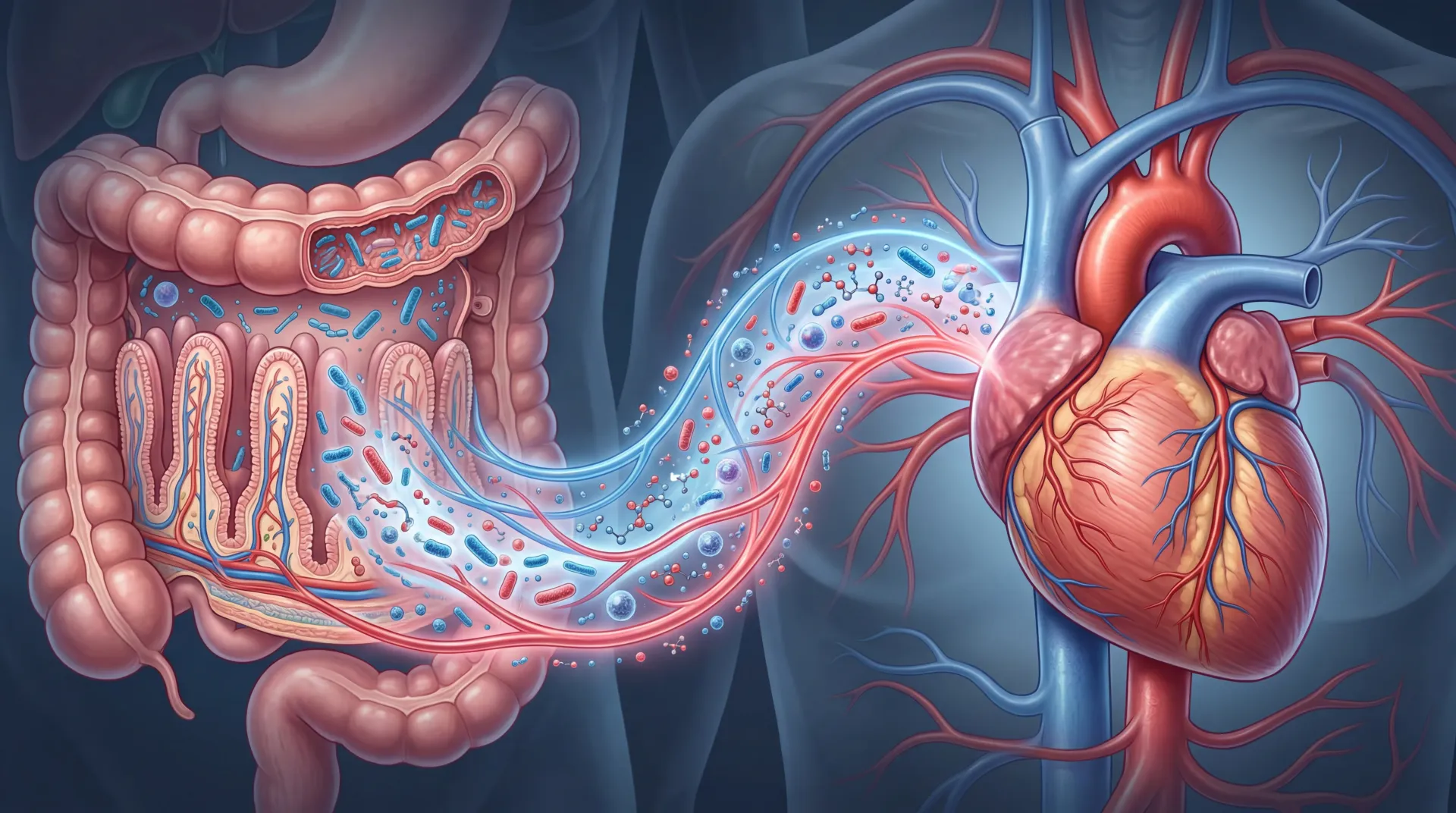
Roughly 38 trillion microorganisms live in your gut. What makes that number matter for your heart is what those organisms do after you finish a meal. They break down nutrients and release metabolites that enter your bloodstream, travel to your arteries, and either protect them or quietly eat away at them from the inside.
