What a CGM actually does (and doesn't do)
A continuous glucose monitor is a coin-sized sensor that sits just under the skin, usually on the upper arm or abdomen. A tiny filament measures glucose in interstitial fluid, the liquid that surrounds your cells, and transmits readings to a phone app every one to five minutes. The technology was first cleared by the FDA in 1999 for people with diabetes who need to track their insulin response in real time.
The key distinction: CGMs measure interstitial glucose, not blood glucose. There is a lag of several minutes between what is happening in your bloodstream and what the sensor detects. For someone managing insulin doses, that difference is manageable. For someone trying to "optimize" their response to a specific meal, it introduces noise that can mislead more than inform.
CGMs also cannot tell you why your glucose moved. A spike after lunch might reflect the carbohydrate load, poor sleep the night before, acute stress, the pace at which you ate, or the composition of your gut microbiome. The number alone does not diagnose anything. According to the Cleveland Clinic, even for people with diabetes, CGM data requires clinical interpretation alongside other health markers to be actionable.
Why healthy people started wearing glucose monitors
The sales pitch writes itself: see how food, exercise, and sleep affect your blood sugar in real time, then adjust. Biohacking communities adopted CGMs early. Venture-backed startups built subscription businesses around selling this idea to people who had never received a diabetes diagnosis.
Two shifts drove adoption. The quantified-self movement had already normalized wearing sensors for sleep and heart rate, so adding glucose felt like a natural extension. Meanwhile, research on metabolic inflammation and insulin resistance made people aware that metabolic dysfunction often starts years before a formal diabetes diagnosis. Companies like Levels and Nutrisense capitalized on both trends by pairing a telehealth prescription with an app and a monthly subscription. No endocrinologist required.
There is also a signaling component. Wearing a CGM says something about how seriously you take your health, similar to sporting an Oura ring or getting biological age testing. Whether the data these devices produce translates into actual health improvements for people whose blood sugar is already normal is a different question.
What "normal" glucose actually looks like on a CGM
If you strap on a CGM for the first time, your readings will bounce around. That can be alarming if you do not know what to expect. A multicenter study of 153 healthy, nondiabetic people wearing Dexcom G6 sensors established what normal actually looks like:
| Metric | Value |
|---|---|
| Mean glucose (all ages except 60+) | 98-99 mg/dL |
| Mean glucose (age 60+) | 104 mg/dL |
| Time between 70-140 mg/dL | 96% (median) |
| Time above 140 mg/dL | 2.1% (~30 min/day) |
| Time below 70 mg/dL | 1.1% (~15 min/day) |
| Coefficient of variation (glucose variability) | 17 +/- 3% |
In practical terms, a healthy person spends about 96% of their day with glucose between 70 and 140 mg/dL. They dip above 140 for about half an hour daily and below 70 for about 15 minutes. The remaining variability is ordinary physiology: your body is constantly adjusting glucose in response to meals, movement, hormones, and circadian rhythm.
A large study from the PREDICT research program involving 4,135 participants found similar patterns. Median glycemic variability was 14.8%, and participants spent about 96% of time in the standard ADA range of 3.9-7.8 mmol/L. When a stricter range was applied (3.9-5.6 mmol/L), the time dropped to 75%, meaning people spent roughly a quarter of their day outside even a tighter window. That quarter of the day is not pathological. It is eating, exercising, sleeping, and living.
Key numbers to keep in mind: Healthy people spend about 96% of their time with glucose between 70-140 mg/dL. Post-meal spikes above 140 mg/dL are normal and brief, lasting roughly 30 minutes per day. A glucose variability coefficient of around 17% is standard.
