What Are Oxalates and Why Should You Care?
Oxalic acid is a small organic compound that plants produce naturally during photosynthesis. Once it loses its hydrogen atoms in your body, it becomes oxalate — a negatively charged ion with a strong appetite for calcium. That binding tendency is the root of nearly every health concern associated with this molecule.
Your body encounters oxalate from two directions. The liver synthesizes it as a metabolic byproduct of glyoxylate metabolism, and this endogenous production accounts for roughly 60 to 80 percent of total plasma oxalate. The remainder enters through food — green vegetables, nuts, grains, cocoa, and tea all deliver varying amounts. In a typical Western diet, daily oxalate intake falls somewhere between 100 and 200 milligrams.
What makes oxalate different from other dietary compounds is that your body cannot break it down. Mammals lack the enzymes to metabolize oxalic acid further, so every molecule that enters the bloodstream must exit through the kidneys. When urinary oxalate concentrations climb too high, calcium and oxalate ions find each other in the renal tubules and form crystals. Those crystals can grow into kidney stones, damage tubular cells, or trigger inflammatory cascades that progressively scar kidney tissue.
Quick fact: Oxalate is 15 to 20 times more potent than calcium at driving kidney stone formation. Even a modest 5 mg/day increase in urinary oxalate can double the risk of developing a kidney stone.
The lifetime risk of developing at least one kidney stone sits between 10 and 15 percent in developed countries, and calcium oxalate stones make up roughly 80 percent of all cases. That prevalence has climbed steadily over recent decades, paralleling dietary shifts toward higher oxalate consumption and lower calcium intake. The mechanics of how oxalate moves through your body, and what tips the balance toward crystal formation, have direct implications for long-term kidney health.
Where Do Oxalates Hide? A Complete Food Map
Not all plant foods deliver oxalate equally. The concentration varies enormously depending on the species, growing conditions, and which part of the plant you eat. A single serving of spinach can dump more oxalate into your gut than an entire day of eating lower-oxalate vegetables. Knowing which foods sit at the top of the scale lets you make targeted adjustments rather than eliminating entire food groups.
Research published in Nutrients (2020) compiled oxalate measurements across hundreds of food samples. The following table captures the most significant dietary sources:
| Food | Oxalate (mg per 100g) | Category |
|---|---|---|
| Amaranth grain | 1,090 | Very High |
| Spinach | 658 | Very High |
| Rhubarb | 433 | Very High |
| Almonds | 407 | Very High |
| Tofu | 274 | High |
| Cocoa powder | 111 | High |
| Rice bran | 112 | High |
| Hazelnuts | 100 | High |
| Peanuts | 95 | High |
| Beets | 61 | Moderate |
| Soybeans | 77 | Moderate |
| Potatoes | 50 | Moderate |
| Pistachios | 49 | Moderate |
| Raspberries | 19 | Low |
Spinach deserves particular attention. In large prospective cohorts tracking over 240,000 participants, spinach alone contributed more than 40 percent of total dietary oxalate intake. A normal serving of 50 to 100 grams delivers between 500 and 1,000 milligrams — five to ten times the amount that meaningfully raises urinary oxalate levels. Green smoothie culture has amplified this issue; blending multiple cups of raw spinach into a single drink concentrates the oxalate dose in ways that whole-food eating rarely achieves.
Case reports illustrate the extremes. One patient developed acute kidney injury after drinking 16 eight-ounce glasses of black iced tea daily. Another required dialysis following weeks of consuming 500 grams of fresh rhubarb per day. Star fruit juice — containing up to 829 mg/dL of oxalate — has caused kidney failure in multiple documented cases, particularly in people with pre-existing kidney disease.
One factor that dramatically changes how much oxalate you actually absorb is solubility. Research comparing turmeric (rich in soluble oxalate) against cinnamon (mostly crystalline oxalate) found absorption rates of 8.2 percent versus 2.6 percent respectively, even with identical total oxalate content. Foods where oxalate already sits bound to calcium — forming insoluble crystals — pose less risk than those carrying free, soluble oxalate.
