Your arteries have GLP-1 receptors, and that changes everything
When semaglutide or liraglutide enters your bloodstream, it does not just travel to your gut and pancreas. GLP-1 receptors sit on the surface of cardiomyocytes, vascular endothelial cells, smooth muscle cells lining your arteries, and several types of immune cells. That distribution pattern matters because it means these drugs have direct access to the tissues that drive cardiovascular disease, not only indirect access through weight loss or blood sugar control.
Think of chronic cardiovascular disease like a slow-burning fire inside your artery walls. Immune cells called macrophages pile in, oxidize LDL cholesterol, form foam cells, and build unstable plaques. GLP-1 receptor activation interrupts this process at multiple points simultaneously.
The anti-inflammatory effects are striking. In cardiovascular outcome trials, semaglutide reduced high-sensitivity C-reactive protein (hs-CRP) by 20 to 30 percent. That is not a minor shift in a lab value. CRP reflects systemic inflammation, and a reduction of that magnitude rivals what some dedicated anti-inflammatory drugs achieve. The mechanism runs deeper than just lowering a blood marker: GLP-1 receptor activation suppresses the NLRP3 inflammasome, a molecular alarm system inside macrophages that triggers release of potent inflammatory proteins IL-1-beta and IL-18. When the NLRP3 inflammasome stays quiet, the entire inflammatory cascade that destabilizes arterial plaques loses its fuel supply.
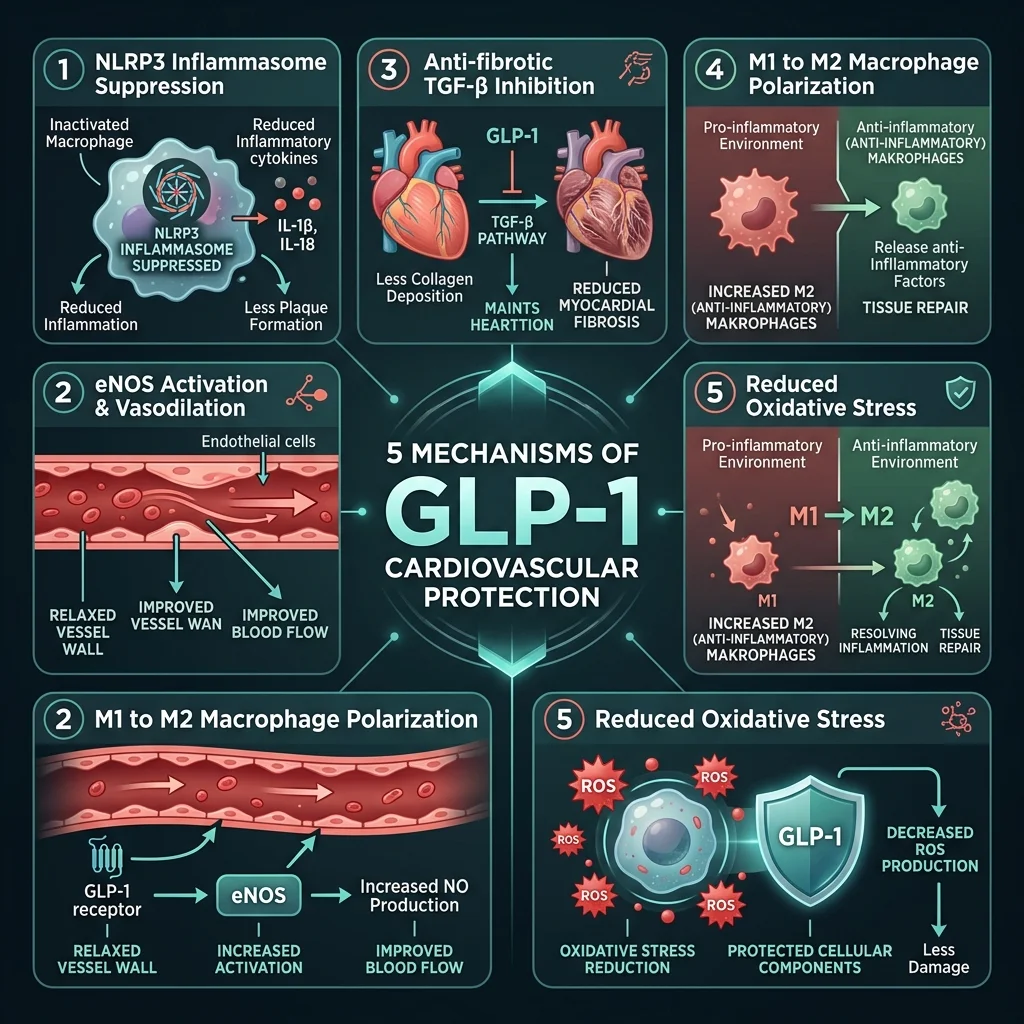
GLP-1 receptor agonists also push macrophages from their aggressive, pro-inflammatory M1 state toward a calmer, tissue-repairing M2 phenotype. This macrophage polarization shift directly stabilizes atherosclerotic plaques by reducing the kind of internal erosion that causes heart attacks.
