Why Vetting Your Peptide Source Matters More Than the Peptide Itself
A customer paid $340 for semaglutide from a vendor with a polished website and "99.8% purity" on every product page. He sent a sample to Janoshik for independent analytical testing. The result: 38% actual purity, with the balance being mannitol and unidentified filler.
He is not an outlier. An analysis referenced by BBC News, citing data from analytical testing firm Finnrick, found that approximately 8% of tested research peptide samples contained bacterial endotoxin contamination. Endotoxins do not show up on a standard HPLC purity test. In small doses they cause fever and body aches. In larger amounts, they trigger septic shock.
The peptide you choose matters. But the source you buy it from determines whether you are injecting the peptide you chose, a fraction of it mixed with filler, or something contaminated with bacterial fragments that your body will react to violently. These seven red flags are the tells that separate vendors worth trusting from those worth avoiding.
Red Flag 1: No Certificate of Analysis (or One That Doesn't Add Up)
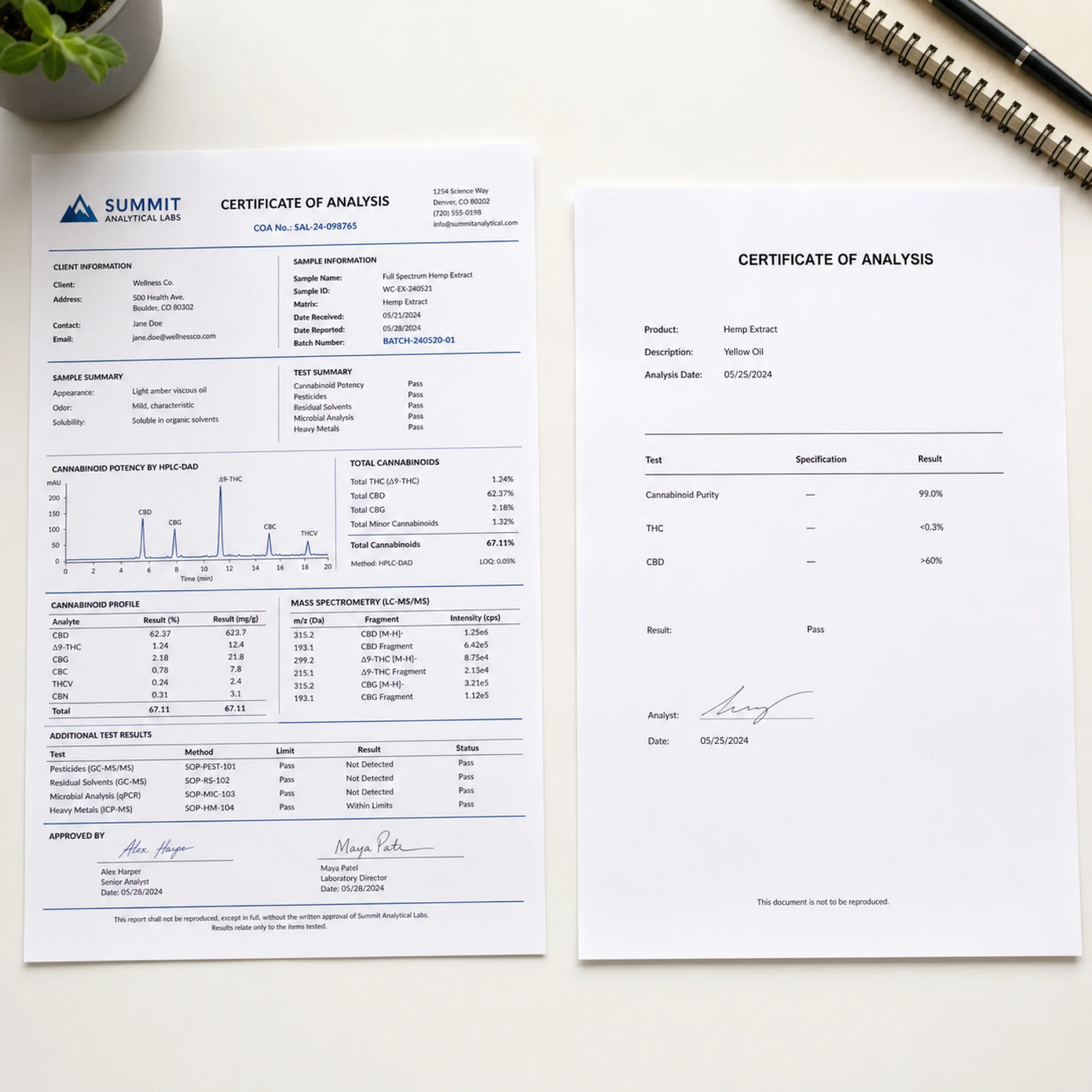
A Certificate of Analysis is the single most important document in the research peptide supply chain. As HonestPeptide's verification guide puts it, a COA "transforms an unlabeled white powder into a scientifically verifiable reagent."
