, dry mouth, fatigue, headache, sweating, rash, muscle spasm, hot flashes, and shortness of breath during monitoring. None were reported in those 2 adults during the study. That is reassuring only within the narrow boundaries of that study: brief exposure, screened participants, known product source, and close observation.
Short half-life is another detail people overread. The Lee/Burgess paper's pharmacokinetic summary says BPC-157 has a bloodstream half-life of less than 30 minutes in cited animal work. That may matter for dosing and trial design, but it does not automatically mean the downstream biological signals vanish in half an hour. A match can burn out quickly and still start a campfire. Safety depends on the repair, immune, vascular, and inflammatory processes the peptide nudges while it is present.

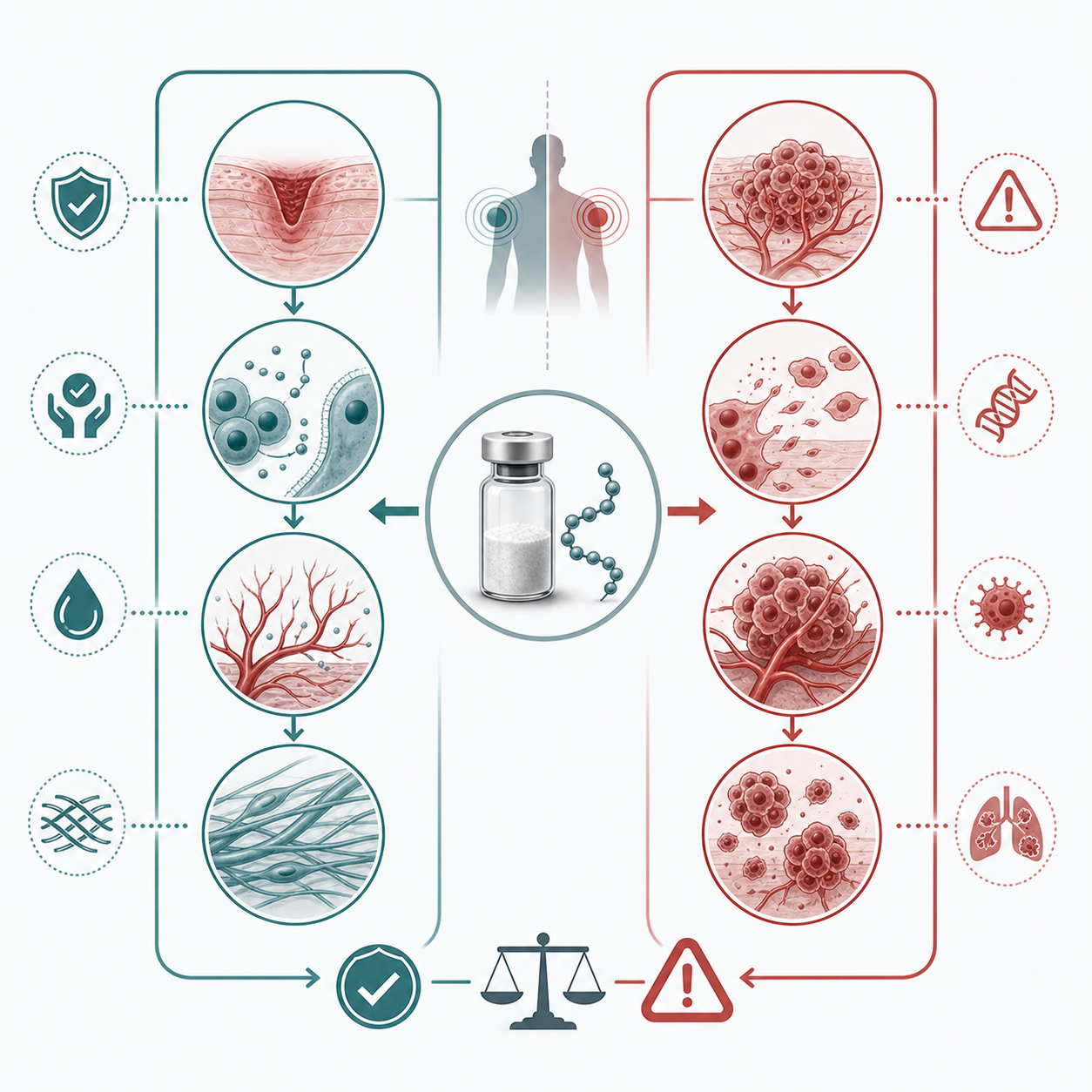
The Cancer, Angiogenesis, and Healing-Signal Question
This is the section most peptide marketing skips, because the mechanism cuts both ways. BPC-157 is interesting partly because it appears to support angiogenesis, the growth of new blood vessels, through pathways involving VEGFR2 and nitric oxide in preclinical work. For wound repair, that can sound good. New tissue needs a blood supply the way a construction site needs roads.