Cardiovascular disease remains the leading cause of death worldwide. The World Health Organization attributed 32% of all global deaths in 2022 to cardiovascular conditions, claiming 19.8 million lives. One in three of those deaths occurred prematurely, in people under 70. And while we have spent decades refining cholesterol medications, blood pressure drugs, and surgical interventions, CVD rates remain stubbornly high. Which is why researchers started looking in less obvious places. What they found sitting in the intestinal tract has changed the conversation.
The "gut-heart axis" describes a two-way communication link between intestinal bacteria and the cardiovascular system. It is not a metaphor. Bacterial DNA has been recovered directly from atherosclerotic plaques, and the metabolites that gut bacteria manufacture from food now appear in the same risk-prediction models as LDL cholesterol and blood pressure. The question is no longer whether your gut affects your heart. It is how much, and what you can do about it.
What your bacteria actually produce when you eat
When food reaches the large intestine, the trillions of bacteria living there go to work. The metabolites they generate depend heavily on what you ate and which bacterial species are present. Two categories of metabolites have received the most research attention in cardiovascular health: trimethylamine N-oxide (TMAO) and short-chain fatty acids (SCFAs). They work in opposite directions.
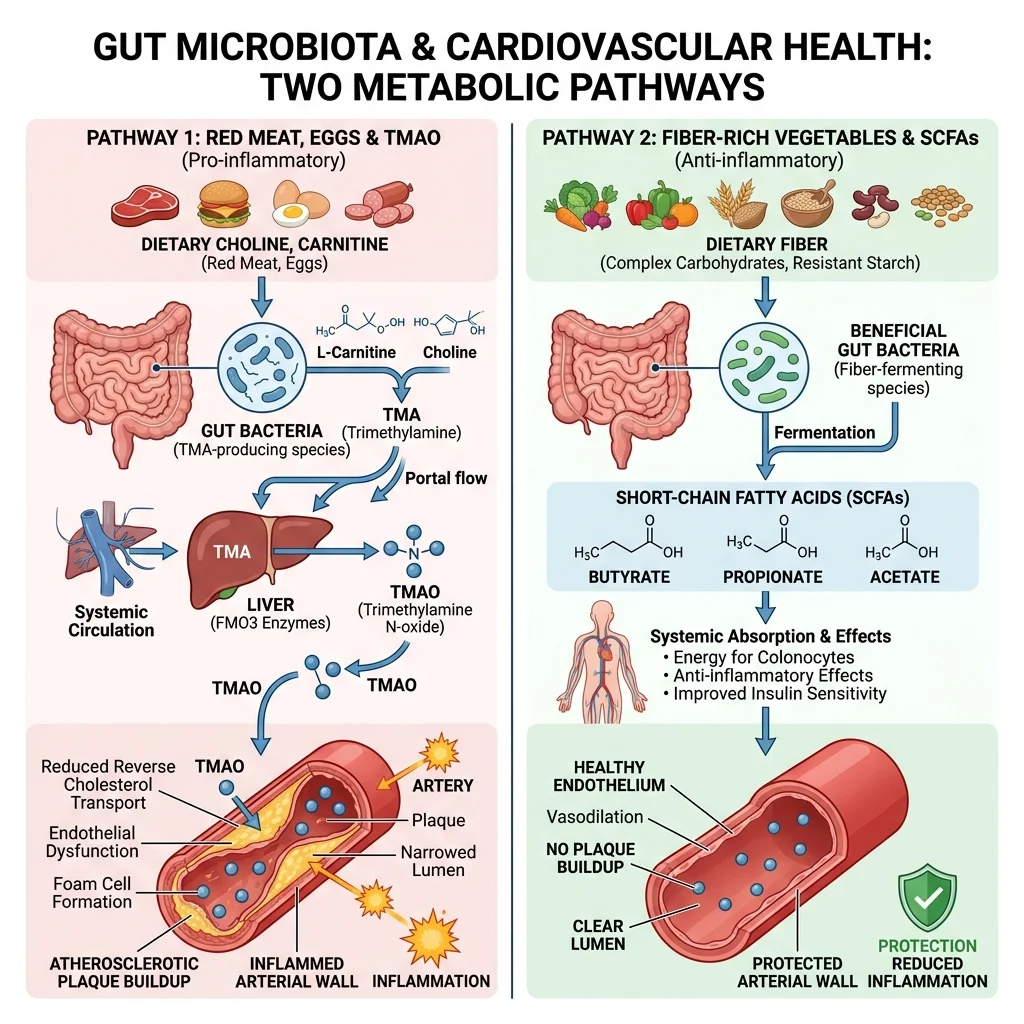
TMAO comes from bacterial digestion of choline, phosphatidylcholine, and L-carnitine, nutrients concentrated in red meat, egg yolks, and full-fat dairy. Gut bacteria convert these precursors into trimethylamine (TMA), which the liver then oxidizes into TMAO using an enzyme called flavin monooxygenase 3 (FMO3). TMAO enters the bloodstream and begins interacting with arterial walls.
