What Makes Akkermansia Different from Traditional Probiotics?
If you have been following gut health research over the past few years, you have probably noticed one bacterial name appearing with increasing frequency: Akkermansia muciniphila. First isolated from a human stool sample in 2004 by researchers at Wageningen University in the Netherlands, this microbe has gone from obscurity to being called "one of the most promising next-generation probiotics" in the scientific literature. But what makes it fundamentally different from the Lactobacillus and Bifidobacterium strains lining supplement store shelves?
It comes down to where it lives and what it eats. Most commercial probiotics are tourists in your gut. You swallow them, they drift through, and the vast majority never settle down. Akkermansia is different. It sets up shop directly in the mucus layer coating your intestinal lining and feeds almost exclusively on mucin, the glycoprotein that makes mucus thick and protective. This bacterium is not passing through. It is part of the infrastructure.
Akkermansia belongs to the phylum Verrucomicrobia, making it taxonomically unrelated to virtually every commercial probiotic on the market. In healthy adults, it accounts for roughly one to four percent of total gut bacteria, which may sound modest until you realize that individual species rarely command that kind of real estate. It is among the top twenty most abundant species in the human gut, and it begins colonizing the intestine within the first year of life, even appearing in breast milk as a likely vehicle for mother-to-infant transfer.
Key Distinction: While traditional probiotics (Lactobacillus, Bifidobacterium) are largely transient and ferment dietary fibers, Akkermansia is a permanent resident that feeds on the mucus layer itself, directly maintaining the gut's structural integrity from within.
Here is the part that trips people up: Akkermansia eats mucus, yet having more of it around is linked to a thicker mucus layer, not a thinner one. The reason is that when Akkermansia chews through old mucin, it nudges goblet cells into producing fresh replacement mucus. Picture a gardener pruning dead branches so the plant sends out new growth. The constant turnover keeps the intestinal lining in better shape than if it were left alone. No traditional probiotic does anything like this.
Your Gut Has a Bouncer, and Its Name Is Akkermansia
Your intestinal lining does not get enough credit. It is a single-cell-thick wall with a contradictory assignment: let nutrients in while keeping bacteria, toxins, and undigested proteins out. When the wall springs leaks, researchers call it "increased intestinal permeability," and the downstream effects are not subtle. Bacterial fragments slip into the bloodstream, the immune system ramps up, and the resulting low-grade inflammation can drag metabolic health down with it.