On the vascular side, these drugs increase production of nitric oxide by enhancing the phosphorylation of endothelial nitric oxide synthase (eNOS). More nitric oxide means better vasodilation, lower blood pressure, and reduced platelet stickiness. Clinical measurements show GLP-1 agonists reduce systolic blood pressure by 2 to 5 mmHg and trim LDL cholesterol by 5 to 10 percent. Neither number sounds dramatic on its own, but layered on top of the anti-inflammatory and anti-thrombotic effects, they add up to meaningful vascular protection.
At the cellular level inside heart muscle, GLP-1 receptor activation triggers pro-survival signaling through the PI3K/Akt and protein kinase A (PKA) pathways. In porcine models of heart attack, the GLP-1 agonist exenatide reduced infarct size by 20 to 25 percent by ramping up these protective cascades. The drugs also dial down oxidative stress: semaglutide cut NOX4 expression (a major source of damaging reactive oxygen species) by 40 percent in endothelial cells and reduced superoxide production by 35 percent in vascular smooth muscle cells in preclinical models.
The practical takeaway: GLP-1 drugs are not just weight-loss medications that happen to help the heart because people get lighter. They interact directly with cardiovascular tissue through at least five distinct biological pathways. When researchers adjusted for weight loss and blood sugar changes in the SELECT trial using time-dependent Cox regression, the cardiovascular benefit was barely affected. The protection appears to come from the drug itself, not just from what the scale says.
17,604 patients, one clear signal: the SELECT trial results
Before SELECT, every major GLP-1 cardiovascular outcome trial enrolled patients with type 2 diabetes. The question lingering over the field was whether these drugs protected the heart only because they improved diabetic metabolic dysfunction, or whether they worked through mechanisms available to anyone with obesity and cardiovascular disease. SELECT answered that question.
The trial enrolled 17,604 adults aged 45 and older who had established cardiovascular disease and a BMI of 27 or higher but no diabetes. Half received weekly injections of semaglutide 2.4 mg; half received placebo. Researchers followed them for a mean of 39.8 months.
The primary endpoint was MACE: a composite of cardiovascular death, nonfatal heart attack, or nonfatal stroke. Semaglutide reduced this composite by 20 percent (hazard ratio 0.80, 95% CI 0.72 to 0.90, P less than 0.001). In absolute numbers, 6.5 percent of semaglutide patients experienced a MACE event compared with 8.0 percent on placebo. For a drug originally developed for blood sugar control, that 1.5 percentage point absolute reduction across roughly 3.3 years represents a substantial shift in cardiovascular outcomes.