SCFAs, on the other hand, are the product of bacterial fermentation of dietary fiber. The three main SCFAs are acetate, propionate, and butyrate, produced in a rough ratio of 3:1:1. They feed the cells lining your colon, reduce inflammation, and send protective signals to your blood vessels. This is a big part of why fiber keeps showing up in every heart-health recommendation you have ever read.
The balance between these two metabolic outputs is determined by three things: what you eat, which bacteria colonize your gut, and how well your intestinal lining holds up. All three are modifiable. That is the part worth paying attention to.
TMAO: the metabolite driving plaque buildup
The connection between TMAO and cardiovascular disease was first uncovered by Dr. Stanley Hazen's group at the Cleveland Clinic, who used metabolomics to identify that plasma levels of TMAO, choline, and betaine could predict cardiovascular risk in humans. The data since then has piled up fast.
A meta-analysis spanning 20 studies and over 31,000 participants found a significant, dose-dependent positive correlation between plasma TMAO levels and both cardiovascular events and mortality. At the individual level, elevated TMAO has been associated with a 74% increased risk of major adverse cardiac events and a 66% increase in all-cause mortality.
Normal plasma TMAO is approximately 0.55 micromoles per liter. Levels above 6 micromoles per liter are associated with significantly increased cardiovascular risk. In patients on hemodialysis, TMAO levels can reach 43 micromoles per liter or higher.
How does TMAO actually damage arteries? The mechanisms are multiple and well-documented:
| Mechanism | What happens | Clinical consequence |
|---|---|---|
| Foam cell formation | TMAO upregulates scavenger receptors CD36 and SRA1 on macrophages | Cholesterol-laden foam cells accumulate in arterial walls |
| Inflammatory activation | TMAO triggers NFkB signaling, NLRP3 inflammasome, and mitochondrial ROS | Chronic vascular inflammation accelerates plaque growth |
| Reverse cholesterol transport inhibition | TMAO reduces expression of cholesterol transporters in the intestine | Less cholesterol is removed from arteries and excreted |
| Platelet hyperactivity | TMAO enhances platelet activation and clotting tendency | Increased risk of clot-related heart attack and stroke |
| Plaque destabilization | TMAO reduces fibrous cap thickness and collagen deposition | Plaques become more vulnerable to rupture |
That last mechanism is particularly alarming. A 2025 study published in Arteriosclerosis, Thrombosis, and Vascular Biology used single-cell RNA sequencing to show that TMAO supplementation in mice upregulated apoptotic genes and downregulated collagen formation in vascular smooth muscle cells. The result: thinner fibrous caps over atherosclerotic plaques, meaning those plaques are more likely to rupture and cause a heart attack. Worth remembering: most heart attacks happen when a plaque ruptures, not when an artery slowly closes off.
A five-year follow-up of 2,235 patients with stable coronary artery disease found that those with the highest TMAO levels had a four-fold greater risk of dying from any cause compared to those with the lowest levels. That risk persisted after adjusting for traditional risk factors.
