Your Bedtime Might Matter More Than Your Sleep Hours
You have probably heard that adults should get seven to nine hours of sleep. That number comes up in nearly every conversation about health, and it is well-supported. But there is a second variable that most people overlook entirely, one that mounting research suggests may carry its own independent cardiovascular risk: when you fall asleep.
Think about it this way. Two people both get seven and a half hours of sleep. One drifts off at 10:30 PM and wakes at 6 AM. The other falls asleep at 1 AM and sleeps until 8:30 AM. Same duration, very different timing. And according to a large-scale UK Biobank cohort study published in the European Heart Journal — Digital Health, those two people do not carry the same cardiovascular risk.
This is a shift in how researchers are thinking about sleep and heart disease. For decades, the focus sat almost exclusively on duration — too short or too long was bad, and the sweet spot was somewhere around seven to eight hours. But the American Heart Association's 2025 scientific statement formally recognized that sleep health is multidimensional. Duration is just one piece. Timing, regularity, continuity, and even subjective satisfaction all play roles in cardiometabolic outcomes.
What follows is a close look at what the current evidence says about sleep timing and your heart — not as a substitute for getting enough sleep, but as an additional piece of the cardiovascular puzzle that most people are not tracking.
What 88,000 Wristbands Revealed About Bedtime and Heart Risk
The most striking data on sleep timing and cardiovascular disease comes from a 2021 study led by Shahram Nikbakhtian and colleagues at the University of Exeter. They analyzed accelerometer data from 88,026 UK Biobank participants who wore wrist-mounted devices for seven continuous days, giving researchers objective measurements of when people actually fell asleep — not self-reported estimates, which tend to be unreliable.
Over an average follow-up of 5.7 years, 3,172 participants developed cardiovascular disease, including heart attacks, heart failure, stroke, and chronic ischemic heart disease. The researchers then looked at whether the time participants fell asleep predicted who would develop these conditions.