From Gut to Kidney: How Oxalates Travel Through Your Body
The journey from your plate to your kidneys involves several steps where things can go right or badly wrong. Under normal circumstances, only about 5 to 15 percent of the oxalate you eat actually crosses the intestinal wall into your bloodstream. The rest passes through your digestive tract and leaves in stool, often bound to calcium in insoluble complexes.
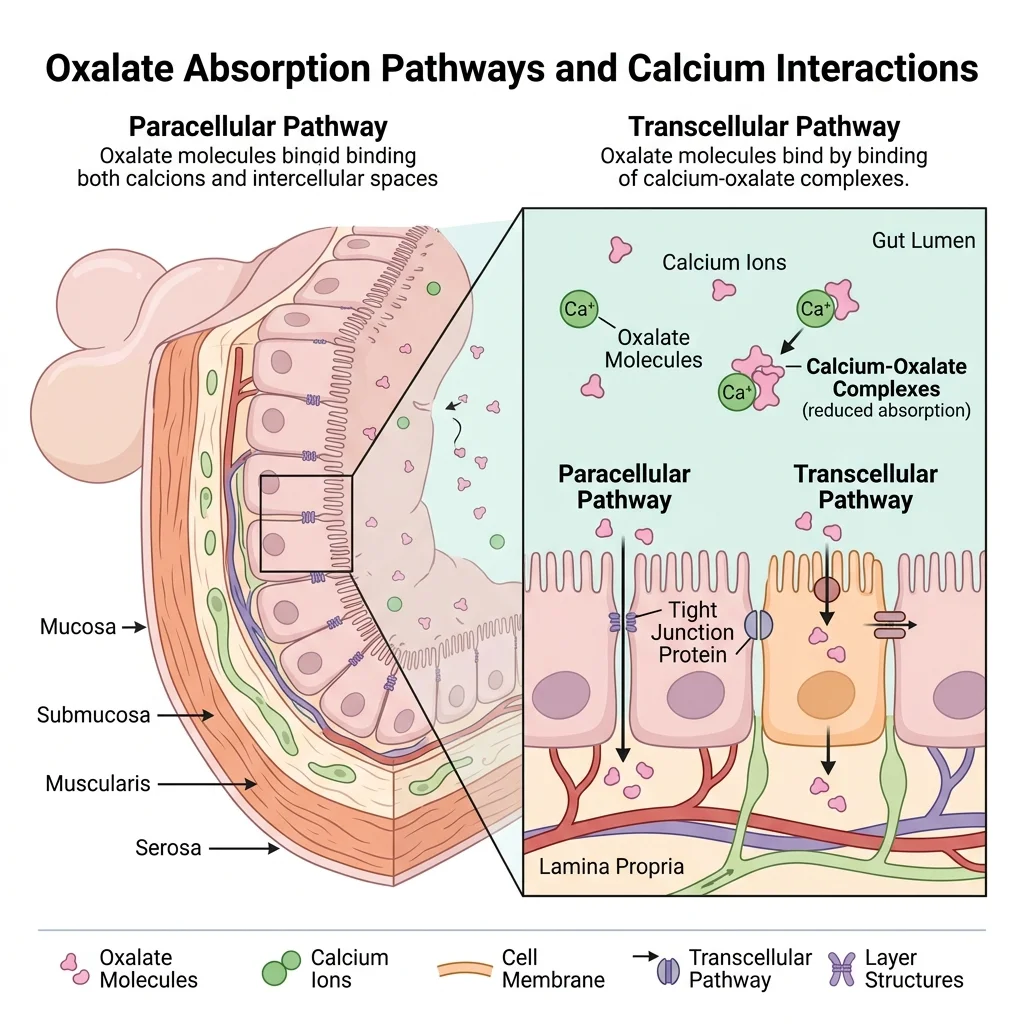
Absorption happens through two routes. The passive paracellular pathway moves oxalate between intestinal cells through tight junctions — this dominates in the colon. The active transcellular pathway uses specialized transport proteins from the SLC26 family to shuttle oxalate through cells. The transporter SLC26A6, found mainly in the small intestine, actually pumps oxalate back into the gut lumen — essentially acting as a safety valve. When researchers knocked out this gene in mice, the animals developed severe hyperoxaluria and kidney stones.
