Your Gut Produces a Hormone That Lasts 11 Minutes. These Drugs Hijack It for a Week.
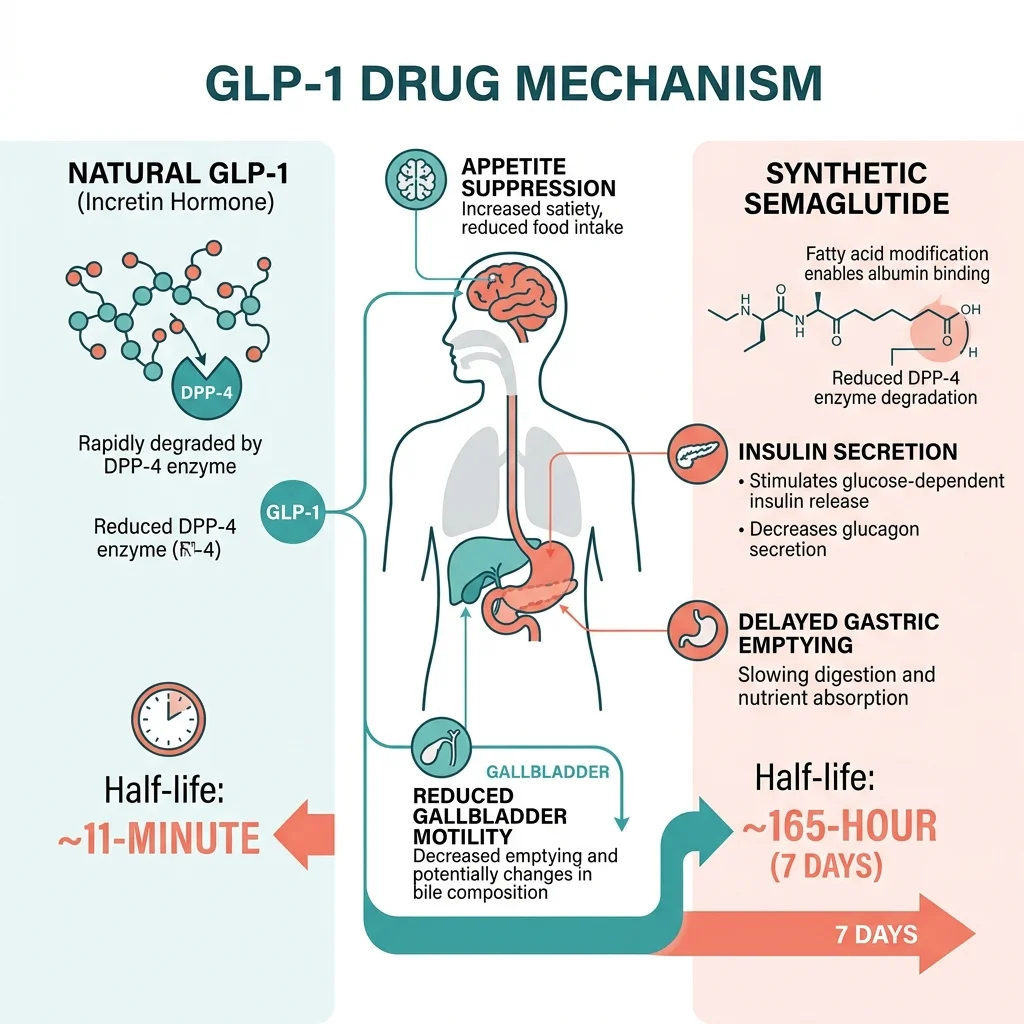
Every time you eat, specialized L-cells in your lower intestine release a hormone called glucagon-like peptide-1 (GLP-1). This hormone does three things at once: it tells your pancreas to release insulin, signals your brain that you're full, and slows the speed at which food leaves your stomach. Under normal conditions, GLP-1 survives for a maximum of 11 minutes before enzymes chew it apart. That brevity is by design. Your body wants the satiety signal to match the meal, not linger for days.