Akkermansia shores up this barrier in at least three ways, and the details help explain the excitement around this organism.
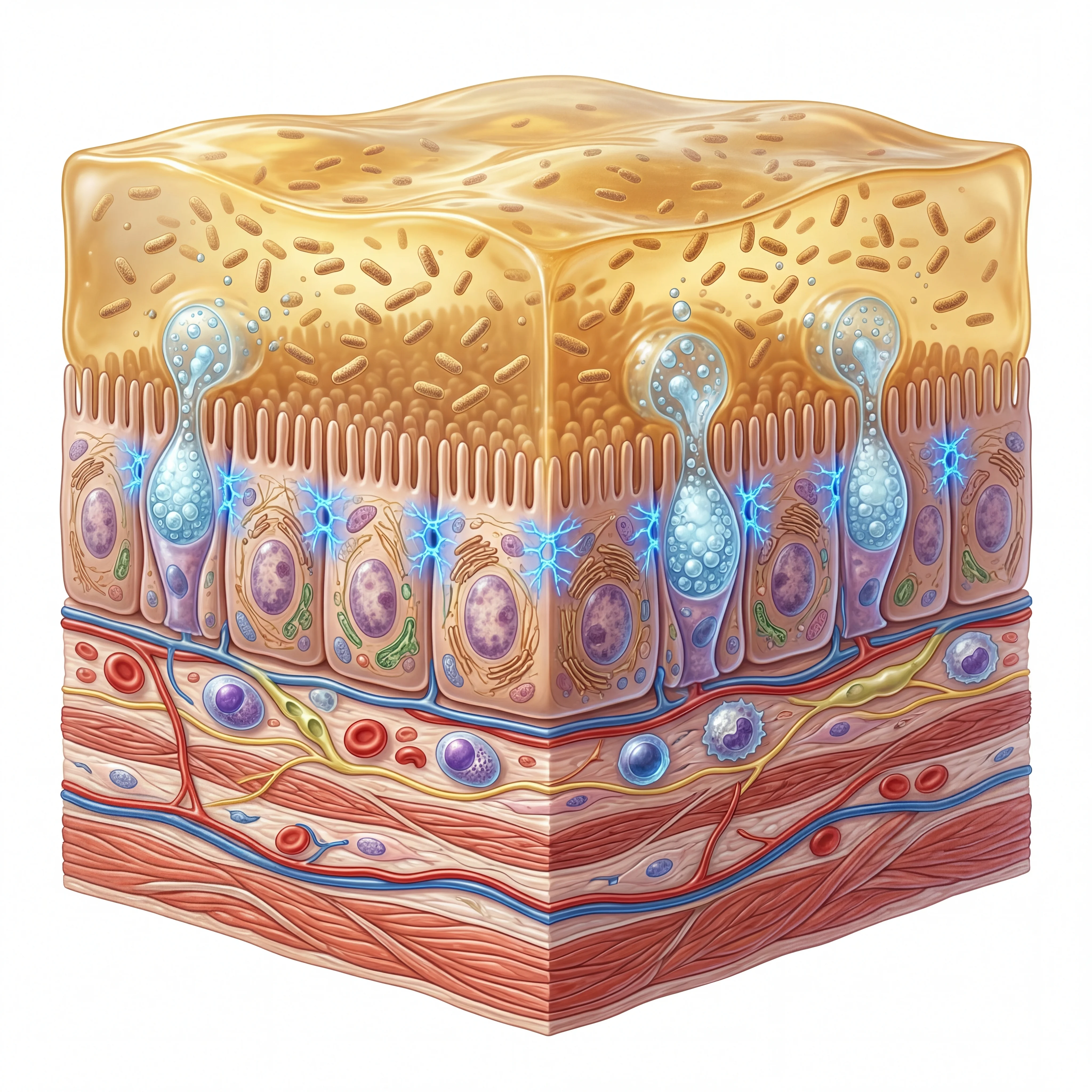
First, it stimulates mucus renewal. As Akkermansia breaks down older mucin, the byproducts signal goblet cells to ramp up production of fresh mucus. Research published in Gut Pathogens (2024) showed that Akkermansia colonization upregulates a molecular sensor called the NLRP6 inflammasome, which accelerates a cellular recycling process (autophagy) within goblet cells. The net effect is a more robust and continuously refreshed mucus shield.
Second, it strengthens the seals between intestinal cells. The cells lining your gut are held together by protein complexes called tight junctions. In preclinical models, Akkermansia supplementation increased the expression of tight junction proteins while also boosting the number of mucin-producing goblet cells. With tighter seals and more mucus, the barrier becomes substantially harder to breach.
Third, it produces short-chain fatty acids that fuel barrier cells. When Akkermansia ferments mucin, the primary metabolic outputs are acetate and propionate. These short-chain fatty acids serve as direct energy sources for the intestinal epithelial cells tasked with maintaining barrier integrity. They also exert anti-inflammatory effects by reducing the amount of lipopolysaccharide (LPS), a bacterial toxin, that leaks from the gut into circulation. Animal studies have documented up to a fifty percent reduction in circulating LPS following Akkermansia supplementation.
| Barrier Mechanism | How Akkermansia Contributes | Downstream Effect |
|---|---|---|
| Mucus layer thickness | Stimulates goblet cell renewal via NLRP6 inflammasome | Stronger physical shield against pathogens |
| Tight junction integrity | Upregulates tight junction protein expression | Reduced intestinal permeability ("leaky gut") |
| SCFA production | Produces acetate and propionate from mucin fermentation | Energy for barrier cells, reduced systemic LPS |
| Immune modulation | Activates TLR2 signaling, promotes regulatory T cells | Anti-inflammatory environment in the gut wall |
The immune angle is worth paying attention to as well. Akkermansia's outer membrane protein Amuc_1100 plugs into Toll-like receptor 2 (TLR2) on immune cells, dialing down inflammatory signaling. Another secreted protein, AmTARS, pushes macrophages toward their anti-inflammatory M2 form and ramps up production of interleukin-10, one of the body's built-in brakes on inflammation. So Akkermansia is not only building a physical wall; it is also coaching the local immune cells to stay calm. For other angles on immune support, our piece on quercetin for immunity and inflammation covers complementary territory.