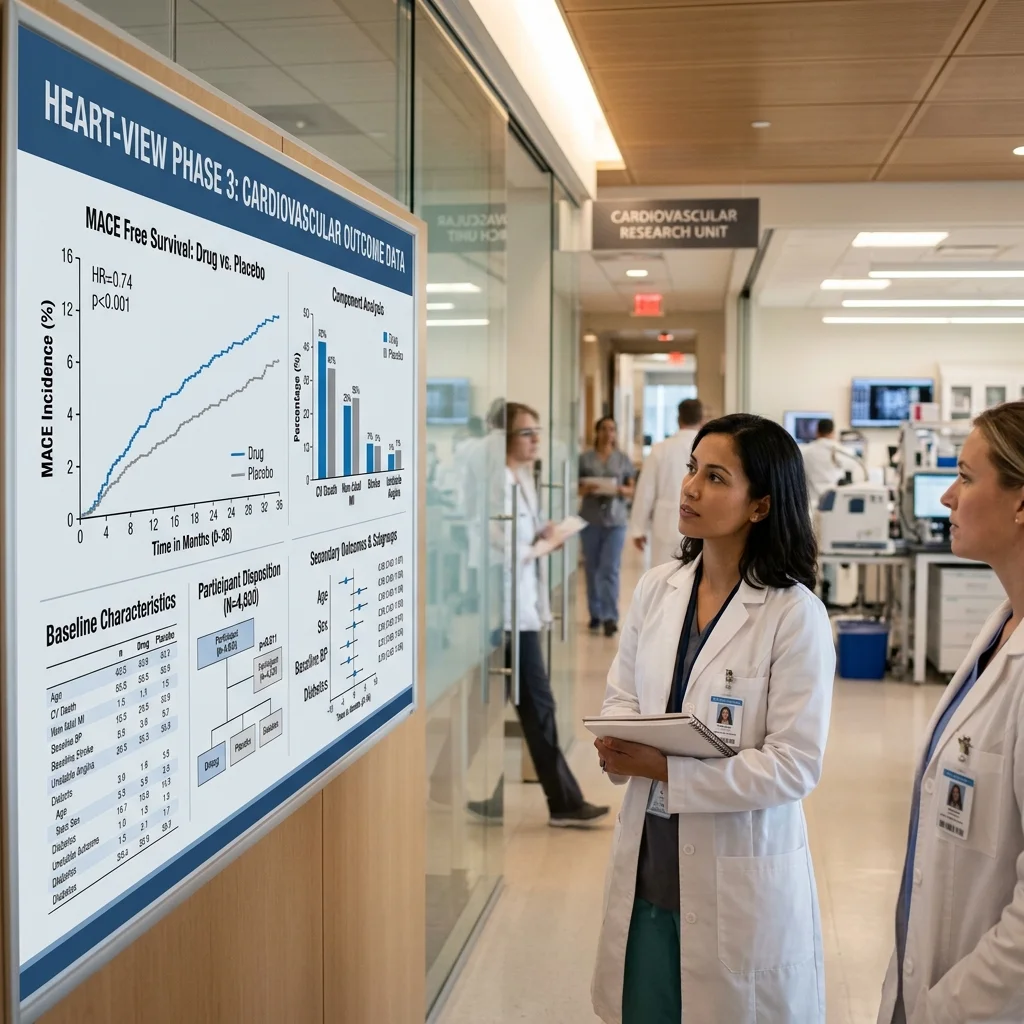
SELECT was not a small signal buried in secondary endpoints. The P-value crossed the significance threshold decisively, and the confidence interval did not flirt with 1.0. The trial was funded by Novo Nordisk, which is worth noting for transparency, but the results were published in the New England Journal of Medicine and have since been scrutinized, replicated in subgroup analyses, and cited in cardiology guidelines.
Quick stat: 6.5% of semaglutide patients had a heart attack, stroke, or cardiovascular death over 3.3 years, compared with 8.0% on placebo. The 20% MACE reduction held regardless of age, sex, or BMI.
What SELECT did not answer is how long patients need to stay on treatment to maintain that protection. That question generated its own line of research, which we address in the myth-versus-reality section below.
Heart failure patients saw the biggest benefit, and nobody expected that
Heart failure was not the original target. But when researchers ran prespecified analyses of the SELECT data, the heart failure subgroup jumped off the page. Of the 17,604 SELECT participants, 4,286 (24.3 percent) had a history of heart failure at enrollment. Among those patients, semaglutide reduced MACE by 28 percent (HR 0.72, 95% CI 0.60 to 0.87), compared with the 20 percent reduction in the overall population.
