What Is BPC-157 and Where Does It Come From?
Your stomach produces a protein called Body Protection Compound. BPC-157 is a synthetic fragment of that protein -- specifically, a chain of 15 amino acids (GEPPPGKPADDAGLV) with a molecular weight of 1,419 daltons. Croatian researcher Predrag Sikiric first isolated and described it in 1993, and his lab at the University of Zagreb has driven much of the research since.
Most peptides dissolve in stomach acid within minutes. BPC-157 doesn't. It resists gastric enzyme degradation for over 24 hours, which means it remains active when taken orally. That's unusual. Insulin and growth hormone require injection because they can't survive the stomach. BPC-157 can.
The peptide occurs naturally in human gastric juice at very low concentrations. Its endogenous role appears to involve maintaining the integrity of the stomach lining and supporting mucosal homeostasis. Researchers noticed early on that this gut-protective activity seemed to extend far beyond the stomach, which is what sparked three decades of investigation into its effects on tendons, muscles, bones, blood vessels, and the nervous system.
If you're exploring the broader world of therapeutic peptides, our guide to peptides for health including BPC-157 and thymosin beta-4 covers the legal and practical landscape.
How BPC-157 Works at the Cellular Level
BPC-157 doesn't act through a single receptor or pathway. Instead, it triggers a cascade of overlapping biological signals, which partly explains why its effects appear across so many different tissue types. A 2025 systematic review in HSS Journal mapped these pathways by analyzing 21 preclinical studies on mechanism of action.
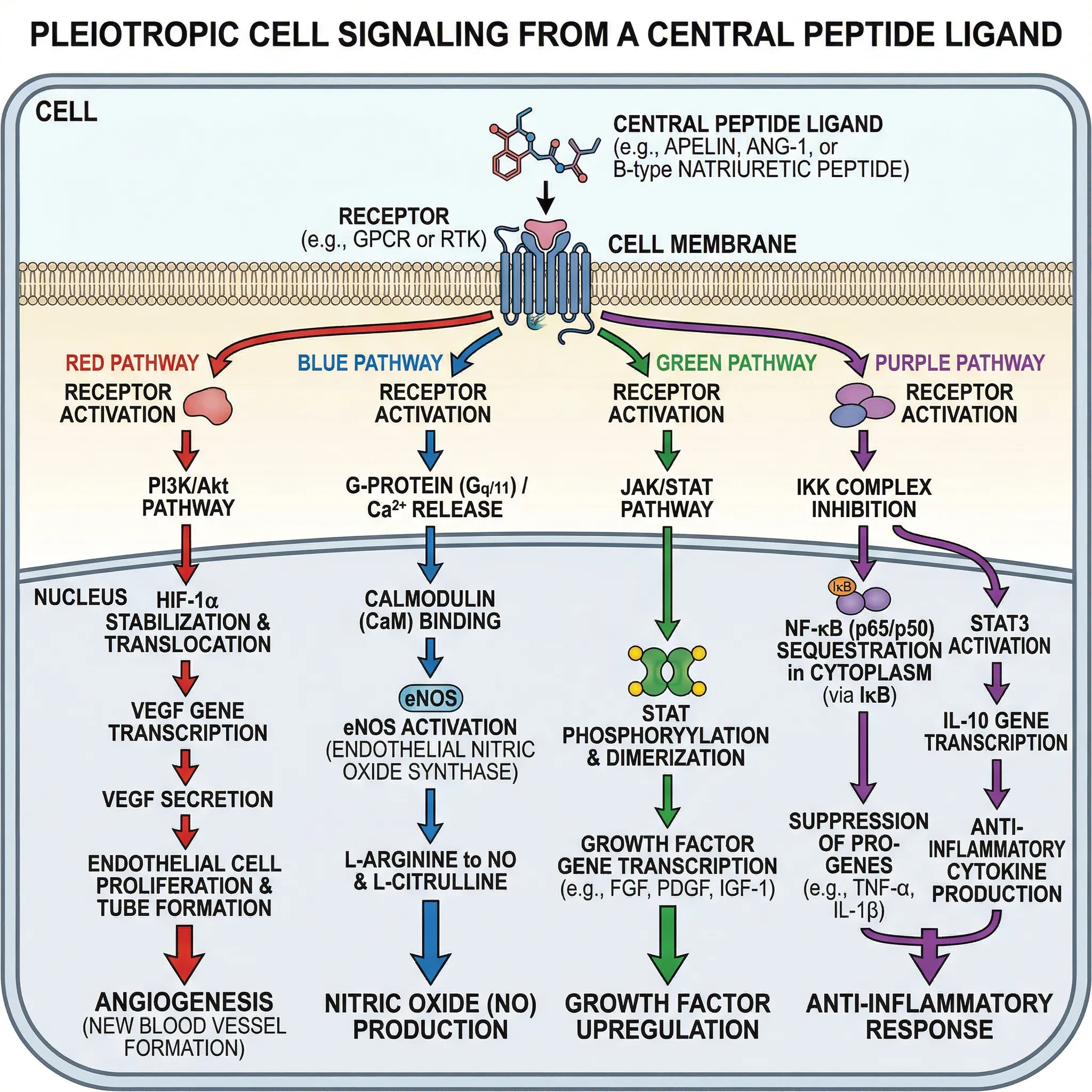
The four main pathways are:
| Pathway | What it does | Why it matters for healing |
|---|---|---|
| VEGF / VEGFR2 | Stimulates new blood vessel formation | Brings oxygen and nutrients to injured tissue, especially tendons and ligaments with poor blood supply |
| Nitric oxide (Akt-eNOS) | Increases NO production and vessel dilation | Improves blood flow, reduces oxidative stress, supports endothelial cell survival |
| ERK1/2 signaling | Promotes cell proliferation and migration | Drives fibroblast activity needed for tissue repair and collagen synthesis |