Key finding: Falling asleep between 10:00 and 10:59 PM was associated with the lowest cardiovascular disease incidence. Every other timing window carried measurably higher risk.
The numbers are worth examining closely. Compared to the 10:00–10:59 PM reference group, falling asleep at midnight or later carried a 25% higher risk of developing cardiovascular disease (hazard ratio 1.25). Falling asleep between 11:00 and 11:59 PM carried a 12% increased risk (HR 1.12). And perhaps most surprisingly, falling asleep before 10:00 PM was associated with a 24% higher risk (HR 1.24).
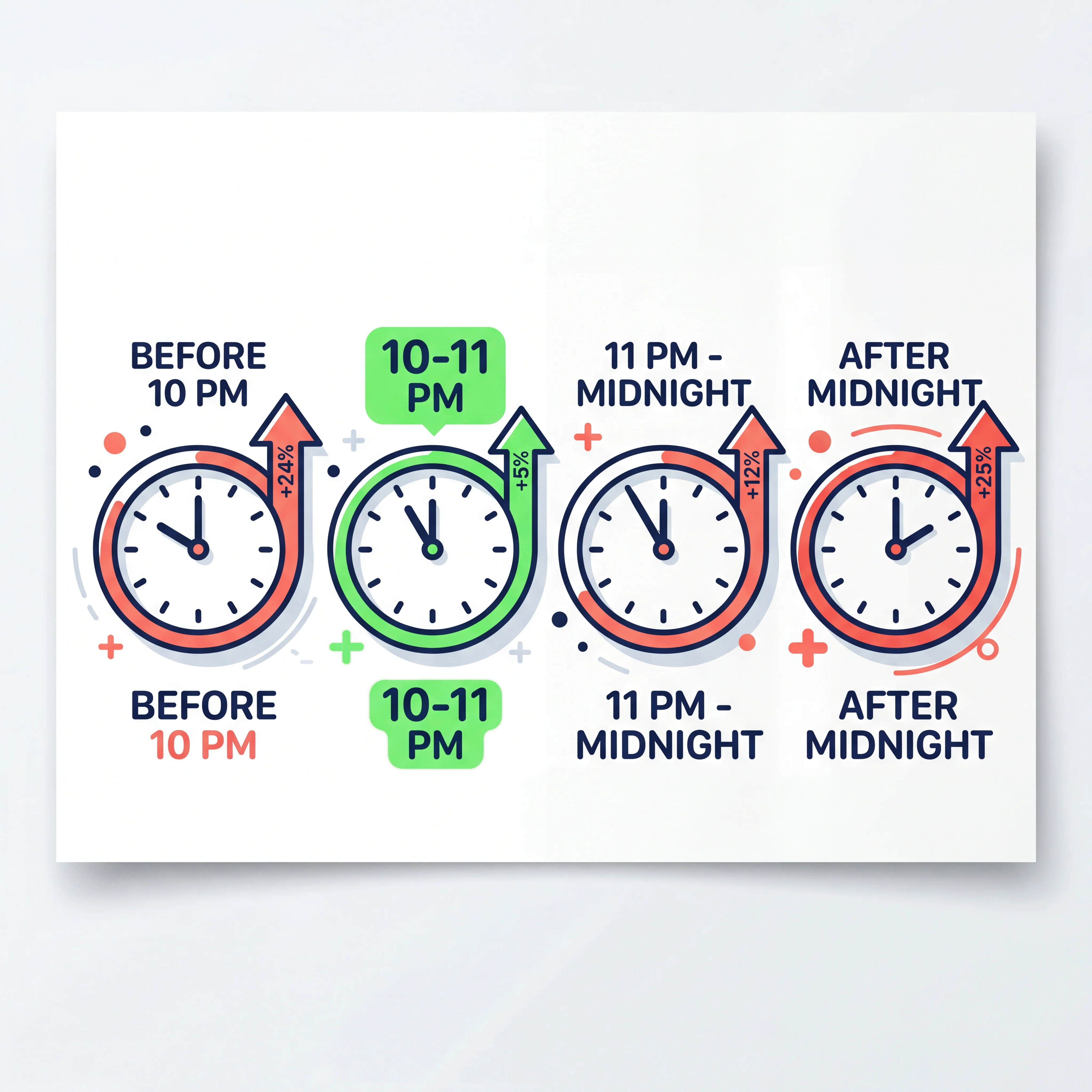
This U-shaped pattern — where both early and late bedtimes elevated risk — held up even after the researchers controlled for sleep duration, sleep irregularity, BMI, diabetes, hypertension, smoking status, chronotype, blood pressure, and cholesterol. In other words, the association between sleep onset timing and heart disease was independent of these established risk factors.
| Sleep Onset Time | Hazard Ratio (Fully Adjusted) | Statistical Significance |
|---|---|---|
| Before 10:00 PM | 1.24 (95% CI: 1.10–1.39) | P < 0.005 |
| 10:00–10:59 PM (reference) | 1.00 | — |
| 11:00–11:59 PM | 1.12 (95% CI: 1.01–1.25) | P = 0.04 |
| Midnight or later | 1.25 (95% CI: 1.02–1.52) | P = 0.03 |
One detail that stood out was a sex-based difference. The association between sleep timing and cardiovascular risk was substantially stronger in women. For women who fell asleep at midnight or later, the hazard ratio jumped to 1.63 — a 63% increased risk. For men, only falling asleep before 10 PM reached statistical significance (HR 1.17). The reasons for this difference remain unclear, though the researchers speculated it may relate to how the endocrine system responds to circadian disruption, or to the older age profile of the cohort (since cardiovascular risk rises sharply for women after menopause).
Your Heart Runs on a Clock, and It Does Not Forgive Disruption
To understand why bedtime matters independent of sleep duration, you need to understand that your heart is not just a pump that operates the same way around the clock. It has its own circadian machinery, and that machinery expects certain things to happen at certain times.