How short-chain fatty acids protect your heart
If TMAO is the metabolite you want less of, SCFAs are the ones you want more of. They are produced when beneficial gut bacteria ferment dietary fiber, resistant starch, and other non-digestible carbohydrates. The three primary SCFAs each contribute differently to cardiovascular protection.
| SCFA | Key cardiovascular actions | Primary bacterial producers |
|---|---|---|
| Butyrate | Maintains gut barrier, inhibits HDACs (reducing inflammation), reduces atherosclerotic plaque area by up to 50% in animal models | Faecalibacterium prausnitzii, Roseburia intestinalis, Coprococcus |
| Propionate | Reduces blood pressure via vasodilation, modulates lipid metabolism, attenuates appetite | Bacteroides, Dialister, Veillonella |
| Acetate | Regulates fat oxidation, central appetite control, anti-inflammatory via HDAC inhibition | Akkermansia muciniphila, Bifidobacterium, Prevotella |
Butyrate is worth lingering on. It is the primary fuel for colonocytes, the cells that line your colon, but it does a lot more than feed them. As a potent histone deacetylase (HDAC) inhibitor, butyrate suppresses pro-inflammatory cytokines like TNF-alpha, IL-12, and interferon-gamma while boosting anti-inflammatory IL-10. In mice, butyrate pretreatment reduced atherosclerotic plaque area in the aortic arch by 50%, decreased oxidized LDL uptake, and reduced foam cell deposition.
If you are familiar with the gut-brain axis, the gut-heart axis operates through similar machinery. The same bacterial metabolites that affect neurotransmitter signaling also regulate vascular inflammation, blood pressure, and cholesterol metabolism. The overlap between the two is bigger than most people think.
The clinical data on fiber and blood pressure adds weight to the SCFA story. A meta-analysis found that dietary fiber consumption reduced systolic blood pressure by 0.9 mmHg and diastolic by 0.7 mmHg on average. Beta-glucan fibers had a stronger effect, reducing systolic BP by 2.9 mmHg and diastolic by 1.5 mmHg. Those numbers look small on paper. But shift the average blood pressure of an entire population by even 1-2 mmHg and the number of heart attacks and strokes drops measurably.
There is also evidence that SCFAs help reduce cholesterol availability in the intestine by converting cholesterol to coprostanol, a form that can be excreted from the body rather than absorbed. And in animal studies, soluble dietary fiber reduced TMA production by 40.6% and TMAO by 62.6%, suggesting that fiber does not just produce beneficial metabolites; it may actively suppress the harmful ones.
Leaky gut as a silent cardiovascular threat
The conversation about leaky gut syndrome tends to focus on digestive symptoms, food sensitivities, and autoimmune conditions. But intestinal permeability plays a more direct role in cardiovascular disease than most cardiologists would have guessed ten years ago.
Your intestinal lining is a single layer of cells held together by tight junction proteins. When that barrier is functioning properly, it allows nutrients through while keeping bacteria and their inflammatory components, particularly lipopolysaccharide (LPS), contained within the gut. When the barrier breaks down, which can happen through poor diet, chronic stress, antibiotic use, or dysbiosis, LPS and bacterial fragments leak into the bloodstream.
| Factor | Healthy gut barrier | Compromised gut barrier |
|---|---|---|
| Tight junction integrity | Intact, regulated paracellular transport | Loosened junctions, increased permeability |
| Mucus layer | Thick, protective, butyrate-stimulated | Thinned, insufficient bacterial separation |
| LPS translocation | Minimal, contained to gut lumen | Systemic entry, triggers low-grade inflammation |
| Cardiovascular impact | Baseline inflammatory state | Endothelial activation, vascular remodeling |
This creates a condition researchers call "metabolic endotoxemia": chronic, low-grade inflammation driven by bacterial products in the bloodstream. It does not produce the obvious symptoms of an acute infection, but it does activate endothelial cells lining the arteries, recruit immune cells to vessel walls, and promote the same inflammatory cascade that underlies atherosclerosis.
A human study examining gut permeability in hypertension found that markers of increased gut permeability and LPS were elevated in hypertensive individuals, with a strong correlation between gut permeability and systolic blood pressure. Patients with chronic heart failure show similar patterns: decreased beneficial bacteria and increased pathogenic species including Campylobacter and Salmonella, alongside markers of intestinal barrier dysfunction.