Glucotypes and hidden spikes: the Stanford findings
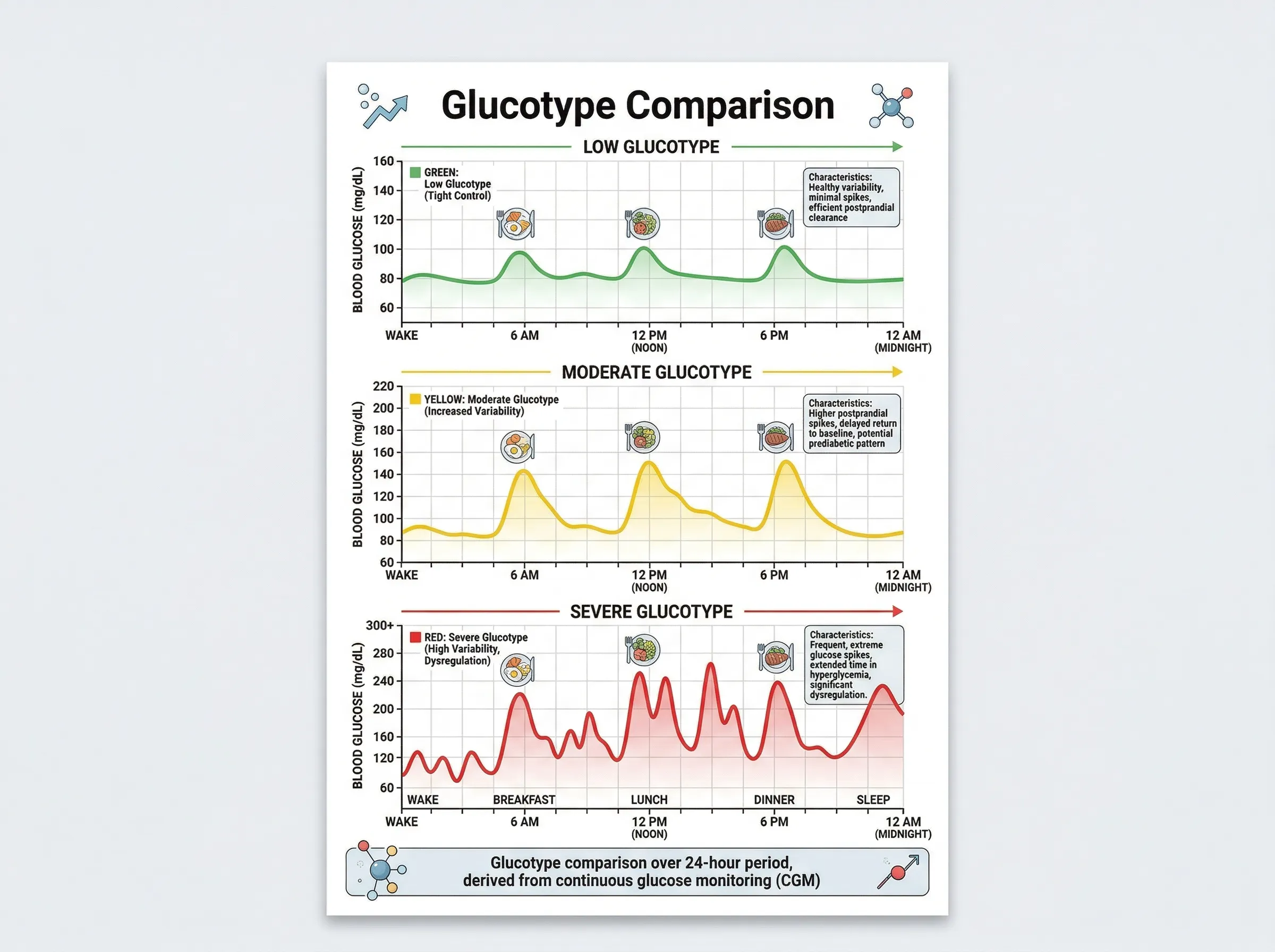
The most-cited study fueling the wellness CGM movement came from Stanford in 2018. Michael Snyder's lab fitted 57 participants with continuous glucose monitors and recorded nearly 500,000 measurements over two to four weeks. Using spectral clustering analysis of glucose patterns, the researchers classified people into three "glucotypes" based on the variability of their glucose curves:
| Glucotype | Variability | Mean glucose in class | Clinical significance |
|---|---|---|---|
| Low | Minimal fluctuation | 77 mg/dL | Stable, healthy pattern |
| Moderate | Intermediate swings | 96 mg/dL | Some elevated responses |
| Severe | Large, frequent spikes | 122 mg/dL | Prediabetic-range excursions |
The finding that generated the most attention: 25% of people classified as normoglycemic by standard tests (fasting glucose, HbA1c, oral glucose tolerance test) still exhibited the severe glucotype. These individuals hit prediabetic glucose levels up to 15% of the time and diabetic-range levels about 2% of the time. Standard blood tests had missed these patterns entirely.