| Anti-inflammatory | Reduces TNF-alpha, IL-6, COX-2 | Limits tissue damage from excessive inflammation after injury |
One finding that keeps appearing across studies: BPC-157 upregulates growth hormone receptor expression in tendon fibroblasts. A 2014 Chang et al. study found that treating tendon cells with BPC-157 increased growth hormone receptor expression up to sevenfold within three days. This amplified the cells' response to circulating growth hormone, boosting proliferation through the JAK2-STAT signaling pathway. In practical terms, BPC-157 appears to make tendon cells more receptive to the repair signals your body already produces.
BPC-157 also shifts macrophage activity from the pro-inflammatory M1 phenotype toward the reparative M2 phenotype. M1 macrophages are the aggressive first responders -- useful early, destructive if they linger. M2 macrophages handle cleanup and lay the scaffolding for new tissue. BPC-157 appears to accelerate this transition, which may explain why it reduces fibrosis and promotes cleaner healing in animal models.
Then there's the duration question. BPC-157's plasma half-life is under 30 minutes, yet its biological effects appear to persist for weeks to months. In one spinal cord injury study, functional improvements lasted 360 days after a single treatment. The working theory is that BPC-157 activates gene expression programs -- flipping molecular switches that initiate self-sustaining repair cascades -- rather than working through continuous receptor occupancy.
Gut Repair: The Original Use Case
BPC-157's story begins in the gut, and that remains where some of the most compelling preclinical data lives. The peptide was first studied as a gastroprotective agent -- something that shields stomach and intestinal lining from damage. If you've dealt with leaky gut or intestinal permeability issues, the mechanism is relevant.
In rat models, BPC-157 has consistently protected against gastric lesions induced by NSAIDs (ibuprofen, diclofenac, celecoxib), alcohol, and stress. One study found that BPC-157 rescued NSAID-induced cytotoxicity by stabilizing intestinal permeability and enhancing cytoprotective pathways. It maintained tight junction integrity -- the seals between intestinal cells that prevent undigested proteins and bacteria from leaking into the bloodstream.