Your body's master clock sits in the suprachiasmatic nucleus (SCN) of the hypothalamus. It coordinates 24-hour rhythms across virtually every organ system by responding to light exposure and synchronizing downstream clocks. But here is the part most people do not realize: your heart has its own local circadian clocks, operating in the cardiomyocytes themselves, driven by the same core clock genes (BMAL1, CLOCK, NPAS2) found throughout the body.
Research published in Nature Communications demonstrated that these cardiac clocks directly govern daily rhythms in the sinoatrial (SA) and atrioventricular (AV) nodes — the structures that control your heart's electrical timing. The study found that the autonomic nervous system and the cardiomyocyte clock contribute to SA and AV node function in different proportions, which means the cardiac conduction system is inherently sensitive to decoupling when your behavioral routine shifts abruptly.
This is not abstract biology. It has direct clinical consequences. Fatal cardiac arrhythmias, including ventricular fibrillation and sudden cardiac death, are more likely to occur during specific windows of the day, particularly in the early morning hours between 6 AM and noon. When your sleep-wake timing is misaligned with your internal clock, you disrupt the carefully orchestrated rhythm that keeps your heart's electrical system stable.
During normal sleep, your blood pressure drops by 10 to 20% — a pattern called nocturnal dipping that gives your cardiovascular system a critical recovery period. As the CDC notes, when sleep is disrupted or poorly timed, blood pressure stays elevated for longer stretches, and that sustained pressure accelerates damage to blood vessel walls over time.
Shift Work, Social Jetlag, and the Cardiovascular Toll
If sleep timing matters, then people who routinely sleep at the wrong time should show elevated cardiovascular risk. And they do — the evidence from shift workers is sobering.
A comprehensive review in Trends in Endocrinology and Metabolism documented that shift work is associated with increased risk of hypertension, ischemic stroke, coronary heart disease, and sudden cardiac death. The mechanism goes beyond simple sleep deprivation. When people work through the night and sleep during the day, their internal circadian clock stays anchored to the light-dark cycle while their behavior runs on an opposite schedule. This mismatch — termed circadian misalignment — triggers a cascade of metabolic changes.

Experimental studies have shown that even short-term circadian misalignment (forcing healthy people onto shifted schedules in a lab setting) produces measurable increases in inflammatory markers, blood pressure, and insulin resistance. Social jetlag — the discrepancy between your sleep schedule on workdays versus weekends — produces a milder version of the same effect. According to the AHA's scientific statement, social jetlag has been associated with a 20% higher risk of overweight or obesity, lower HDL cholesterol, higher triglycerides, and decreased insulin sensitivity.
Even daylight saving time transitions — a one-hour shift that most people barely notice — have been associated with a measurable uptick in myocardial infarction and ischemic stroke hospitalizations. If a single hour can register in population-level cardiovascular event data, the implication for people who routinely shift their sleep timing by two or three hours between weekdays and weekends is worth considering.
| Type of Circadian Disruption | Cardiovascular Effects | Magnitude of Risk |
|---|---|---|
| Rotating shift work | Hypertension, coronary heart disease, stroke, sudden cardiac death | Moderate to high (dose-dependent with years of shift work) |
| Social jetlag (2+ hours) | Obesity, insulin resistance, dyslipidemia | 20% increased overweight/obesity risk |
| Daylight saving time | Myocardial infarction, ischemic stroke | Small but population-detectable |
| Late chronotype (night owl) | Higher cardiovascular mortality | Elevated vs. morning chronotype |
Chronotype itself — whether you are naturally a morning person or a night owl — adds another layer. Evening chronotypes tend to have later bedtimes and are more likely to experience circadian misalignment when forced into conventional work schedules. Research cited in the Trends in Endocrinology and Metabolism review found that evening chronotypes face higher cardiovascular mortality risk compared to morning types, even after accounting for sleep duration. The relationship between chronic stress, hormones, and cardiovascular strain likely compounds this effect, since people fighting their natural sleep-wake rhythm live in a state of ongoing physiological tension.
It's Not Just When You Sleep — It's How Consistently
Sleep timing and sleep regularity are related but distinct concepts, and both matter for cardiovascular health. You could go to bed at 10:30 PM every night — an optimal timing — but if you go to bed at 10:30 on weekdays and 2 AM on weekends, the inconsistency itself introduces risk.