The heart failure composite endpoint, which combined cardiovascular death with hospitalization or urgent hospital visits for heart failure, dropped by 21 percent (HR 0.79, 0.64 to 0.98). Cardiovascular death alone fell by 24 percent in this subgroup. These are the kinds of numbers that get cardiologists to rethink treatment algorithms.
The benefits held across heart failure subtypes. Patients with heart failure with reduced ejection fraction (HFrEF) saw a 35 percent MACE reduction, while those with heart failure with preserved ejection fraction (HFpEF) saw a 31 percent reduction. HFpEF, which accounts for roughly half of all heart failure cases, has been notoriously difficult to treat. For decades, most drugs that worked for HFrEF failed in HFpEF trials. Semaglutide worked in both.
| Population | MACE Reduction | Hazard Ratio (95% CI) |
|---|---|---|
| SELECT overall (no diabetes) | 20% | 0.80 (0.72-0.90) |
| SELECT with heart failure | 28% | 0.72 (0.60-0.87) |
| SELECT HFrEF subgroup | 35% | 0.65 (0.49-0.87) |
| SELECT HFpEF subgroup | 31% | 0.69 (0.51-0.91) |
The dedicated STEP-HFpEF trials built on this signal. A pooled analysis of the STEP-HFpEF and STEP-HFpEF DM trials, which together enrolled 1,145 patients with obesity-related HFpEF across 129 sites in 18 countries, showed semaglutide improved the Kansas City Cardiomyopathy Questionnaire Clinical Summary Score by 7.5 points over placebo (P less than 0.0001). That score measures how much heart failure limits daily life. A 5-point change is considered clinically meaningful. Patients also walked 17.1 meters farther in six minutes and lost 8.4 percent more body weight than the placebo group.
An echocardiography substudy drilled into what was happening structurally inside the heart. After 52 weeks, semaglutide patients showed a 6.13 mL reduction in left atrial volume compared with placebo (P=0.0013), along with less right ventricular enlargement and improved measures of diastolic function (E/A ratio and E/e' average). The left atrium stretches and enlarges as heart failure worsens; shrinking it back suggests the disease process is being reversed, not just masked.