A legitimate COA must answer four questions: Is this the right peptide? How pure is it? What batch did it come from? Who tested it?
What a credible COA looks like:
- Third-party testing from an independent, accredited laboratory (not in-house)
- Unique batch or lot number that matches the label on your vial
- HPLC purity percentage with a chromatogram showing the analysis
- Mass spectrometry confirmation of molecular weight
- Testing date within the last 6-12 months
What should make you suspicious:
- No COA available at all ("we'll send it on request" and never do)
- Generic COA with no batch number or a batch number that doesn't match your vial
- In-house testing only (vendor testing their own product is not independent verification)
- COA from a lab that does not exist or has no verifiable website
- Years-old testing dates that do not match current inventory
Serious verification requires checking the COA against the third-party tester's website. Unscrupulous suppliers photoshop results routinely. If the vendor claims Janoshik testing, verify the report ID directly on Janoshik's site.
One critical nuance: a research-grade COA is not a sterility certificate, not a pyrogen (endotoxin) certificate, and not a GMP batch record. It tells you about purity and identity. It tells you nothing about whether the product is safe to inject.
Red Flag 2: Pricing That's Too Good to Be True
Peptide synthesis has a cost floor. Solid-phase peptide synthesis (SPPS), HPLC purification, lyophilization, quality testing, and packaging under any kind of quality standard costs real money. When a vendor undercuts the market by 60-70%, something has been removed from the process.
As Bachem's quality control reference documents, proper peptide analysis requires identity confirmation (mass spectrometry, amino acid analysis), purity assessment (HPLC at 210-220nm), and content determination (net peptide content, water content, counterion quantification). Each test has a cost. Skip them and the sticker price drops accordingly.
| Peptide | Suspicious Price | Typical Market Range | What's Probably Missing |
|---|---|---|---|
| BPC-157 (5mg) | Under $15 | $35-80 | Purification, third-party testing, sterility |
| Semaglutide (4-week) | Under $50 | $100-250 (compounded) | Pharmaceutical-grade API, proper storage chain |
| TB-500 (5mg) | Under $15 | $40-90 | Full sequence verification, endotoxin testing |
| CJC-1295/Ipamorelin kit | Under $30 | $80-200 | Independent batch COA, proper lyophilization |
The cheapest vendor is almost never the best value. A $15 vial with 38% actual peptide content delivers less active ingredient than a $50 vial with 98% purity. You pay less per vial and more per microgram of what you actually wanted.
Red Flag 3: Therapeutic Claims on a "Research Chemical"
A research chemical vendor that writes "for fat loss," "heals tendons," or "reverses aging" on product listings has made a regulatory error that reveals either ignorance or disregard for the rules. Either one should concern you.
Research peptides are legally sold under an exemption that requires them to be marketed exclusively for laboratory research. The moment a vendor makes a therapeutic claim, they have reclassified their product as an unapproved drug under Section 505(a) of the FD&C Act. The "research use only" disclaimer no longer applies.
The FDA has used exactly this logic in its March 2026 warning letters to vendors like Gram Peptides and Prime Sciences. If a vendor is making therapeutic claims, the FDA considers their "research only" defense already broken. That vendor is operating on borrowed time.
Red Flag 4: Crypto-Only Payment and Unmarked Shipping
Legitimate businesses can get merchant accounts. Credit card processors (Visa, Mastercard, Stripe, Square) perform due diligence on the businesses they serve. A vendor that cannot secure credit card processing either sells products that payment processors consider too risky or has been dropped by processors for chargebacks and fraud complaints.
Cryptocurrency-only payment removes the buyer's ability to dispute a charge. If your vial arrives underdosed, contaminated, or not at all, there is no chargeback mechanism. Wire transfers carry the same risk.