The NIH reported on a study from the Multi-Ethnic Study of Atherosclerosis (MESA) that tracked nearly 2,000 adults ages 45 to 84 using actigraphy for seven days, then followed them for about five years. The results were striking: participants with the most irregular sleep schedules were nearly twice as likely to develop cardiovascular disease as those with more consistent patterns. This remained true after adjusting for sleep apnea and other established cardiovascular risk factors.
The AHA's 2025 scientific statement reinforced these findings. In large-scale studies, greater consistency in sleep-wake timing was associated with a 22% to 57% lower risk of cardiovascular death. Day-to-day variability in sleep timing was independently linked to higher risk of cardiovascular disease, high blood pressure, inflammation, and obesity.
Worth noting: In UK Biobank data, irregular sleep timing was linked to a higher risk of Type 2 diabetes even in people who got sufficient total sleep — with the highest risk among those who had both insufficient sleep and very irregular patterns.
This is where duration and timing intersect. A 2024 NHANES analysis published in the Journal of the American Heart Association examined the joint effects of sleep onset time and sleep duration in 6,696 adults. Late sleep onset combined with insufficient duration was the worst combination: men who fell asleep after midnight and slept less than seven hours had 74% higher odds of metabolic syndrome. Women reporting late onset with excessive duration had more than fivefold higher odds of hypertriglyceridemia. These associations were most pronounced in adults aged 40 to 59.

For anyone trying to improve their sleep habits, the takeaway is that consistency may matter as much as duration. Going to bed and waking up at roughly the same time every day — including weekends — appears to be one of the more protective things you can do for your cardiovascular system. This aligns with what sleep researchers at OHSU and other institutions have been finding about the relationship between circadian alignment and long-term health.
Sleep Timing Myths Versus What the Data Actually Shows
| Common Belief | What Research Actually Shows |
|---|---|
| "As long as I get 8 hours, it does not matter when" | Sleep onset timing is an independent cardiovascular risk factor. People sleeping the same number of hours but at different times carry different CVD risk (Nikbakhtian et al., 2021). |
| "Going to bed early is always better" | Falling asleep before 10 PM was associated with 24% higher CVD risk — similar to falling asleep after midnight. Both extremes carry elevated risk. |
| "Weekend sleep catch-up fixes everything" | Social jetlag (shifting sleep by 2+ hours on weekends) is associated with obesity, insulin resistance, and metabolic disruption. Regularity matters more than catch-up. |
| "Night owls just need more discipline" | Chronotype has a genetic component. Evening types forced into early schedules experience chronic circadian misalignment, which independently raises cardiovascular risk. |
| "Sleep timing only matters for shift workers" | Even 1-hour shifts (daylight saving time) produce detectable cardiovascular effects. Anyone with inconsistent bedtimes is experiencing a milder version of shift-work disruption. |
Practical Strategies to Protect Your Heart Through Better Sleep Timing
The research points toward a relatively clear set of principles. Your heart benefits when you fall asleep in the 10:00–11:00 PM window, maintain consistent timing seven days a week, and align your sleep schedule with your body's light-dark expectations. Here is how to work toward that in practice.
Anchor your wake time first. Most sleep experts recommend fixing your wake-up time rather than your bedtime. Your wake time sets the downstream cascade of cortisol, melatonin, and body temperature rhythms that determine when you will naturally feel sleepy roughly 14 to 16 hours later. If you wake at 6:30 AM consistently, your body will start winding down around 10:00–10:30 PM on its own.
Get bright light within 30 minutes of waking. Morning light exposure is the single most powerful signal for resetting and strengthening your circadian clock. This does not require a special light therapy device — walking outside for 10 to 15 minutes works. The CDC specifically recommends morning or lunchtime walks for this purpose. For anyone exploring how other lifestyle factors like regular cardiovascular exercise interact with sleep quality, morning exercise combined with light exposure creates a particularly effective circadian anchor.