Drugs like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro, Zepbound) are synthetic versions of this hormone, but engineered to resist that enzymatic breakdown. Semaglutide incorporates two amino acid substitutions and a fatty acid chain that latches onto albumin in the blood, stretching its half-life to roughly 165 hours, about one week. Think of it like replacing a sticky note on your fridge ("I'm full") with a billboard bolted to your kitchen wall.
That persistence is what makes GLP-1 receptor agonists effective. In healthy people, the natural incretin effect, the boost in insulin secretion triggered by eating, accounts for 50-70% of the postprandial insulin response. In people with type 2 diabetes, that contribution shrinks to 20-30%. GLP-1 drugs restore and amplify that diminished signal.
The weight loss follows from three overlapping mechanisms: reduced appetite via brain signaling, delayed gastric emptying, and improved insulin sensitivity. WHO-commissioned Cochrane reviews analyzing data from thousands of participants found tirzepatide produced about 16% body weight reduction after 12-18 months, semaglutide about 11%, and liraglutide 4-5%.
But a hormone that touches receptors in your brain, stomach, pancreas, kidneys, gallbladder, and eyes doesn't restrict its effects to the ones you want. The same mechanism that slows gastric emptying to reduce appetite can, in some patients, slow it to the point of paralysis. The same receptor activation that stimulates insulin secretion also bathes the pancreas in continuous stimulation it wasn't designed to handle for years at a time. Understanding the side effects requires understanding this biology, because these aren't random malfunctions. They are the predictable consequences of turning a brief hormonal whisper into a perpetual shout.
Pancreatitis and GLP-1 Drugs: Two Decades of Contradictory Data
The pancreatitis question has haunted GLP-1 drugs since the early days of exenatide. Singh et al. published data in 2013 showing that GLP-1-based therapies were associated with increased odds of hospitalization for acute pancreatitis. Four years later, Storgaard et al. conducted a meta-analysis and found no evidence of increased risk. The two findings aren't as contradictory as they appear, and the resolution matters for anyone taking these drugs.
A 2025 propensity-matched study by Dr. Mark Ayoub and colleagues at Charleston Area Medical Center examined 969,240 patients with type 2 diabetes, matching GLP-1 users against non-users on demographics, comorbidities, and medications known to cause pancreatitis. The GLP-1 group did better, not worse: a lower lifetime pancreatitis risk of 0.3% versus 0.4% in non-users (p<0.001). At every time point measured, from 6 months to 5 years, the GLP-1 group showed equal or lower rates.
That same year, Dr. Joyce Hanyue Gu and Dr. Mark Samarneh published a different kind of analysis, one focused not on whether the drugs cause pancreatitis, but whether the risk increases with cumulative exposure. Using FDA Adverse Event Reporting System (FAERS) data, they found a clear dose-dependent relationship. At a cumulative dose threshold of 200mg, the odds ratio for pancreatitis was 1.66 (95% CI 1.22-2.26). At 5,300mg cumulative, it jumped to an odds ratio of 3.11 (95% CI 1.33-7.24).
Key takeaway: The average GLP-1 user doesn't face elevated pancreatitis risk compared to other diabetes patients. But patients who accumulate large cumulative doses over extended treatment, precisely the scenario of long-term weight management, may face meaningfully higher risk.
Large cardiovascular outcomes trials reinforce the population-level safety. LEADER, SUSTAIN-6, and EXSCEL found no significant difference in confirmed acute pancreatitis versus placebo, and REWIND reported less than 0.1% incidence per year. But these trials typically lasted 2-5 years. The dose-dependence data suggests the risk profile may shift for patients on decade-long treatment regimens, the kind increasingly common as these drugs expand from diabetes management to lifelong weight maintenance.
For patients, this means pancreatitis risk isn't a binary yes-or-no question. It depends on duration, dose accumulation, and individual factors like gallstone history and alcohol use. And that nuance rarely survives the 15-minute prescriber visit.