But biology does not label a road "for healing only." Tumors also use angiogenesis and migration pathways when they grow or spread. Michalina Jozwiak and colleagues wrote in a 2025 Pharmaceuticals review that BPC-157 may not be the right choice in situations where hidden cancer cells could be present because of those overlapping signals. Victor Prisk, MD, makes the same cautious argument from an orthopedic perspective: absence of proof that BPC-157 causes cancer is not the same thing as proof that long-term use is harmless for cancer risk.
There is a counterargument. Predrag Sikiric and colleagues argue that BPC-157 modulates angiogenesis rather than simply pushing it upward, and they cite animal models where the compound appears protective rather than tumor-promoting in their interpretation. That disagreement matters. It tells you this is not a settled yes-or-no risk; it is a plausible concern sitting in a thin human evidence base.
| Safety question | Why people are optimistic | Why caution remains |
|---|---|---|
| Angiogenesis | New blood vessels can help injured tissue rebuild in models | The same machinery can matter in tumor growth and invasion |
| Inflammation | Animal work suggests anti-inflammatory effects across injury models | Long-term immune effects in humans are not well characterized |
| Healing speed | Preclinical tendon, ligament, muscle, and gut data are broad in the Sikiric literature | Broad animal effects have not translated into broad approved human use |
Practically, this is where a first-time user should slow down most. If you have active cancer, a recent cancer history, unexplained masses, unusual bleeding, or symptoms you have not had medically evaluated, using a pro-healing experimental peptide is a poor first move. Diagnosis comes before tinkering.
Product Quality and Dosing Risks May Be the Bigger Problem
For many first-time users, the largest immediate risk may not be the peptide molecule. It may be the vial. STAT's Undark investigation describes how unapproved peptides are often sold as "research chemicals," a label that can make human use look like a loophole instead of a medical decision in the gray market.
The difference between a clinic-sourced, tested product and an online powder is not cosmetic. In the IV pilot, the authors used BPC-157 from a 503A compounding pharmacy, described a certificate of analysis, and said the product had been independently verified to be free of endotoxins before infusion. That kind of sourcing does not make the treatment approved, but it removes some avoidable hazards.
This is why clinic anecdotes and online anecdotes are not interchangeable. If a monitored clinic case uses a tested vial, excludes high-risk patients, records baseline labs, and checks symptoms afterward, the risk environment is completely different from a first-time user mixing powder at home. Same peptide name, different experiment.
Unregulated peptide quality can be ugly. A safety discussion from New Regeneration Orthopedics cites a Drug Testing and Analysis paper reporting that 30% of online peptides had incorrect amino-acid sequences and 65% exceeded endotoxin safety thresholds in tested products. Even if those numbers do not apply to every seller, they show why "my friend used this brand" is not a safety protocol.
| Risk source | What can go wrong | What a careful user would verify |
|---|---|---|
| Identity | The vial may not contain the peptide or sequence claimed on the label | Independent mass spectrometry or equivalent identity testing |
| Sterility | Injection can introduce contamination into tissue | Sterile compounding process and clear lot-level documentation |
| Endotoxin | Bacterial fragments can trigger inflammatory reactions if levels are high | Lot-specific endotoxin testing, not a generic purity badge |
| Dose | No FDA label defines dose, route, duration, or stop rules | Clinician-supervised plan and documented monitoring |
A useful rule: if you cannot explain where the peptide came from, what test proves its identity, what test proves sterility, what dose you are taking, and when you will stop, you are not doing a health experiment. You are guessing with a needle.