From Blood Sugar to Body Fat: The Metabolic Evidence
The barrier protection alone would be interesting. But what really caught the medical community's attention is what happens to metabolism when Akkermansia levels change. The research has progressed from "people with diabetes have less of this bacterium" to actual placebo-controlled trials in humans, and the data is worth a close look.
The observational foundation is broad. Multiple studies have consistently found that people with obesity, type 2 diabetes, metabolic syndrome, and cardiovascular disease tend to harbor significantly lower levels of Akkermansia compared to metabolically healthy individuals. In animal models, high-fat diets slashed Akkermansia populations by a hundredfold within just four weeks. While correlation never proves causation, the consistency of this pattern across diverse populations and conditions demanded intervention trials.

The landmark human trial came from Depommier and colleagues, published in Nature Medicine in 2019. In this proof-of-concept study, overweight and obese volunteers received either live Akkermansia, pasteurized (heat-killed) Akkermansia, or placebo daily for three months. The pasteurized form produced the most striking results: significantly improved insulin sensitivity, reduced plasma insulin levels, and lower total cholesterol. There were also modest reductions in body weight, fat mass, and hip circumference compared to baseline. The live form showed benefits too, but the pasteurized version outperformed it on several metabolic markers.
Why Pasteurized Works: Heat-killing the bacteria does not destroy the beneficial outer membrane protein Amuc_1100. This protein remains stable at pasteurization temperatures and can still interact with TLR2 receptors to trigger metabolic improvements, without the need for live bacterial colonization.
The mechanistic picture has grown richer since that trial. A protein secreted by Akkermansia, designated P9, has been shown to stimulate secretion of GLP-1, the same hormone targeted by blockbuster diabetes and weight-loss drugs like semaglutide. P9 also promotes thermogenesis in brown adipose tissue, the metabolically active fat that burns calories to generate heat. The connection to bioactive peptides is worth noting: like pharmaceutical peptides, these bacterial proteins exert precise hormonal effects despite being produced by a gut microbe rather than a lab.
| Metabolic Marker | Effect in Human Trial (Depommier 2019) | Mechanism |
|---|---|---|
| Insulin sensitivity | Significantly improved | Reduced endotoxemia, TLR2-mediated signaling |
| Plasma insulin | Decreased | Improved beta-cell function via reduced inflammation |
| Total cholesterol | Decreased | Altered bile acid metabolism, reduced hepatic inflammation |
| Body weight | Slight reduction | GLP-1 stimulation, brown fat thermogenesis |
| Fat mass | Slightly reduced vs. baseline | P9 protein activating brown adipose tissue |
What ties this together is the gut barrier connection from the previous section. The metabolic improvements likely trace back, at least partly, to reduced "metabolic endotoxemia," the low-grade inflammation that happens when bacterial LPS leaks through a compromised intestinal wall into the bloodstream. Seal the barrier, and you cut off the inflammatory signals driving insulin resistance and fat storage. It is a fix-the-plumbing approach to metabolism, and it has a certain elegance to it. For more on nutritional strategies that complement metabolic health, see our coverage of electrolytes and metabolic function.
Myth vs. Reality: Sorting Akkermansia Hype from Science
Whenever a microbe gets popular, the claims outrun the data. Akkermansia is no exception. Here is what the research actually says versus what shows up in supplement ads and wellness TikToks.
| Claim | Reality | Evidence Level |
|---|---|---|
| "Akkermansia is a miracle cure for obesity" | The human trial showed modest reductions in weight and fat mass, not dramatic weight loss. It is a metabolic modulator, not a magic bullet. | One human RCT; consistent animal data |
| "More Akkermansia is always better" | There appears to be an upper ceiling. In healthy mice already at normal levels, supplementation did not increase abundance further. Excessive enrichment in certain conditions may actually be harmful. | Strong animal evidence; theoretical human concern |
| "You can only get Akkermansia from supplements" | Dietary strategies including polyphenol-rich foods, caloric management, and prebiotic fibers can meaningfully increase your natural Akkermansia levels. | 29 human clinical trials reviewed |
| "Pasteurized is inferior to live bacteria" | In the landmark trial, pasteurized Akkermansia actually outperformed the live form on several metabolic markers. The key protein Amuc_1100 survives heat treatment. | One human RCT |
| "Akkermansia is safe for everyone" | Caution is warranted for people with active inflammatory bowel disease, active infections, or those in post-antibiotic recovery phases. | Strong animal evidence; clinical guidelines pending |
Can You Feed Your Way to More Akkermansia?