Butyrate is the gut lining's primary repair signal. It orchestrates tight junction proteins, stimulates mucin production, and feeds colonocytes the energy they need to maintain barrier function. When butyrate-producing bacteria decline, which happens on low-fiber, high-fat diets, the barrier weakens and cardiovascular risk climbs. Traditional nutrition science spent decades focused on fat and cholesterol. This mechanism, the slow erosion of the gut barrier, was hiding in plain sight the whole time.
Myth vs. reality: what we know and what we don't
This field moves fast, and some of the claims circulating online have outrun the data. A few things worth straightening out:

| Claim | Reality |
|---|---|
| TMAO is a definitive cause of heart disease | TMAO is an independent risk factor with strong associative data and plausible mechanisms, but whether it is a direct cause or a marker of other processes remains debated. Seafood contains high TMAO yet is cardioprotective, which complicates the picture. |
| Everyone should avoid eggs and red meat completely | TMAO production depends on your specific gut bacteria, not just food intake. Some people eating the same diet produce far more TMAO than others. Vegetarians and vegans produce very little TMAO regardless of choline intake. |
| Probiotics can cure heart disease | A randomized trial found that B. lactis Probio-M8 combined with statins improved LDL cholesterol and cardiac metrics beyond statins alone, but probiotics are supplements to established treatments, not replacements. |
| TMAO testing should be routine | TMAO testing is available through Cleveland HeartLab and may add useful information for some patients, but it is not yet part of standard cardiovascular screening guidelines. |
| Only bacteria matter in the gut-heart connection | Most research has focused on bacteria while neglecting viruses, archaea, and fungi. The full picture of the gut-heart axis likely involves microbial interactions we have not yet characterized. |
The biggest limitation of current research is that most studies are observational. They can show associations between microbiome composition and cardiovascular outcomes, but establishing clear cause-and-effect in humans is harder. Person-to-person variability in gut microbiome composition adds another layer of complexity. What works for one individual's bacterial ecosystem may not work for another's.
Still, the mechanistic evidence from animal models and cell culture is strong enough to act on. And here is the thing that makes the gut-heart axis practical even without perfect data: the dietary changes that reduce cardiovascular risk through the microbiome (more fiber, more plants, less processed red meat) are the same changes that reduce risk through every other known pathway. You do not need to wait for a definitive clinical trial to eat more beans and fewer hot dogs.
Feeding your microbiome for a healthier heart
So what do you actually do with all this? The research points to a handful of strategies. Some aim at lowering TMAO. Others boost SCFA production. The best ones do both at once.
A systematic review of randomized clinical trials identified the Mediterranean diet, high-fiber diets, polyphenol-rich diets, and diets containing polyunsaturated fatty acids as the most promising for reducing CVD risk factors through microbiome modulation. The Mediterranean diet is especially interesting because it naturally contains DMB (3,3-dimethyl-1-butanol), a compound found in extra virgin olive oil and red wine that inhibits TMA lyase, the bacterial enzyme that starts the TMAO production chain. In animal studies, DMB added to drinking water reduced both TMAO levels and arterial plaque formation.
The anti-inflammatory eating patterns that nutritionists have recommended for years turn out to be the same ones that cultivate a heart-protective microbiome. That is probably not a coincidence.
Fiber intake matters more than most people think. The standard recommendation is 25-30 grams of fiber daily. Most adults eat about half that. Each gram of fiber that reaches the large intestine becomes substrate for SCFA-producing bacteria. Diversity matters too: different fiber types (soluble, insoluble, resistant starch) feed different bacterial communities, which is why variety in fruits, vegetables, legumes, and whole grains outperforms any single fiber supplement.