Shipping tells a similar story. Legitimate supplement and research chemical companies ship in branded packaging with return addresses and tracking numbers. Vendors shipping in plain envelopes with no return address from countries with weak pharmaceutical regulation are making their supply chain intentionally opaque. That opacity does not benefit the buyer.
Red Flag 5: No Batch Numbers, No Lot Tracking, No Accountability
Every vial should carry a batch or lot number. This number links the physical product in your hands to a specific production run, a specific set of raw materials, and a specific COA. Without it, there is no traceability.
Traceability matters when something goes wrong. If a batch is contaminated, a batch number allows the vendor (or a regulator) to identify every unit from that production run and issue a recall. If your vial has no batch number, and you develop an injection-site infection or an adverse reaction, there is no way to trace the problem back to its source, alert other buyers, or determine what you were exposed to.
Pharmaceutical-grade manufacturers like Bachem assign batch numbers to every production run and maintain complete batch records from synthesis through shipment. Research-grade vendors operating at any reasonable quality standard do the same. Vendors who ship vials without batch numbers have no quality system worth the name.
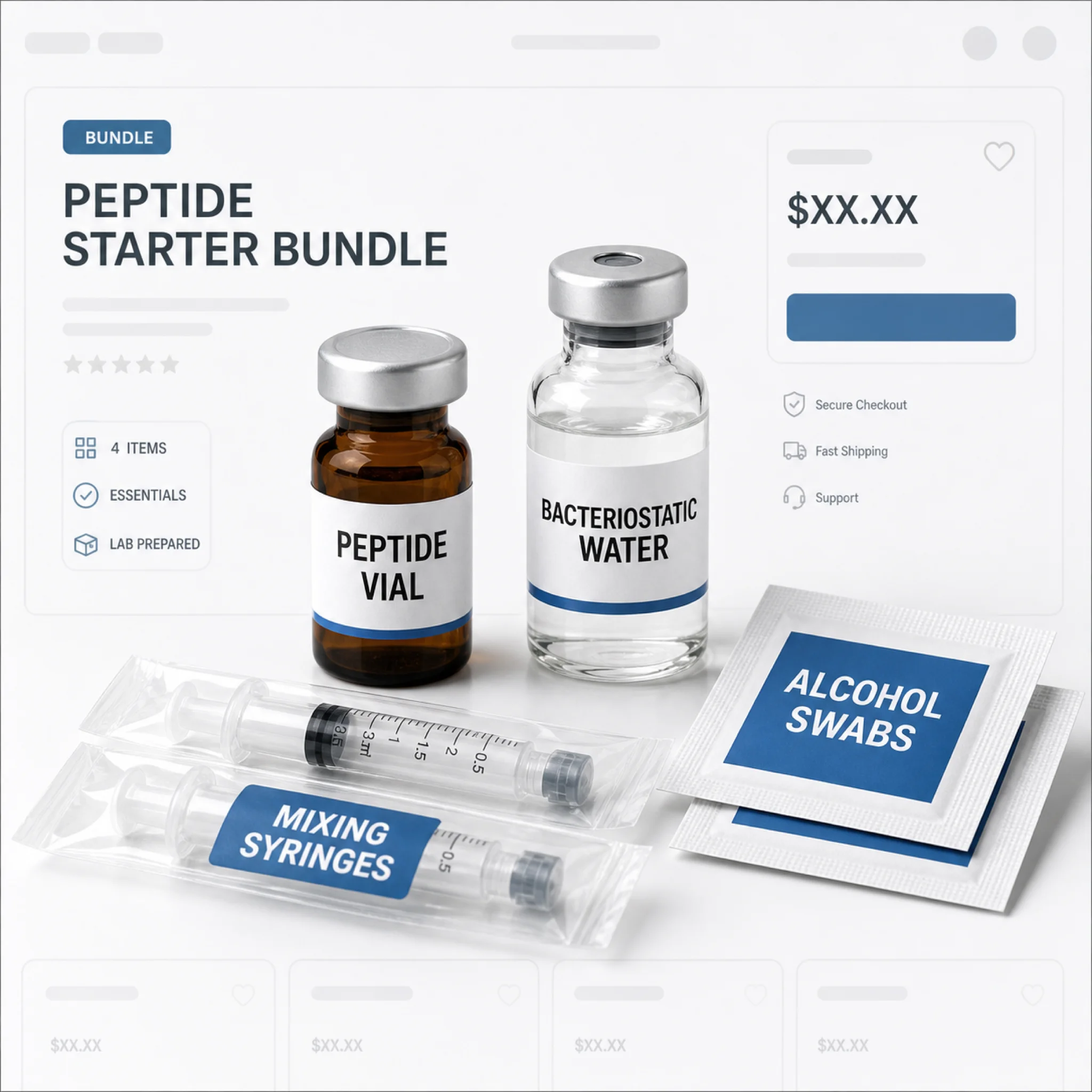
Red Flag 6: Bacteriostatic Water Bundles With "Research Use Only" Labels
This is the red flag the FDA has explicitly weaponized.