Who Should Avoid BPC-157 or Get Medical Guidance First
Some groups should not treat BPC-157 as a casual trial. The IV pilot excluded people with chronic kidney disease, diabetes, hypothyroidism, liver failure, pregnancy, or heart disease from its safety study. If a study excludes your situation, do not use that study as reassurance for your situation.
People with active cancer, recent cancer treatment, unexplained lumps, unexplained weight loss, or abnormal screening results deserve special caution because the angiogenesis question is unresolved in human safety terms. The same goes for anyone on immunosuppressants, anticoagulants, chemotherapy, fertility treatment, or complex heart, kidney, liver, or endocrine medication. The review literature does not provide a reliable interaction map for those combinations in humans.
Competitive athletes have a separate problem. OPSS states that BPC-157 is prohibited in sport and is an unapproved drug found in wellness products under anti-doping concern. Even a "research" label will not help if the substance creates a testing or eligibility problem.
- Do not start without medical guidance if you have active cancer, recent cancer, pregnancy, kidney disease, liver disease, heart disease, diabetes, thyroid disease, autoimmune disease, or unexplained symptoms.
- Do not self-inject a product without lot-specific identity, sterility, and endotoxin documentation.
- Do not combine BPC-157 with prescription drugs, hormones, or other peptides without a clinician reviewing interactions and monitoring.
- Do not use it for sport if anti-doping rules apply to you.

A Practical Risk Checklist for First-Time Users
If you are still considering BPC-157, make the decision boring and documented. A risky supplement decision usually feels urgent: start now, heal faster, do not overthink it. A safer medical decision creates friction on purpose.
- Define the actual diagnosis. Tendon pain, gut symptoms, or joint pain should be evaluated before an experimental compound enters the picture.
- Check the evidence for your exact use case. A bladder injection report does not prove safety for shoulder injections, oral capsules, or long-term cycling.
- Screen personal red flags. Cancer history, pregnancy, kidney disease, liver disease, heart disease, diabetes, thyroid disease, immune disorders, and complex medications deserve clinician review.
- Demand lot-level testing. Ask for identity, sterility, endotoxin, concentration, and beyond-use-date documentation, not a seller screenshot.
- Write stop rules before starting. Stop for rash, breathing symptoms, swelling, fever, worsening pain, unusual bleeding, severe headache, chest symptoms, neurologic symptoms, or any sign of infection.
- Do not stack unknowns. Starting BPC-157 with TB-500, growth-hormone secretagogues, hormones, or multiple supplements makes it harder to know what caused a problem.
The best answer for many first-time users is to wait. That is not fear; it is sequencing. Exhaust the boring proven options, understand your diagnosis, and only then discuss experimental therapies with someone who is accountable for monitoring you.
If you do discuss it with a clinician, bring specific questions instead of asking, "Is this safe?" Ask what diagnosis you are treating, what evidence exists for that exact condition, what baseline labs or screening matter, what product source would be used, what side effects would stop treatment, and what outcome would count as failure. A serious answer should sound specific. A sales answer usually sounds easy.

Frequently Asked Questions
Is BPC-157 FDA-approved?
No. OPSS describes BPC-157 as an unapproved drug found in some health and wellness products, and the FDA has not approved it for human medical use as a treatment.
Does BPC-157 have human safety studies?
Yes, but they are small and incomplete. The most direct IV pilot reported no side effects in 2 healthy adults after 10 mg and 20 mg infusions, while a planned 42-person safety and pharmacokinetics trial has a canceled results submission on ClinicalTrials.gov for the pilot and for the trial record.
Can BPC-157 cause cancer?
No human study proves that BPC-157 causes cancer. The concern is theoretical but serious enough to respect: some repair pathways linked to BPC-157, including angiogenesis-related signaling, overlap with pathways tumors can exploit in biology.
Are oral capsules safer than injections?
Oral use avoids needle-related infection and injection-site problems, but it does not solve the larger issues: no FDA-approved label, uncertain human dosing, variable product quality, and limited long-term safety data in people.
What is the biggest first-time-user mistake?
The biggest mistake is treating source quality as an afterthought. A product with the wrong sequence, contamination, or uncertain concentration changes the risk calculation before the peptide's biology even enters the discussion according to product-safety reporting.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.