When "Slowed Digestion" Becomes Stomach Paralysis
Delayed gastric emptying is supposed to be a feature of GLP-1 drugs, not a bug. By slowing the movement of food from stomach to intestine, these medications reduce post-meal glucose spikes and promote satiety. GLP-1 receptors on vagal afferent nerves transmit inhibitory signals to the brainstem, which then modulates vagal motor output back to the stomach, reducing motility. Under normal therapeutic conditions, this creates a gentle brake on digestion.
In some patients, that brake locks. Gastroparesis, delayed stomach emptying without mechanical obstruction, occurs in fewer than 2% of GLP-1 users according to Dr. Pranav Periyalwar, a gastroenterologist at National Jewish Health who has watched cases climb alongside prescription rates. Symptoms include severe nausea, bloating, abdominal pain, and the unsettling experience of retained food sitting in the stomach for far longer than it should.
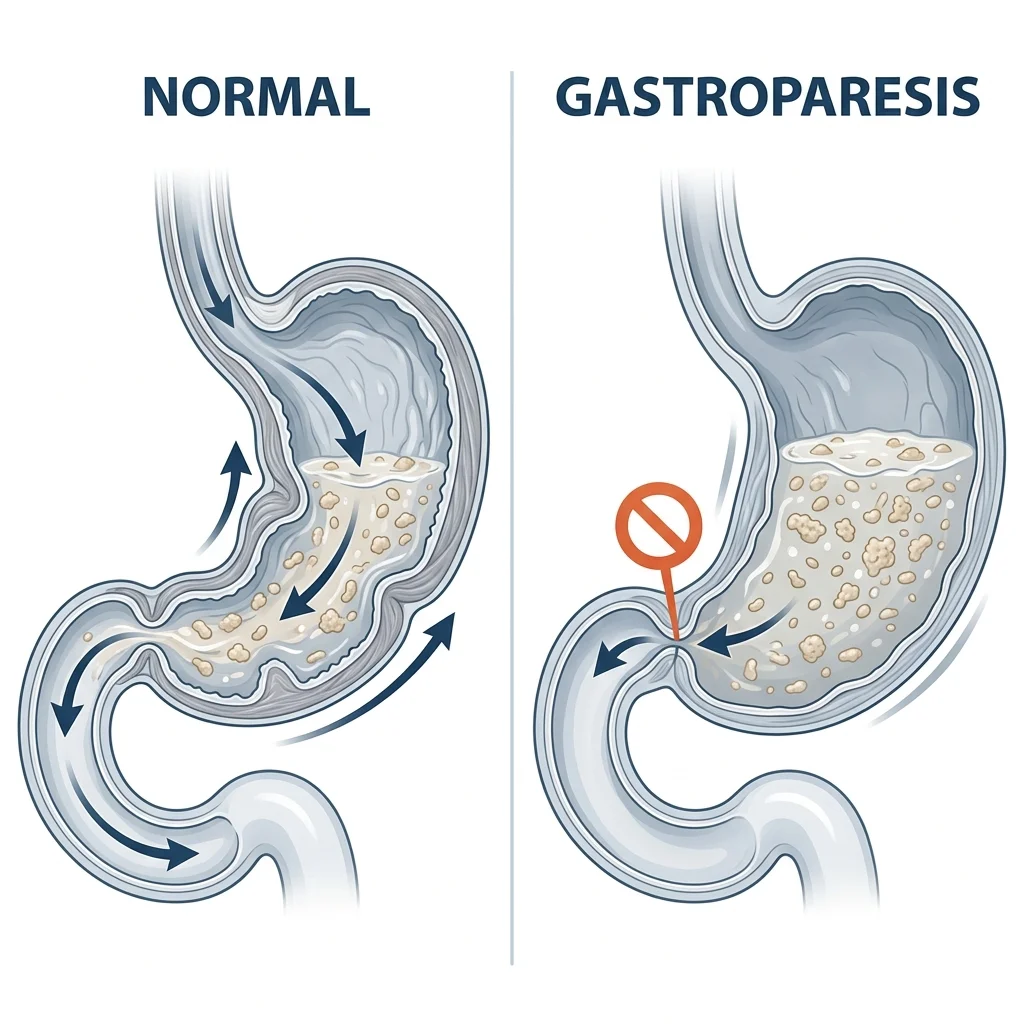
A published case report details what this looks like clinically. A 53-year-old woman started semaglutide 0.5mg weekly for weight loss and lost 40 pounds in four months. She presented with three weeks of nausea and abdominal pain. CT scan showed no obstruction. An endoscopy found semisolid food still in her stomach fundus. A repeat endoscopy 24 hours later, after clear liquids only, still found retained food, a finding highly consistent with gastroparesis. She stopped semaglutide. Her symptoms resolved completely within one month.