Before reaching for a supplement, it is worth knowing that your dietary choices exert powerful influence over Akkermansia populations. A systematic review of 29 human clinical trials involving over 1,400 participants identified several dietary interventions that reliably shift Akkermansia abundance.
Polyphenol-rich foods top the list. Pomegranate extract and resveratrol, the polyphenol found in grape skins and red wine, both increased Akkermansia levels in controlled trials. The mechanism likely involves polyphenols reaching the colon largely intact and selectively nourishing Akkermansia while inhibiting competing species. Cranberries, green tea, and dark chocolate are also polyphenol-dense foods that may support Akkermansia growth, though direct trial data for these specific foods is still emerging. Our guide on turmeric benefits covers another polyphenol powerhouse with complementary gut effects.

Prebiotic fibers matter, but the type is critical. Inulin, a prebiotic fiber found in chicory root, garlic, onions, and artichokes, increased Akkermansia abundance in multiple trials. However, a low-FODMAP diet actually decreased Akkermansia levels, which creates a tension for people managing irritable bowel syndrome who may be avoiding the very foods that nourish this bacterium. Polydextrose and sodium butyrate supplementation also showed positive effects.
Caloric restriction and fasting may help. The same systematic review found that caloric restriction protocols increased Akkermansia abundance, possibly because fasting shifts the gut toward mucin-fueled metabolism when dietary substrates are scarce. This is a scenario where Akkermansia's unique ability to thrive on mucin gives it a competitive advantage over bacteria that depend on dietary fiber.
| Dietary Strategy | Effect on Akkermansia | Evidence Strength |
|---|---|---|
| Pomegranate extract | Increased | Human RCT |
| Resveratrol | Increased (in Caucasians) | Human RCT (ethnic variability noted) |
| Inulin (prebiotic fiber) | Increased | Multiple human trials |
| Caloric restriction | Increased | Human clinical trial |
| Polydextrose | Increased | Human RCT |
| Sodium butyrate | Increased | Human RCT |
| Low-FODMAP diet | Decreased | Human RCT |
| High-fat Western diet | Decreased (up to 100-fold) | Consistent animal evidence |
One surprising finding: certain other probiotics can boost Akkermansia indirectly. Bifidobacterium animalis subspecies lactis increased Akkermansia abundance roughly a hundredfold in animal studies, likely by producing short-chain fatty acids that stimulate mucin production and thus provide more substrate for Akkermansia to thrive on. Interestingly, Lactobacillus rhamnosus did not produce the same effect, suggesting that the cross-feeding relationship is species-specific. For broader context on how probiotics interact, our guide to probiotic foods and supplements offers a wider view.
The Supplement Question: Pasteurized, Live, or Neither?
Akkermansia supplements are now available to consumers, but picking one is not as straightforward as grabbing a bottle off the shelf. The formulation differences are real and worth understanding.
Pasteurized Akkermansia has the strongest regulatory and clinical backing. The European Food Safety Authority (EFSA) approved pasteurized A. muciniphila as a safe novel food, specifically the heat-treated form. In the Depommier trial, participants taking 10 billion pasteurized cells daily for three months experienced no adverse effects. Formal toxicology testing, including a 90-day subchronic study in rats and bacterial mutation assays, confirmed no safety concerns.
Live Akkermansia presents manufacturing challenges. The bacterium is technically anaerobic, meaning it cannot survive prolonged oxygen exposure, though research shows over 90 percent can endure up to an hour of ambient air. Maintaining viability through supplement manufacturing, shipping, and storage is substantially harder than for robust Lactobacillus strains. Some companies have developed proprietary encapsulation technologies to address this, but consistent viability across commercial products remains an open question.
Dosing appears to matter. Animal studies demonstrated clear dose-dependence: a lower dose of 4 million CFU failed to improve glucose tolerance, while 200 to 400 million CFU showed consistent metabolic benefits. The human trial used 10 billion cells daily. Below-threshold doses may offer no measurable benefit.
There is also an important ceiling effect. In animal models, supplementing healthy subjects that already had normal Akkermansia levels did not push abundance higher or produce additional metabolic benefits. This suggests supplementation is most relevant for individuals whose levels are depleted, particularly those consuming high-fat diets, living with metabolic disorders, or experiencing reduced microbial diversity.