Minimize social jetlag. Keep your weekend bedtime within 30 to 60 minutes of your weekday bedtime. A two-hour Friday-to-Saturday shift might feel harmless, but over months and years, the metabolic and cardiovascular costs accumulate. If you must shift your timing, do it gradually — 15 minutes at a time rather than a sudden two-hour jump.
Manage evening light exposure. Blue-enriched light from screens suppresses melatonin production and delays your circadian phase. Dim your lights and reduce screen brightness in the two hours before your target bedtime. If you work late or use devices extensively, blue-light filtering (either built-in to the device or via glasses) can help, though it is less effective than simply reducing light intensity.
Time your meals. Late-night eating can delay your circadian clock and disrupt the nocturnal blood pressure dipping that your cardiovascular system depends on. Finish your last substantial meal at least two to three hours before bed. This aligns with broader evidence on how cardiovascular-protective lifestyle practices work best when timed appropriately within the circadian cycle.
If you are a natural night owl, work with your biology, not against it. Chronotype has a genetic component and cannot simply be overridden by willpower. If your natural tendency is to fall asleep around midnight, forcing a 9 PM bedtime will likely produce fragmented, low-quality sleep — which carries its own cardiovascular risks. Instead, aim for the earliest bedtime you can sustain naturally while still falling asleep within 15 to 20 minutes. Gradually shifting bedtime earlier by 15-minute increments over weeks is more sustainable than abrupt changes.
Frequently Asked Questions
What is the best time to go to sleep for heart health?
Based on the largest study to date — an analysis of 88,026 UK Biobank participants — falling asleep between 10:00 and 10:59 PM was associated with the lowest incidence of cardiovascular disease. Both earlier (before 10 PM) and later (after 11 PM) bedtimes carried measurably higher risk, even after controlling for sleep duration and other cardiovascular risk factors.
Can sleeping too early be bad for your heart?
Yes, according to the UK Biobank data. Falling asleep before 10:00 PM was associated with a 24% higher risk of cardiovascular disease compared to the 10:00–10:59 PM window. The researchers suggest this may indicate that very early bedtimes reflect or contribute to circadian rhythm disruption, though the exact mechanism is still being studied.
Does going to bed at the same time every night really matter?
The evidence says yes. The MESA study found that people with the most irregular sleep schedules were nearly twice as likely to develop cardiovascular disease. The AHA reported that consistent sleep-wake timing was associated with a 22% to 57% lower risk of cardiovascular death. Even small variations — shifting by an hour or two on weekends — can produce measurable metabolic effects over time.
Is sleep timing more important for women than men?
The UK Biobank study found that the association between late bedtime and cardiovascular disease was substantially stronger in women. Women who fell asleep at midnight or later had a 63% higher risk (HR 1.63), while men in the same timing group did not show a statistically significant increase. Researchers are still investigating whether hormonal differences, menopausal status, or other factors explain this disparity.
Should shift workers be especially concerned about heart health?
Yes. Shift work is an established cardiovascular risk factor, associated with higher rates of hypertension, coronary heart disease, stroke, and sudden cardiac death. The risk is dose-dependent — more years of shift work correlate with greater cardiovascular risk. Shift workers should discuss cardiovascular monitoring with their doctors and focus on sleep regularity, light exposure management, and meal timing as partial countermeasures.
Related Articles
- Sauna Bathing for Heart Health: What 20 Years of Finnish Research Actually Proves — How heat exposure supports cardiovascular function and complements other heart-protective habits.
- Sleep Optimization and Circadian Rhythm Technology — A deeper look at the tools and strategies for aligning your sleep with your body's internal clock.
- The Hidden Toll of Low-Level Stress: How Chronic Tension Wrecks Your Hormones — Why ongoing stress compounds the cardiovascular effects of poor sleep timing.
- Zone 2 Cardio for Fat Burning and Longevity — How moderate cardiovascular exercise interacts with sleep quality and circadian health.
- Best Ways to Improve Your Sleep — Practical sleep hygiene strategies that work alongside optimal timing.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.