In seven warning letters issued March 31, 2026 (to Gram Peptides, Prime Sciences, Mile High Compounds, and others), the FDA laid out a clean legal argument: when a vendor sells peptides labeled "research use only" alongside bacteriostatic water for injection, the pairing constitutes evidence that the products are intended for human use. The "research only" disclaimer is treated as a sham.
Bacteriostatic water has one purpose: reconstituting lyophilized drugs for injection. A laboratory researcher doing cell culture work uses sterile water or buffer solutions, not bacteriostatic water with benzyl alcohol preservative. The preservative is there specifically because the solution is meant to sit in a multi-use vial that a human draws from repeatedly.
If the vendor you are considering sells peptide kits bundled with bacteriostatic water, mixing needles, and insulin syringes while simultaneously claiming the products are "not for human consumption," they are running the exact playbook the FDA is now targeting with enforcement actions.
Red Flag 7: No Verifiable Track Record or Only Telegram Support
A vendor's reputation trail tells you more than their marketing copy. Legitimate vendors accumulate verifiable reviews across multiple independent platforms (Reddit peptide communities, Trustpilot, independent review sites). They have domain histories older than six months. They respond to customer inquiries through email or live chat with identifiable humans, not anonymous Telegram accounts.
Pepdex's vendor verification guide identifies two specific variants of this red flag: sketchy domain history (check WHOIS registration date and whether the domain has been frequently transferred) and Telegram-only customer support (designed to avoid accountability and paper trails).
The combination of a new domain, no organic reviews, aggressive social media marketing, and encrypted-messaging-only support follows a pattern: pop-up operations that sell until complaints accumulate, then rebrand under a new name. The peptide is the same mystery powder either way. The brand name changes every 6-12 months.
Before ordering from any vendor, search their name on Reddit (r/Peptides, r/ResearchChemicals), check Trustpilot or similar review aggregators, and look at the WHOIS data for their domain. If the company has existed for less than a year and has no verifiable independent reviews, you are their product test subject, not their customer.
Frequently Asked Questions
Is it safe to buy peptides from overseas vendors?
The risk depends on the specific vendor, not the country. Some overseas manufacturers produce high-quality research peptides. But importing peptide APIs faces increased FDA scrutiny (Import Alert 66-80 updated March 2026), and overseas purchases eliminate your ability to verify the manufacturing facility, dispute charges, or seek recourse if the product is contaminated. The further the vendor is from any regulatory jurisdiction you can access, the higher the risk.
Can I get my peptides tested independently?
Yes. Independent labs like Janoshik Analytical offer HPLC purity testing ($50-100 per sample), mass spectrometry identity confirmation, and in some cases endotoxin screening. Send a sample before using a new vendor or a new batch. The cost of a single test is far less than the cost of injecting contaminated or substituted material.
What does "99% purity" actually mean on a peptide COA?
It typically means HPLC purity: 99% of the UV-absorbing material in the sample is the target peptide. It does not mean the vial is 99% peptide by weight (counterions and moisture reduce net peptide content, often to 60-80%). It says nothing about sterility, endotoxins, heavy metals, or residual solvents. A COA that shows only HPLC purity is an incomplete picture of quality.
Should I stop buying from a vendor that just received an FDA warning letter?
An FDA warning letter means the agency has documented specific legal violations and given the vendor a deadline to correct them. The vendor may comply, shut down, or continue operating and face escalated enforcement. At minimum, a warning letter tells you the FDA considers the vendor's products to be unapproved drugs sold in violation of federal law. That is useful information for your risk assessment.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.