Critically, some of these structural improvements were weight-independent. The improvements in E-wave velocity, E/e' ratio, and right ventricular size did not correlate with the degree of weight loss. The researchers concluded semaglutide appeared to be "disease modifying" in obesity-related HFpEF. That language is carefully chosen in medicine. It means the drug may alter the underlying biology, not just treat symptoms.
One finding that should quiet concerns about safety in heart failure: serious adverse events were actually less frequent with semaglutide (161 events) than with placebo (301 events) in the pooled STEP-HFpEF analysis. The drug did not just avoid harming heart failure patients; the treated group had fewer serious problems overall.
The liver problem hiding inside heart disease
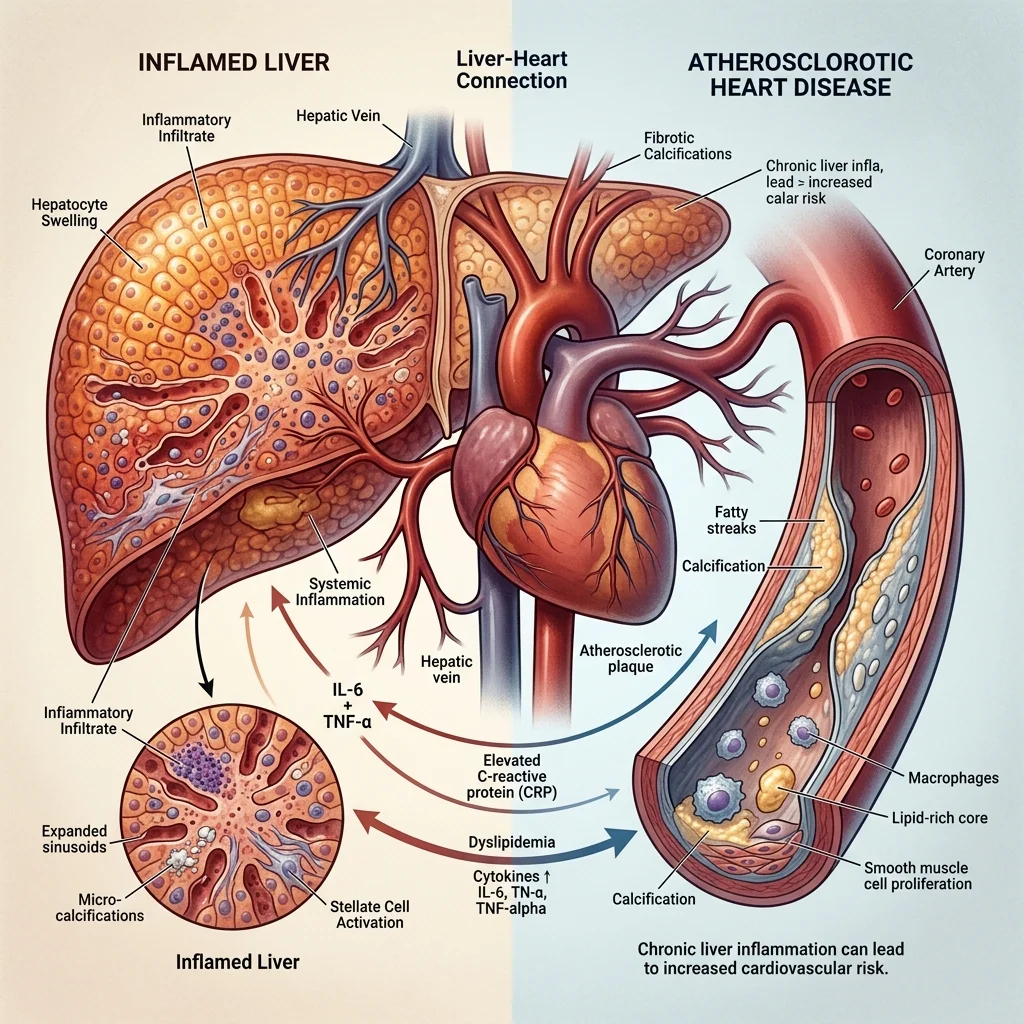
Cardiovascular disease and liver disease do not just coexist. They fuel each other. Metabolic dysfunction-associated steatohepatitis (MASH), the inflammatory form of fatty liver disease, affects more than 250 million people worldwide. And here is the fact that reframes the entire condition: cardiovascular disease, not liver failure, is the leading cause of death in people with MASH.
The link is not coincidental. A chronically inflamed liver pumps inflammatory cytokines, clotting factors, and atherogenic lipoproteins into the bloodstream. The severity of liver fibrosis correlates directly with the incidence of major cardiovascular events. Think of the liver as an upstream factory: when it malfunctions, it ships inflammatory products downstream into the arteries and heart.
The ESSENCE phase 3 trial tested semaglutide 2.4 mg in adults with MASH and moderate-to-advanced liver fibrosis (stages F2 and F3). The results, published in the New England Journal of Medicine (2025;392(21):2089-2099), earned semaglutide FDA accelerated approval for MASH. The secondary analyses presented at the AASLD Liver Meeting 2025 revealed something surprising: even patients who lost 2 percent or less of their body weight on semaglutide still saw liver injury resolve. In that minimal weight-loss subgroup, 48.4 percent of semaglutide patients experienced resolution of liver injury compared with 25.8 percent on placebo.
Professor Philip Newsome of King's College London, the trial's co-chief investigator, put it plainly: "These data suggest that the effects of semaglutide 2.4 mg in this study may not be solely dependent on weight loss."