That reversibility is typical. "Stopping the GLP-1 or cutting down the dose will usually improve it," Dr. Periyalwar told National Jewish Health. The condition differs from diabetic gastroparesis, which results from years of nerve damage and tends to be chronic. GLP-1-induced gastroparesis is pharmacological, driven by receptor overstimulation, and it typically resolves when the drug clears the system.
But "typical" carries important caveats. Semaglutide remains in circulation for about five weeks after the last dose due to its long half-life. A study by Kalas et al. found that 50% of healthy participants showed delayed gastric emptying while taking GLP-1 drugs. That's not gastroparesis in most cases, but it explains why the broader "motility issues" Dr. Periyalwar describes, constipation, alternating diarrhea, esophageal discomfort, are far more common than the less-than-2% gastroparesis figure suggests.
The surgical implications are serious. The American Society of Anesthesiologists recommends holding GLP-1 agonists one week before elective surgery because retained gastric contents create aspiration risk during anesthesia induction. Published case reports document patients who followed standard pre-operative fasting rules but still had food in their stomachs, creating dangerous situations during intubation.
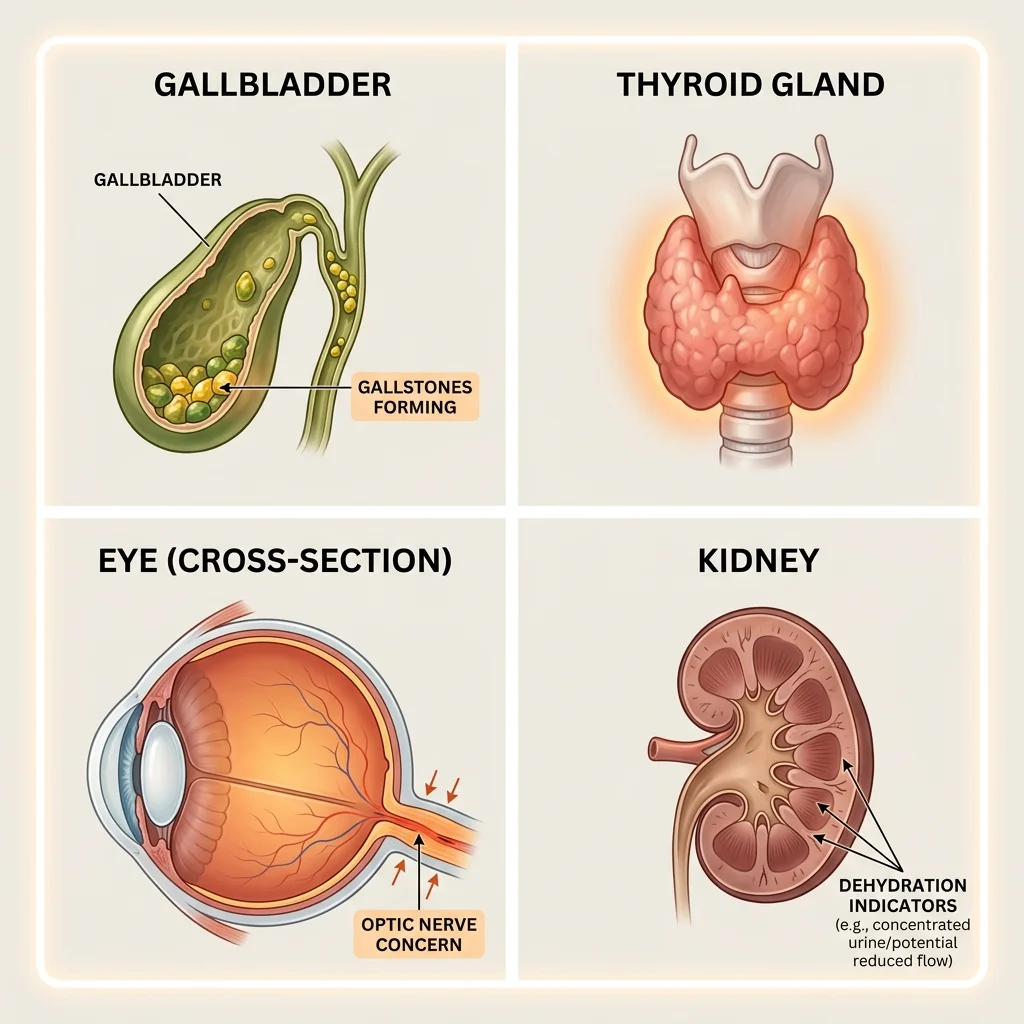
Beyond the Stomach: Gallbladder, Thyroid, Eyes, and Kidneys
GI symptoms dominate the conversation around GLP-1 side effects, which makes sense since they're by far the most common. In the STEP-1 trial of semaglutide 2.4mg, participants reported nausea in 20-44%, diarrhea in 15-30%, vomiting in 10-25%, and constipation in 10-24%. These rates led to treatment discontinuation in 5-9% of participants across major trials. For most people, these symptoms diminish with time and dose escalation. What deserves more attention are the less common but more consequential organ-specific risks.
| Side Effect | Incidence | Severity | Reversible? |
|---|---|---|---|
| Nausea/vomiting/diarrhea | 20-44% | Mild-moderate | Usually improves over weeks |
| Gallbladder disease | 37% increased risk (RR 1.37) | Moderate-severe | May require cholecystectomy |