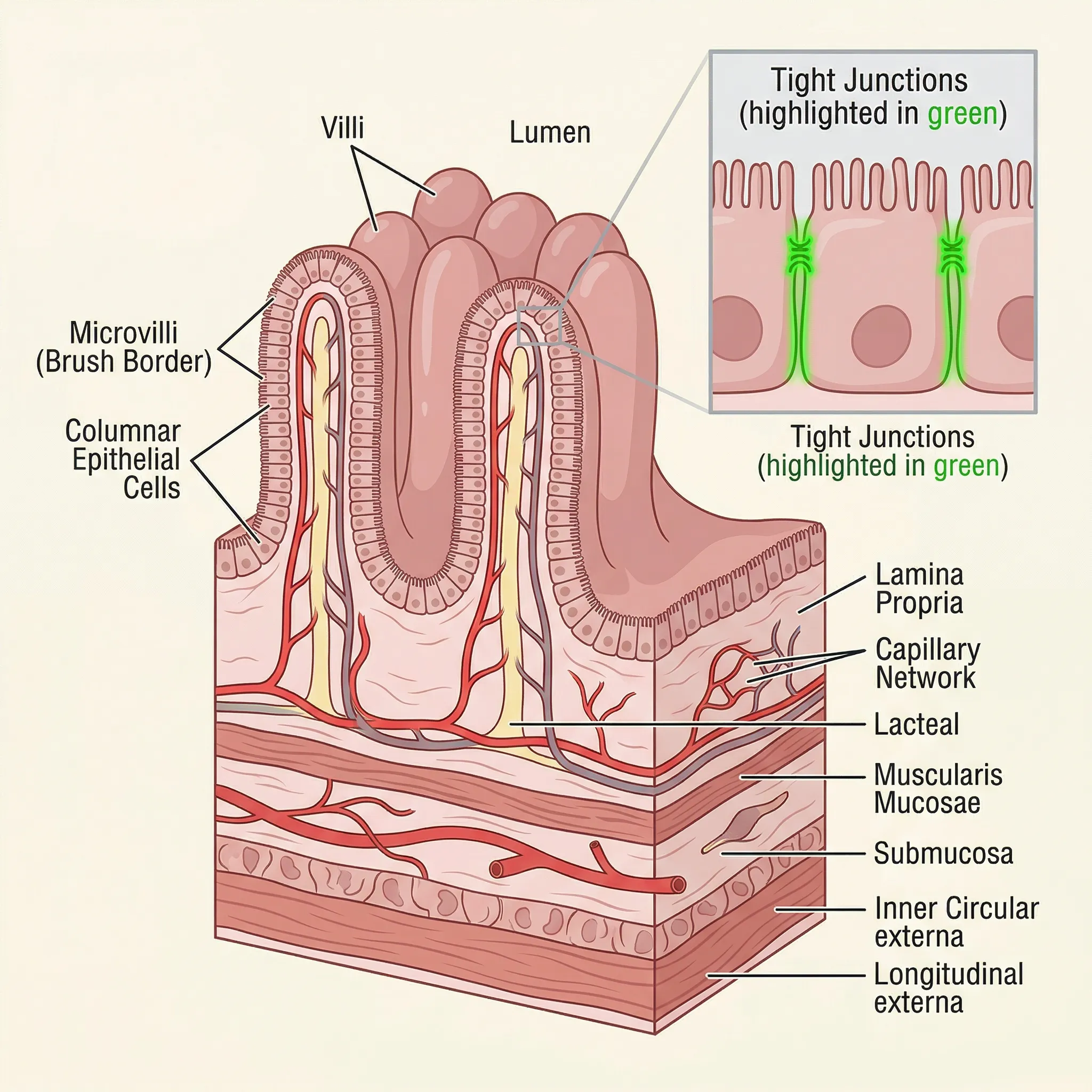
The peptide was previously tested in human clinical trials for ulcerative colitis, and more recently for multiple sclerosis -- both conditions with inflammatory components affecting tissue integrity. The specific trial results haven't been published in full, which is a gap worth noting. What we do know from the preclinical work is that BPC-157 counteracted colitis in multiple animal models, reduced inflammatory markers, and promoted mucosal regeneration.