A prespecified analysis of the SELECT trial, published in Nature Medicine, connected the liver and heart data directly. Researchers stratified SELECT participants by their Fibrosis-4 (FIB-4) score, a blood-test-based estimate of liver fibrosis risk. Among patients with intermediate fibrosis risk (FIB-4 of 1.3 or higher), semaglutide reduced MACE by 26 percent (HR 0.74, 95% CI 0.63 to 0.88, P=0.0004). That exceeded the 20 percent reduction seen in the overall trial population. In the small group with the highest fibrosis scores (FIB-4 above 2.67), the trend pointed toward a 34 percent MACE reduction (HR 0.66), though the subgroup was too small for statistical significance.
| Fibrosis Risk Category | Patients | MACE Reduction | P-value |
|---|---|---|---|
| Overall SELECT population | 17,604 | 20% | <0.001 |
| FIB-4 ≥ 1.3 (intermediate risk) | 6,567 | 26% | 0.0004 |
| Age-adjusted FIB-4 | 3,664 | 21% | 0.035 |
| FIB-4 > 2.67 (high risk) | 509 | 34% (trend) | 0.11 |
Semaglutide also improved liver-specific markers: a 28 percent greater reduction in the Fatty Liver Index compared with placebo, along with a 17 percent drop in GGT (a liver enzyme that rises with liver damage and independently predicts cardiovascular events).
The clinical implication is that for patients with both fatty liver disease and cardiovascular risk, semaglutide may be treating two conditions with one drug. And the patients who appear to benefit most from cardiovascular protection are exactly the patients with the worst liver disease. That is not a coincidence. It is probably because reducing liver-driven inflammation has an outsized impact on arterial health.
What the evidence actually shows, and what it does not
The cardiovascular data on GLP-1 receptor agonists is strong, but it is not without caveats. A clear-eyed look at the evidence means acknowledging both sides.
| Claim | Evidence Status |
|---|---|
| GLP-1 drugs reduce MACE in people with obesity and CVD | Confirmed: SELECT, 17,604 patients, 20% reduction |
| Benefits work independently of weight loss | Supported: Cox regression adjustments, weight-independent echo findings |
| Protection requires continuous treatment | Confirmed: Two large studies show rapid erosion on discontinuation |
| GLP-1 drugs cure heart disease | Not supported: Protection is ongoing, not curative |
| All GLP-1 drugs have identical CV effects | Unproven: Most data is from semaglutide; class effect is assumed but not confirmed for all agents |
| Benefits are free of serious side effects | Partially true: Fewer serious AEs than placebo in HFpEF, but 16.6% discontinuation rate in SELECT vs 8.2% placebo |
The discontinuation problem deserves particular attention. Researchers at Washington University School of Medicine in St. Louis followed 333,687 U.S. veterans with type 2 diabetes for three years in a study published in BMJ Medicine. The findings were blunt: stopping GLP-1 treatment for as little as six months was linked to a measurable increase in cardiovascular risk. After two years off the drugs, MACE risk climbed by 22 percent compared with continuous use.
Dr. Ziyad Al-Aly, the study's senior author, described the dynamic as "metabolic whiplash." When patients stop, inflammation rebounds, blood pressure creeps back up, and cholesterol worsens. Weight regain is visible; the metabolic reversal happening underneath is not. Restarting the drug helped, but only partially. Patients who interrupted treatment and resumed averaged a 12 percent risk reduction, compared with 18 percent for those who never stopped.
This creates a difficult reality. With 36 to 81 percent of patients discontinuing GLP-1 drugs in the first year, often due to cost, side effects, or supply shortages, the cardiovascular benefits demonstrated in controlled trials may not fully translate to real-world outcomes. Al-Aly put it directly: "There is enormous exuberance about starting GLP-1 drugs, but not nearly enough attention to what happens when people stop."
The funding question also matters. Every major GLP-1 cardiovascular outcome trial, including SELECT, STEP-HFpEF, and ESSENCE, was funded by Novo Nordisk. The trials were large, well-designed, and published in top journals with independent event adjudication committees. But independent replication from non-industry-funded studies would strengthen the evidence base. The Veterans Affairs discontinuation studies, which were independently funded, at least confirm the cardiovascular effects are real enough that stopping the drugs measurably worsens outcomes.
Another gap: most of the mechanism data showing direct cardioprotection comes from animal studies and cell cultures. The human clinical trial data is convincing on outcomes, but the precise biological mechanisms operating in living human hearts remain partly inferred from preclinical work.
The next wave of trials and unanswered questions
The GLP-1 cardiovascular story is moving fast, but several questions remain open. The ESSENCE trial's Part 2 is ongoing, with expected readout in 2029, and will report on whether semaglutide reduces actual liver-related clinical events (not just biopsy improvements) over 240 weeks. That data will clarify whether resolving MASH inflammation translates into fewer liver transplants, less cirrhosis, and, importantly, fewer cardiovascular deaths in the MASH population.