| Gastroparesis | <2% | Moderate | Usually resolves after stopping |
| Acute pancreatitis | <0.1% per year | Severe | Variable; requires hospitalization |
| Diabetic retinopathy worsening | 3.0% vs 1.8% placebo | Moderate-severe | Linked to rapid glucose improvement |
| Acute kidney injury | <1% | Severe | Usually reversible with hydration |
| NAION (vision loss) | Rare (under investigation) | Severe | Often permanent |
Gallbladder disease has the strongest evidence base after GI complaints. A meta-analysis by He et al. spanning 76 randomized controlled trials with 103,371 patients found GLP-1 RAs associated with a 37% increased risk of gallbladder and biliary diseases (RR 1.37). The risk was dose-dependent and duration-dependent, reaching RR 1.56 at higher doses and RR 2.29 in weight loss trials compared to 1.27 in diabetes trials. The mechanism involves suppressed cholecystokinin secretion and reduced gallbladder contractility, leading to bile stasis and gallstone formation. Rapid weight loss compounds this, as it independently promotes cholesterol supersaturation in bile.
Thyroid concerns carry a black box warning. Rodent studies showed GLP-1 RAs caused thyroid tumors including thyroid cancer, prompting an FDA warning. Whether this translates to humans remains unclear. Population-based studies have yielded mixed results. Bezin et al. found that GLP-1 RA use for 1-3 years was associated with an adjusted hazard ratio of 1.58 for all thyroid cancer and 1.78 for medullary thyroid carcinoma. Other large studies found no significant association. No causal link has been established.
Vision complications present two distinct risks. In SUSTAIN-6, semaglutide users showed a 3.0% rate of diabetic retinopathy complications versus 1.8% with placebo (HR 1.76). Researchers attribute this to rapid HbA1c reduction rather than direct drug toxicity. Separately, a retrospective study found concerning rates of nonarteritic anterior ischemic optic neuropathy (NAION): HR 4.28 in diabetic patients and HR 7.64 in overweight/obese patients on semaglutide versus non-users. This is a single retrospective study and needs replication, but NAION causes sudden, often permanent vision loss.