The breakfast sub-study was even more provocative. When 30 participants alternated between cornflakes with milk, a peanut butter sandwich, and a protein bar, 80% of them spiked into prediabetic territory after the cereal. The peanut butter sandwich, with more fat, fiber, and protein, produced far milder responses across the group. Individual variation was substantial, though. Some people spiked hard after the protein bar but stayed flat after cereal. Others showed the reverse.
The individual variation matters. But so does what the study did not show: that identifying your glucotype and changing your diet in response prevents diabetes or cardiovascular disease. The researchers wrote that "longer-term studies are warranted" to determine whether severe glucotype classification predicts progression. Those longitudinal studies remain unpublished.
The wellness CGM boom: Levels, Nutrisense, and Dexcom Stelo
Companies moved fast to bridge the gap between "interesting research finding" and "you should buy this." Several now sell CGM subscriptions to non-diabetic consumers:
| Company | Model | Approximate cost | Approach |
|---|---|---|---|
| Levels | Subscription + app | $199/month | Scores meals on glucose response |
| Nutrisense | Subscription + dietitian | $225/month | CGM paired with nutrition coaching |
| Dexcom Stelo | OTC, no prescription | ~$99/month | Standalone sensor, basic app |
| Signos | Subscription + app | $199/month | CGM-guided weight management |
The Dexcom Stelo marked a turning point. The FDA cleared it in March 2024 as the first over-the-counter CGM, available to adults 18 and older who do not use insulin, including people without diabetes. No prescription, no doctor visit. The FDA did note that users "should not make medical decisions based on the device's output without talking to their healthcare provider."
The math explains the rush. Roughly 37 million Americans have diabetes and about 96 million have prediabetes. Over 200 million adults have neither. Even a small slice of that third group, converted into monthly subscribers, represents substantial revenue.
A 2025 systematic review examining CGM use for cardiovascular prevention in non-diabetic populations found just seven studies meeting inclusion criteria. The evidence showed CGM could personalize exercise timing (walking before a person's typical post-meal glucose peak reduced glucose, insulin, and C-peptide levels) and serve as a motivational tool. But the reviewers concluded that "direct evidence on changes in traditional cardiovascular risk factors was limited" and called for long-term trials.
The case against CGMs for healthy people
The skeptics include endocrinologists, academic medical centers, and glucose researchers.
Harvard Health's assessment is blunt: "I could find no published study suggesting that monitoring translates into improved health for healthy people without diabetes or other blood sugar problems." The author, a Harvard Medical School professor, called CGMs for healthy people potentially "the latest health monitoring fad wasting effort and money."
Their concerns are specific:
Normal variation gets pathologized. Post-meal glucose of 145 mg/dL in a healthy person is a brief, normal physiological event. Wellness CGM apps often flag this as a "spike" and assign it a poor score. Over time, this framing can convince people that their healthy bodies are malfunctioning. The comprehensive review in the Journal of Diabetes Science and Technology noted there is "an absence of consensus standards for defining abnormal values" in non-diabetic CGM users and "an absence of consensus standards for how to respond to abnormal values."
Anxiety and disordered eating. Research in Diabetes Technology and Therapeutics found the constant stream of glucose data can increase anxiety. When apps frame every glucose fluctuation as something to fix, some users start restricting foods, timing meals obsessively, or exercising after eating specifically to flatten their curves. For people predisposed to eating disorders, this is a real risk. The Cleveland Clinic flags "information overload" and "diabetes distress or burnout" as known CGM drawbacks, even for diabetic patients who medically need the devices.

No randomized trial showing benefit. The study that would settle the debate, a randomized controlled trial comparing health outcomes between healthy CGM users and non-users over several years, does not exist. Without it, the argument for CGMs in healthy populations rests on plausibility and self-reported behavior change, not measured outcomes.
Cost with uncertain return. At $100-$225 per month, a year of CGM monitoring runs $1,200-$2,700. Insurance does not cover it for non-diabetic use. For the same money, a person could fund a gym membership, regular bloodwork panels, or consultation with a registered dietitian, interventions with far stronger evidence behind them.
Access and equity concerns. Some critics argue that marketing CGMs to healthy, affluent consumers diverts manufacturing capacity and public attention from the people who medically depend on them. Millions of diabetic patients worldwide still face cost barriers to CGM access.