Dual-agonist drugs are expanding the field. Tirzepatide, which activates both GLP-1 and GIP receptors, has produced greater weight loss than semaglutide in head-to-head trials and is now generating its own cardiovascular outcome data. A review in PMC notes that tirzepatide is showing significant MACE reduction in its own CVOT. Whether the dual mechanism produces additive cardiovascular benefit beyond what GLP-1 alone delivers remains to be fully quantified.

Several practical questions need answers. How long does someone need to stay on a GLP-1 drug before the cardiovascular protection becomes durable enough to survive discontinuation? The current data suggests that less than 18 months of treatment provides no lasting cardiovascular benefit after stopping. Can lower maintenance doses preserve protection with fewer side effects? Do different GLP-1 receptor agonists (liraglutide, dulaglutide, oral semaglutide) offer equivalent cardiovascular protection, or is it specific to injectable semaglutide at the 2.4 mg dose studied in SELECT?
For clinicians, the current evidence already supports a shift in how these drugs are prescribed. GLP-1 receptor agonists are no longer just diabetes drugs or weight-loss drugs that happen to help the heart. For patients with established cardiovascular disease and obesity, they are cardiovascular drugs that happen to cause weight loss. The distinction matters for how insurance covers them, how doctors prescribe them, and how patients think about adherence.
Frequently Asked Questions
Do GLP-1 drugs protect the heart only through weight loss?
No. Multiple lines of evidence suggest weight-independent cardioprotection. When SELECT trial researchers adjusted for weight loss and blood sugar changes, the cardiovascular benefit persisted. The STEP-HFpEF echocardiography substudy found improvements in diastolic function and right ventricular size that did not correlate with degree of weight loss. And the ESSENCE MASH trial showed liver injury resolution even in patients who lost less than 2 percent of body weight. GLP-1 receptors on heart cells, blood vessel linings, and immune cells enable direct anti-inflammatory, anti-fibrotic, and vascular protective effects that operate regardless of what happens on the scale.
What happens to cardiovascular protection if I stop taking a GLP-1 drug?
Protection erodes quickly. A study of 333,687 veterans published in BMJ Medicine found that stopping for just six months increased MACE risk by 4 to 8 percent compared with continuous use. After two years off, risk climbed by 22 percent. Resuming the drug restored some protection, but not all of it. The cardiovascular benefits appear to require ongoing treatment, similar to how statins work: the protection is real but not permanent once the drug is withdrawn.
Are GLP-1 cardiovascular benefits proven in people without diabetes?
Yes. The SELECT trial specifically excluded patients with diabetes and still demonstrated a 20 percent reduction in heart attacks, strokes, and cardiovascular death. All 17,604 participants had established cardiovascular disease and overweight or obesity but no history of diabetes. This was the first large trial to confirm GLP-1 cardiovascular protection operates independently of diabetes treatment.
Can GLP-1 drugs help with both liver disease and heart disease simultaneously?
Current data strongly suggests yes. Semaglutide received FDA accelerated approval for MASH based on the ESSENCE trial, and a Nature Medicine analysis of SELECT found that patients with the highest liver fibrosis risk scores experienced a 26 percent reduction in MACE. Since cardiovascular disease is the leading killer of MASH patients, a single drug that addresses both the liver inflammation and the downstream cardiovascular risk may be more efficient than treating each condition separately.
Are there serious heart-related side effects from GLP-1 drugs?
In heart failure populations specifically, semaglutide actually produced fewer serious adverse events than placebo (161 vs. 301 events in the STEP-HFpEF pooled analysis). However, gastrointestinal side effects (nausea, vomiting, diarrhea) cause significant discontinuation: 16.6 percent of semaglutide patients in SELECT stopped due to adverse events compared with 8.2 percent on placebo. There are no signals of increased heart rhythm problems, heart attacks, or strokes from the drug itself. The main cardiovascular concern is what happens when patients stop taking it.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.