Some specific foods and what they do to your gut-heart chemistry:
- Fermented foods (yogurt, kefir, kimchi, sauerkraut): provide live Lactobacillus and Bifidobacterium species that support SCFA production and may have direct antihypertensive effects through ACE-inhibitory peptide production
- Legumes and beans: rich in resistant starch and soluble fiber that selectively feed butyrate-producing bacteria like Faecalibacterium prausnitzii
- Extra virgin olive oil: contains DMB, a natural TMA lyase inhibitor that reduces TMAO production
- Polyphenol-rich foods (berries, green tea, dark chocolate): modulate gut bacterial composition toward more favorable profiles
- Allium vegetables (garlic, onions): contain prebiotic fibers and compounds that support beneficial bacterial growth
Maintaining arterial health involves more than diet alone. The interplay between vitamin K2 and arterial calcification is another piece of the cardiovascular puzzle, and regular physical activity, including practices like sauna bathing for heart health, independently improves both gut microbial diversity and cardiovascular function.
What about probiotic supplements? The evidence is encouraging but still early. The most compelling clinical data comes from a randomized trial of 60 coronary artery disease patients where B. lactis Probio-M8 combined with conventional statin therapy produced significantly greater improvements in serum LDL cholesterol and patient-reported cardiac outcomes compared to statins plus placebo. But probiotic effects are strain-specific, dose-dependent, and need to be maintained long-term. Short-term dramatic changes produce temporary microbiome shifts that revert once you stop.
Frequently asked questions
Can a blood test for TMAO predict my heart disease risk?
TMAO blood testing is available through Cleveland HeartLab and some research institutions. Elevated levels (above 6 micromoles per liter) are associated with increased cardiovascular risk independently of traditional risk factors. However, TMAO testing is not yet included in standard screening guidelines, and levels fluctuate based on recent diet, kidney function, and individual gut bacterial composition. It may be most useful for people with unexplained cardiovascular risk who want additional data points to guide dietary decisions.
Should I stop eating eggs and red meat to lower TMAO?
Probably not. TMAO production depends on which bacteria you carry, not just what you eat. Vegetarians and vegans produce almost no TMAO even when given choline supplements, because they lack the bacterial populations that develop from years of regular red meat consumption. Cutting back on red meat and full-fat dairy while eating more plants and fiber will gradually shift your gut bacteria toward lower TMAO output. You do not need to go fully vegan to see the effect.
How long does it take for dietary changes to affect gut bacteria?
Measurable shifts in gut microbial composition can occur within days of a major dietary change, but these early shifts are unstable. Sustained changes in bacterial community structure typically require 2-4 weeks of consistent dietary modification, and achieving a stable new equilibrium takes months. This is why crash diets and short-term interventions produce temporary microbiome effects that revert once old eating patterns resume.
Are probiotic supplements effective for heart health?
Some probiotic strains show promise. Lactobacillus helveticus has been shown to produce ACE-inhibitory peptides that lower blood pressure, and B. lactis Probio-M8 improved cardiac outcomes when added to statin therapy in a clinical trial. However, benefits are strain-specific, not all commercial probiotics contain strains with cardiovascular evidence, and effects require ongoing supplementation. Probiotics work best as part of a fiber-rich diet that provides substrate for beneficial bacteria to thrive.
Does the gut-heart axis affect people who already have heart disease?
Yes. Patients with existing heart failure show distinct patterns of gut dysbiosis: reduced populations of butyrate-producing bacteria (particularly Eubacterium hallii and Lachnospiraceae) and increased pathogenic species. This dysbiosis correlates with disease severity, inflammation levels, and mortality. Managing gut health through dietary fiber, fermented foods, and potentially targeted probiotics may complement conventional heart failure treatments, though this remains an active area of clinical research.
Related Articles
- The gut-brain axis: how your microbiome affects mood and cognition - The same bacterial metabolites that influence your heart also shape your mental health through parallel signaling pathways.
- Leaky gut syndrome: symptoms, causes, and evidence-based healing - Intestinal permeability plays a direct role in cardiovascular inflammation through bacterial endotoxin translocation.
- Anti-inflammatory eating patterns: Mediterranean vs. DASH vs. plant-based - The dietary patterns with the strongest evidence for gut-mediated cardiovascular protection.
- Vitamin K2 and arterial calcification - Another piece of the arterial health puzzle, working through complementary mechanisms to the gut-heart axis.
- Sauna bathing for heart health - Heat therapy independently improves gut microbial diversity and cardiovascular function.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.