Kidney injury typically occurs as a secondary effect. GI symptoms cause dehydration, dehydration damages kidneys. LEADER reported acute kidney injury in fewer than 1% of patients, and REWIND found no difference in kidney function decline versus placebo. Case reports describe patients who developed renal problems, but most had pre-existing risk factors or severe GI-induced volume depletion.
The mental health story has a clearer ending. The FDA conducted a comprehensive meta-analysis of 91 placebo-controlled trials covering 107,910 patients and found no increased risk of suicidal ideation or behavior with GLP-1 drugs. A separate Sentinel System study of 2,243,138 users confirmed this. In January 2026, the FDA requested manufacturers remove the suicidal behavior warning from GLP-1 drug labels.
Five Groups That Need Extra Scrutiny Before Starting
GLP-1 drugs aren't appropriate for everyone, and the contraindications extend beyond what most prescribers discuss during an initial visit. Some are absolute. Others represent elevated risk that demands closer monitoring.
| Risk Group | Concern | Recommendation |
|---|---|---|
| Personal/family history of MTC or MEN2 | Thyroid cancer risk | Absolute contraindication, do not prescribe |
| History of pancreatitis | Recurrence risk unknown | Manufacturer states it is "not known" if safe |
| Severe gastroparesis | Worsened gastric motility | Not recommended per prescribing information |
| Pre-existing diabetic retinopathy | Rapid glucose improvement may worsen | Monitor closely, especially early in treatment |
| Pregnancy or planning pregnancy | Animal reproductive toxicity data | Stop 2 months before planned conception |
Semaglutide is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia syndrome type 2 (MEN 2). This isn't a soft recommendation. MTC accounts for about 3-4% of thyroid cancers, and MEN2 is a genetic condition affecting roughly 1 in 35,000 people. Prescribers should ask about thyroid cancer family history before writing the first prescription, and patients with elevated calcitonin levels or thyroid nodules need further evaluation.
Patients with a history of pancreatitis occupy an uncomfortable gray zone. Novo Nordisk's own prescribing information states it is "not known if Ozempic can be used in people who have had pancreatitis." Given the dose-dependent risk data, this population warrants particular caution.
Semaglutide is not recommended in patients with severe gastroparesis because it will make the condition worse. Patients with pre-existing motility disorders, including those caused by diabetic neuropathy, should be evaluated before starting treatment. Drug interactions also matter: combining GLP-1 drugs with insulin or sulfonylureas increases hypoglycemia risk, and the delayed gastric emptying can alter the absorption timing of other oral medications.
Women should stop semaglutide two months before planned pregnancy. Animal studies documented reproductive toxicity at maternally toxic doses for semaglutide, liraglutide, and dulaglutide. A multinational cohort study of 51,826 pregnant women found no significant increase in major congenital malformations compared to insulin, but the manufacturer recommendation stands.
The group that gets the least attention is patients with pre-existing kidney vulnerability. Most patients who developed acute kidney injury had risk factors for volume depletion, existing comorbidities, or were taking other nephrotoxic medications. If you're on an ACE inhibitor, a diuretic, and you start a GLP-1 drug that causes persistent vomiting, the dehydration cascade can damage your kidneys before you realize what's happening.
What to Watch For and When to Call Your Doctor
Nausea during the first few weeks of treatment is normal and expected. Nausea that persists for weeks alongside severe abdominal pain radiating to the back warrants an emergency call. Knowing where one ends and the other begins is half the work of safe GLP-1 use.
Stop your GLP-1 medication and contact your doctor immediately if you experience: severe abdominal pain (with or without vomiting) that doesn't resolve, especially if the pain radiates to your back; sudden vision changes or loss in one eye; signs of severe allergic reaction (face/throat swelling, difficulty breathing); symptoms of dehydration that you cannot correct with oral fluids.
Dose titration is the primary defense against GI side effects. Subcutaneous semaglutide starts at 0.25mg weekly, then moves to 0.5mg after four weeks, with further increases to 1mg or 2mg based on tolerance. Gradual escalation reduces the severity and frequency of nausea, vomiting, and diarrhea. Pillarisetti and Agrawal describe a "Three E" approach: Educate patients about expected side effects, Escalate dosage gradually based on individual tolerance, and Effectively manage symptoms with small frequent meals, hydration, and short-term antiemetics when needed.