Who Should Think Twice Before Supplementing
The safety data for Akkermansia in otherwise healthy overweight individuals looks solid. But "generally safe" and "safe for everyone" are very different statements, and the research flags several situations where supplementation could do more harm than good.
Inflammatory bowel disease (IBD). In people with Crohn's disease or ulcerative colitis, the gut barrier is already compromised. Adding a mucin-degrading bacterium to an environment where the mucus layer is damaged and inflamed may backfire. A preclinical study using an IBD mouse model (IL-10 knockout) found that Akkermansia supplementation could actually exacerbate colitis. The logic is straightforward: mucin degradation in a healthy gut stimulates renewal, but in a gut that cannot adequately produce new mucus, it may accelerate erosion.
Active infections. Research showed that in the presence of Salmonella typhimurium infection, Akkermansia pre-colonization made the pathogen more dominant rather than less. This is a case where the mucin-degrading activity may inadvertently create openings for opportunistic pathogens when the immune system is already fighting an invader.
Post-antibiotic recovery. After broad-spectrum antibiotic treatment, the gut microbiota undergoes a fragile rebuilding process. Introducing Akkermansia during this window can interfere with the reconstitution of the microbial community and potentially worsen barrier damage rather than repair it.
Neurological considerations. An intriguing and still-unresolved finding is that patients with Parkinson's disease and multiple sclerosis tend to show elevated Akkermansia abundance compared to healthy controls. This does not mean Akkermansia causes neurological disease, but the correlation is consistent enough that supplementation in these populations should await further research. Supporting overall brain and nervous system health through validated strategies remains the prudent approach.
Endocrine and gynecological disorders. Women with polycystic ovary syndrome (PCOS) or endometriosis may carry elevated IBD risk, with one longitudinal study finding that women with endometriosis were 80 percent more likely to develop IBD. Since Akkermansia supplementation could be counterproductive in IBD, these populations deserve individualized assessment before use.
Frequently Asked Questions
What does Akkermansia muciniphila actually do in the gut?
Akkermansia lives in the mucus layer lining your intestines, where it feeds on mucin glycoproteins. This feeding process stimulates goblet cells to produce fresh mucus, strengthens tight junctions between intestinal cells, and generates short-chain fatty acids that reduce inflammation and support barrier integrity. It essentially maintains the gut's protective shield from within.
Is pasteurized Akkermansia better than the live form?
In the only published human clinical trial, pasteurized (heat-killed) Akkermansia produced stronger improvements in insulin sensitivity and cholesterol levels compared to the live form. The key beneficial protein, Amuc_1100, survives pasteurization and can still activate immune receptors. Pasteurized forms also have the advantage of consistent dosing and EU regulatory approval as a novel food.
Can I increase my Akkermansia levels through diet alone?
Yes. Clinical trials have shown that polyphenol-rich foods such as pomegranate and grape-derived resveratrol, prebiotic fibers like inulin from chicory root and garlic, caloric restriction, and certain probiotic strains like Bifidobacterium animalis subspecies lactis can meaningfully increase Akkermansia populations. High-fat Western diets, conversely, can reduce levels by up to a hundredfold.
Who should avoid Akkermansia supplements?
People with active inflammatory bowel disease, current gastrointestinal infections, or those in post-antibiotic recovery should avoid Akkermansia supplementation until further safety data is available. Additionally, individuals with Parkinson's disease, multiple sclerosis, or estrogen-dependent conditions like endometriosis that carry elevated IBD risk should consult their healthcare provider before use.
How long does it take for Akkermansia to produce noticeable effects?
The landmark human trial administered pasteurized Akkermansia daily for three months before measuring metabolic outcomes. Dietary strategies to boost natural levels may take several weeks to shift microbial populations meaningfully. There is currently no evidence supporting rapid, short-term benefits from Akkermansia supplementation.
Related Articles
- Health Benefits of Probiotic Foods and Supplements - A comprehensive guide to how fermented foods and targeted probiotic strains support digestive health.
- Gut Health Personalization and Microbiome Testing - How emerging testing technologies can help you understand your unique microbial profile.
- Collagen Supplements for Skin, Joints, and Gut - Explores how collagen peptides support the intestinal lining alongside microbial strategies.
- Liver Support Supplements: Milk Thistle, NAC, and Detox - The liver-gut axis and how supporting hepatic function complements gut barrier health.
- Berberine for Blood Sugar and Weight Management - Another evidence-based compound with overlapping metabolic benefits.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.