BPC-157 also interacts with the gut-brain axis. Research published in Neural Regeneration Research showed that the peptide modulates dopamine and serotonin pathways, and counteracted brain damage following stroke in rats when the peptide was administered during reperfusion. This bidirectional relationship between gut health and brain function is part of why BPC-157 researchers describe it as "pleiotropic" -- it influences multiple organ systems simultaneously.
We've covered the gut-brain axis in detail -- the bidirectional communication between your microbiome and your brain that BPC-157 appears to modulate.
Tendons, Muscles, and Bones: The Sports Medicine Evidence
Athletes and clinicians started using BPC-157 for musculoskeletal recovery years ago, despite having almost no human clinical data to justify it. The animal data, to be fair, is extensive.
A 2025 systematic review published in HSS Journal analyzed 36 studies (35 preclinical, 1 clinical) on BPC-157 in orthopaedic sports medicine. The results across tissue types:
| Tissue type | Key findings | Study count |
|---|---|---|
| Achilles tendon | Improved structural, functional, and biomechanical indices after transection; reduced inflammatory infiltrates; promoted tendon-to-bone healing | 4 studies |
| Quadriceps tendon | Resolved myotendinous junction defects, restored motor function | 2 studies |
| Muscle (crush/transection) | Improved load to failure, reduced atrophy, restored muscle fiber diameters | 3 studies |
| MCL (knee ligament) | Reduced valgus instability and contracture, restored biomechanics at 90 days | 1 study |
| Bone (nonunion fracture) | Performed similarly to autologous bone marrow injection in resolving nonunion defects | 1 study |
One finding stands out: BPC-157 maintained its healing effects even in the presence of corticosteroids, which normally impair tissue repair. Krivic et al. (2006) showed that BPC-157 promoted tendon-to-bone healing in rats with Achilles detachment and actively opposed the aggravating effects of corticosteroid treatment. For anyone who's been told to use cortisone injections for a tendon injury, that's a provocative result -- though, again, it's animal data.
The single human study available is a 2021 retrospective review by Lee and Padgett. They followed 16 patients who received intraarticular knee injections containing BPC-157 (either alone or combined with thymosin beta-4). Fourteen of 16 patients (87.5%) reported significant pain relief. For those who received BPC-157 alone, 7 of 12 reported symptom improvement lasting over six months after a single injection of 2 cc at 2000 mcg/mL concentration.
A second human study from 2024 tested intravesical BPC-157 injections in 12 patients with interstitial cystitis who had not responded to standard treatment. All 12 reported significant improvement, with 80-100% symptom resolution at 6 weeks.
The evidence gap in numbers: Over 500 published papers on BPC-157 since 1993, yet only three published human studies -- two small pilot studies and one retrospective review. No randomized controlled trials. No phase II or III clinical trials with published results. The FDA registered one phase I trial in 2016, but its status is listed as unknown.
Dosage Protocols: What Studies Actually Used
There is no established human dosage for BPC-157. No regulatory body has approved a dose, and the handful of human studies used different routes and concentrations. What follows is a summary of what researchers have used -- not a recommendation.

| Setting | Dose | Route | Duration |
|---|---|---|---|
| Rat studies (typical) | 10 ng/kg to 10 mcg/kg | IP, oral (drinking water), IM, topical | Single dose to 90 days daily |
| Safety study (rat/dog) | 6 mcg/kg to 20 mg/kg | IM, IV | Single to 6 weeks |
| Human knee injection | 4,000 mcg (2 cc of 2,000 mcg/mL) | Intraarticular | Single injection |
| Human IV safety study | Up to 20 mg (20,000 mcg) | Intravenous | Single infusion |
| Human-equivalent oral estimate | ~110-180 mcg (based on allometric scaling) | Oral | Not established |
The oral human-equivalent dose comes from Examine.com's analysis of rat studies where oral administration at 10 mcg/kg was effective. Using standard allometric scaling (rat to human), that translates to roughly 1.6 mcg/kg of body weight, or 110 mcg for a 150-pound person and 180 mcg for a 250-pound person. No human pharmacokinetic studies have validated this conversion.