The STEP-4 trial provides some reassurance on the timeline: among patients who continued semaglutide 2.4mg treatment, the prevalence of nausea, diarrhea, and constipation decreased over time while vomiting rates remained low. Most GI side effects peak during dose escalation and settle within the first several months.
| Monitoring Area | Frequency | What to Watch |
|---|---|---|
| Kidney function | Before starting, then with dose changes | Creatinine, eGFR; increased if GI symptoms cause dehydration |
| Pancreatic symptoms | Ongoing | Severe epigastric pain radiating to back |
| Thyroid | Baseline and if symptoms appear | Neck lumps, hoarseness, difficulty swallowing |
| Eyes (diabetic patients) | Before starting and periodically | Vision changes, especially with rapid glucose improvement |
| Gallbladder | Ongoing, especially with rapid weight loss | Right upper quadrant pain, fever, jaundice |
| Pre-surgical | Before any elective procedure | Hold GLP-1 drugs one week prior per ASA guidelines |
Renal function monitoring is particularly important when patients report GI adverse reactions that could lead to dehydration. The cascade from persistent vomiting to volume depletion to acute kidney injury is well-documented, and patients on concurrent diuretics or nephrotoxic drugs face amplified risk.
Long-term monitoring remains an evolving question. The WHO-commissioned Cochrane reviews noted that most studies were funded by pharmaceutical manufacturers and that independent research on long-term outcomes is limited. Eva Madrid, co-lead researcher from Universidad de Valparaiso, stated that "weight regain after stopping treatment may affect the long-term sustainability of the observed benefits" and called for more independent studies from a public health perspective. Patients on these drugs for weight management may be on them indefinitely, and safety data beyond 3-5 years remains thin.

Frequently Asked Questions
Can GLP-1 drugs cause permanent gastroparesis?
In the vast majority of cases, no. GLP-1-induced gastroparesis differs from diabetic gastroparesis in that it is pharmacologically driven rather than caused by nerve damage. Stopping the medication or reducing the dose will usually resolve symptoms, though semaglutide remains in circulation for about five weeks after the last injection. Persistent gastroparesis after complete drug clearance is not typical and would warrant investigation for other causes.
Should I get my pancreas checked before starting a GLP-1 drug?
Standard prescribing guidelines don't require pancreatic screening before starting treatment. However, if you have a history of pancreatitis, discuss this explicitly with your prescriber because the safety data for this population is unknown. During treatment, report any severe abdominal pain that doesn't resolve, especially pain radiating to the back, as this could signal acute pancreatitis requiring emergency evaluation.
Are GLP-1 drugs safe for long-term use (5+ years)?
This is an honest gap in the evidence. The largest cardiovascular outcomes trials lasted 2-5 years, and the WHO-commissioned Cochrane reviews specifically highlighted limited long-term safety data. The dose-dependent pancreatitis risk and the gallbladder disease risk that increases with treatment duration suggest that long-term monitoring is not optional. Researchers have called for more independent studies to answer this question definitively.
Do GLP-1 drugs cause depression or suicidal thoughts?
Current evidence says no. The FDA conducted the most comprehensive analysis to date, a meta-analysis of 91 trials with 107,910 patients plus a Sentinel System study of over 2.2 million users, and found no increased risk. In January 2026, the FDA requested that manufacturers remove suicidal behavior warnings from GLP-1 drug labels. If you experience mood changes, discuss them with your prescriber, but the evidence does not support a causal link between these drugs and psychiatric harm.
Should I stop my GLP-1 medication before surgery?
Yes. The American Society of Anesthesiologists recommends holding GLP-1 agonists one week before elective surgery due to the risk of retained gastric contents and pulmonary aspiration during anesthesia. Always inform your surgical and anesthesia teams that you are taking a GLP-1 drug, even if your last dose was several days ago.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.