Who might genuinely benefit
Writing off CGMs for everyone without diabetes goes too far. There are specific situations where a few weeks of data could genuinely inform decisions:
People with prediabetes. About 84 million Americans have prediabetes, and roughly 90% do not know it. Of those who do know, up to 70% will progress to type 2 diabetes without intervention. CGM data could provide the motivational feedback loop needed to sustain the diet and exercise changes proven to prevent that progression. The Diabetes Prevention Program demonstrated that lifestyle interventions reduce conversion to diabetes by 58% at four years. A CGM is not the intervention itself, but it might make the intervention stick.
People with a family history of type 2 diabetes. The Stanford glucotype data showed that standard tests miss a quarter of people with significant glucose dysregulation. If you have a parent or sibling with diabetes, are overweight, or have other metabolic risk factors, a short trial of CGM monitoring could reveal patterns worth discussing with your doctor. One or two weeks of data may be more informative than another fasting glucose test.
People using metabolic interventions. If you are actively experimenting with resistant starch for blood sugar management, intermittent fasting protocols, or dietary changes aimed at improving metabolic health, a short CGM trial can show whether the intervention is affecting your glucose patterns. This is time-limited, hypothesis-driven monitoring, not indefinite tracking.
The CGM-HYPE study from 2024 highlights another angle. Researchers at Heinrich-Heine University fitted healthy young adults with CGMs for 14 days while running standardized tests of diet, exercise, and stress. Anaerobic exercise produced significantly greater glucose excursions (28.7 mg/dL) compared to aerobic exercise (8.8 mg/dL). The Trier Social Stress Test, a standard psychological stress protocol, triggered a statistically significant glucose response. For people under chronic stress or training intensely, those patterns might be worth knowing about.
A practical framework: Consider a CGM if you have prediabetes, a strong family history of diabetes, or want to evaluate a specific dietary intervention. Use it for 2-4 weeks with a clear question in mind, not as an indefinite lifestyle accessory. Discuss findings with a healthcare provider.
Frequently Asked Questions
How much does a CGM cost without insurance for non-diabetic use?
Over-the-counter options like Dexcom Stelo run approximately $99 per month. Subscription services from companies like Levels and Nutrisense cost $199-$225 per month. Insurance does not cover CGMs for people without diabetes. A full year of monitoring ranges from $1,200 to $2,700 out of pocket.
Can a CGM diagnose prediabetes or diabetes?
No. CGMs are not diagnostic tools. They measure interstitial glucose trends, not fasting plasma glucose or HbA1c, which are the standard diagnostic criteria. However, CGM data showing frequent excursions above 140 mg/dL may indicate a reason to get formal testing through your doctor. The Stanford glucotype research found that 25% of people with normal standard test results still showed significant glucose dysregulation on CGM.
Is it normal for glucose to spike above 140 mg/dL after eating?
Yes. In a multicenter study of 153 healthy people, the median time above 140 mg/dL was about 30 minutes per day. Post-meal glucose rises are a normal physiological response. The concern begins when glucose stays elevated for extended periods or regularly exceeds 180 mg/dL, which is uncommon in people without metabolic dysfunction.
Do CGMs help with weight loss?
There is no strong evidence that CGM use directly causes weight loss in healthy people. Some users report that seeing glucose spikes after certain foods motivates them to change eating habits. A study of obese individuals without diabetes found that using a CGM plus counseling increased motivation to exercise. But no controlled trial has demonstrated sustained weight loss attributable to CGM use alone.
Are there any risks to wearing a CGM if you are healthy?
Physical risks are minimal: occasional skin irritation or adhesive allergy at the sensor site. The larger risk is psychological. The constant data stream can trigger anxiety, orthorexic eating patterns, or unnecessary dietary restriction. Harvard Health has cautioned that biologically insignificant glucose dips could lead to unnecessary snacking, potentially causing weight gain.
Related Articles
- Resistant Starch: The Cheap Gut Health Hack for Blood Sugar and Metabolism — How a specific type of fiber can improve your glucose response and feed beneficial gut bacteria.
- Inflammation and Chronic Disease: A Complete Guide to Anti-Inflammatory Living — The metabolic inflammation that underlies insulin resistance and why it matters before a diabetes diagnosis.
- Autophagy: How to Trigger It Through Fasting, Exercise, and Diet — Fasting protocols and their effect on metabolic health, including glucose regulation.
- Biological Age Testing: How to Measure and Improve Your Pace of Aging — Other biohacking tools for health tracking and what the evidence says about each.
- Akkermansia: The Next-Generation Probiotic for Metabolic Health — The gut-metabolism connection and how specific bacteria influence glucose regulation.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.