Most animal studies dosed BPC-157 once daily. The effective range in animals was remarkably wide -- nanogram to microgram per kilogram doses both showed benefit, which is unusual for pharmacologically active compounds. In the 2025 IV safety study by Lee and Burgess, two healthy adults received up to 20 mg intravenously with no adverse events and plasma levels returning to baseline within 24 hours.
BPC-157 is metabolized primarily in the liver via cytochrome P450 pathways and excreted by the kidneys. Metabolites remain detectable in urine for 4-5 days using mass spectrometry -- relevant for athletes, since WADA-accredited labs can detect it well within their testing windows.
Safety Profile and Regulatory Status
The preclinical safety data on BPC-157 is remarkably clean -- almost suspiciously so. Three dedicated safety studies in animal models found no adverse effects across a wide range of doses and administration routes.
Xu et al. (2020) performed the most comprehensive evaluation: single and repeat intramuscular and intravenous doses ranging from 6 mcg/kg to 20 mg/kg over 6 weeks in both rats and dogs. The results:
- No gross necropsy abnormalities in liver, spleen, thymus, or gastric wall
- No microscopic histopathological changes in liver, spleen, lung, kidney, brain, thymus, prostate, or ovaries
- No acute local irritation at injection sites
- No mutagenic or genotoxic effects (Ames test, chromosomal aberration assays, micronucleus assays)
- No teratogenic effects in pregnant rats (doses 0.2-4 mg/kg between days 6-15 of pregnancy)
- No lethal or toxic dose identified at any level tested
In the three human studies published so far, no adverse effects were reported. The 2025 Lee and Burgess pilot study -- the only one designed specifically to assess safety -- found that IV infusions up to 20 mg produced no clinically meaningful changes in vital signs, ECG, or blood biomarkers for cardiac, hepatic, renal, thyroid, or metabolic function.
That said, the regulatory picture tells a different story about confidence levels:
| Agency | Position | Practical meaning |
|---|---|---|
| U.S. FDA | Category 2 bulk drug substance (2023) | Cannot be legally compounded; not approved for human use; "may pose risk of immunogenicity" |
| WADA | Banned under S0 Unapproved Substances (2022) | Prohibited at all times, in and out of competition |
| NFL, MLB, NCAA, PGA | Non-specific bans on peptide hormones | BPC-157 falls under broader PED prohibitions |
| UFC/USADA | Specific ban via WADA list | Fighters who test positive face sanctions |
The FDA's concern isn't that BPC-157 has been shown to be dangerous. Their concern is that it hasn't been shown to be safe in humans through the standard regulatory process. There's also the issue of product quality. Since BPC-157 products are sold as "research chemicals" and not as drugs or supplements, they aren't subject to manufacturing standards. Studies have found that between 12% and 58% of ergogenic supplements are contaminated with undisclosed substances -- and BPC-157 products purchased online face the same risk.
Anonymous user reports online describe side effects including injection site pain, joint pain, anxiety, heart palpitations, insomnia, fatigue, and mood changes. These could reflect the peptide's known ability to modulate dopamine and serotonin systems, contamination in unregulated products, or both.
Our article on chronic inflammation and disease covers why anti-inflammatory compounds keep drawing research attention.
Myth vs. Fact: What the Evidence Supports
| Claim | Reality |
|---|---|
| "BPC-157 is proven to heal tendons in humans" | Preclinical data in rats is strong and consistent. Human evidence is limited to a single retrospective study of 12 patients with knee pain. No controlled human trials on tendon healing have been published. |
| "It's completely safe with no side effects" | Animal safety studies show no toxicity across wide dose ranges. Three small human studies reported no adverse events. But long-term human safety data doesn't exist, and the FDA has flagged potential immunogenicity concerns. |
| "It's illegal to use BPC-157" | In the U.S., BPC-157 is not a DEA-scheduled substance, so possession isn't illegal. However, selling it for human use violates FDA regulations. WADA-tested athletes face sanctions for use. |
| "Oral BPC-157 is useless -- you have to inject it" | BPC-157 is stable in gastric juice (unlike most peptides), and oral administration showed benefit in rat studies for GI conditions. Injection was used for localized musculoskeletal effects. Both routes appear active in animal models. |
| "BPC-157 could cause cancer through angiogenesis" | A reasonable theoretical concern. BPC-157 promotes VEGF-driven blood vessel growth, which tumors exploit. However, multiple studies show BPC-157 actually inhibited uncontrolled cell proliferation, downregulated VEGF expression in tumor models, and suppressed Ki-67 (a proliferation marker). The peptide appears to selectively modulate angiogenesis depending on tissue context. |
| "It replaces the need for surgery or physical therapy" | No evidence supports this. Even the most optimistic interpretation of animal data positions BPC-157 as a potential adjunct to standard care, not a replacement. |
Frequently Asked Questions
Is BPC-157 FDA-approved for any condition?
No. BPC-157 has no FDA-approved indication for any condition. In 2023, the FDA classified it as a Category 2 bulk drug substance, meaning it cannot legally be used in compounded medications. Products sold online as BPC-157 are marketed as "research chemicals" not intended for human consumption, though that labeling is often misleading.
Can you take BPC-157 orally or does it need to be injected?
BPC-157 is unusual among peptides because it resists degradation by stomach acid, remaining stable in gastric juice for over 24 hours. Animal studies show oral administration is effective, particularly for gastrointestinal conditions. For musculoskeletal injuries, most animal studies used injection (intraperitoneal, intramuscular, or subcutaneous) to deliver the peptide closer to the injury site. Both routes appear biologically active, but no human comparative studies exist.
How long does BPC-157 stay in your system?
The plasma half-life of BPC-157 is under 30 minutes. It is metabolized in the liver and excreted through the kidneys. Metabolites are detectable in urine for 4-5 days using high-resolution mass spectrometry. Despite the short half-life, biological effects in animal studies persist for weeks to months, suggesting the peptide triggers lasting cellular changes rather than requiring continuous presence.
Is BPC-157 banned in sports?
Yes. WADA specifically banned BPC-157 in 2022 under the S0 Unapproved Substances category. This prohibition applies at all times -- in and out of competition. Major U.S. sports leagues (NFL, MLB, NCAA, PGA, UFC) also prohibit it, either by name or under broader peptide hormone bans. USADA testing can detect BPC-157 metabolites in urine for up to 5 days.
What are the risks of using BPC-157 from online sources?
The primary risk is product quality. BPC-157 sold online is unregulated for purity, potency, and sterility. Research has found that 12-58% of ergogenic supplements contain undisclosed contaminants. Without pharmaceutical-grade manufacturing standards, products may contain incorrect doses, degraded peptide, or harmful impurities. There is no way for consumers to verify what they're actually receiving.
Related Articles
- Peptides for Health: BPC-157, Thymosin Beta-4, and What's Legal -- A broader overview of therapeutic peptides and their regulatory status.
- Leaky Gut Syndrome: Symptoms, Causes, and Evidence-Based Healing -- Understanding the intestinal permeability issues that BPC-157 was originally studied to address.
- Inflammation and Chronic Disease: Anti-Inflammatory Living -- The inflammatory pathways that BPC-157 modulates are central to chronic disease.
- Bone Broth Benefits: Gut Healing, Joints, and Skin -- Natural approaches to supporting gut and joint health.
- Autophagy: Fasting, Exercise, and Diet for Cellular Repair -- Another pathway for cellular repair and recovery that complements peptide research.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.