Once absorbed, oxalate travels through the portal circulation to the liver and then distributes systemically. The kidneys filter it from blood and excrete it through the proximal tubule. Plasma oxalate levels peak two to four hours after eating a high-oxalate meal and largely clear within six hours. That short window matters — it means acute spikes in urinary oxalate concentration follow specific meals rather than building up gradually.
The research community initially assumed dietary oxalate contributed only 10 to 20 percent of total urinary oxalate, with the liver generating the rest. Newer controlled feeding studies suggest the real figure may reach 50 percent — a substantially larger dietary influence than previously recognized. This revised estimate strengthens the case for dietary management as a meaningful intervention.
The Gut Microbiome Factor
Your intestinal bacteria play a significant role in managing oxalate levels. The bacterium Oxalobacter formigenes is an obligate anaerobe that uses oxalate as its primary energy source, breaking it down into formate and carbon dioxide. People colonized with O. formigenes show measurably lower urinary oxalate levels, and one study found that colonization decreased the risk of recurrent calcium oxalate stone formation by 70 percent.
The problem is that antibiotics — particularly tetracyclines, macrolides, and fluoroquinolones — readily destroy these bacteria. Patients without O. formigenes show approximately 40 percent higher urinary oxalate levels. In one kidney biopsy study, 52 percent of patients diagnosed with oxalate nephropathy had received antibiotics within the preceding month. While that correlation does not prove causation, it has prompted researchers to investigate whether disruption of the gut microbiome may be an underappreciated trigger for oxalate-related kidney damage.
Beyond O. formigenes, other bacterial species — including certain Bifidobacterium and Lactobacillus strains — also contribute to intestinal oxalate degradation. Maintaining microbial diversity appears to support overall oxalate homeostasis, which is one more reason to think carefully about unnecessary antibiotic use and the health of your intestinal barrier.
Hyperoxaluria: When Oxalate Levels Cross the Line
The clinical definition of hyperoxaluria is straightforward: urinary oxalate excretion exceeding 40 to 45 milligrams per 24 hours. But the condition itself is anything but simple, because the causes span a spectrum from rare genetic disorders to common dietary and surgical factors.
| Type | Cause | Urinary Oxalate | Prevalence |
|---|---|---|---|
| Primary (PH) | Genetic enzyme deficiency (liver) | Often >100 mg/day | 1-3 per million |
| Enteric | Fat malabsorption (surgery, IBD) | Typically 40-80 mg/day | ~250,000 in US |
| Dietary | Excessive high-oxalate food intake | Variable, 40-60 mg/day | Most common form |
| Idiopathic | Unknown mechanism | Variable | Rare |
Primary Hyperoxaluria
Primary hyperoxaluria is a group of three rare autosomal recessive disorders where the liver overproduces oxalate due to defective enzymes. Type 1, caused by mutations in the AGXT gene, accounts for 80 percent of cases and carries the worst prognosis — roughly 80 percent of patients develop end-stage kidney disease by age 30. At the time of diagnosis, 70 percent of adults with PH1 already have significant renal failure. Type 2 (GRHPR mutation) and Type 3 (HOGA1 mutation) follow milder courses, though Type 2 still leads to kidney failure in 20 to 25 percent of patients.
Because the defect sits in the liver, dietary oxalate restriction has limited impact on primary hyperoxaluria. Treatment has historically centered on high-dose pyridoxine (vitamin B6) — effective in about 30 percent of PH1 patients — combined with aggressive hydration and potassium citrate to inhibit crystal formation. That changed in 2020 with the FDA approval of lumasiran, an RNA interference therapy that reduces hepatic oxalate production by an average of 65 percent in PH1, with 84 percent of treated patients achieving normal urinary oxalate levels within six months.
Enteric Hyperoxaluria
Enteric hyperoxaluria develops when gastrointestinal conditions cause fat malabsorption. The mechanism is biochemically simple: undigested fatty acids reaching the colon bind to dietary calcium that would normally trap oxalate. With calcium sequestered by fat, free oxalate absorption can climb from the normal 5 to 10 percent to 30 percent or higher. Bile salts and fatty acids also increase colonic membrane permeability, further boosting uptake.
An estimated 250,000 people in the United States live with enteric hyperoxaluria conditions. Roux-en-Y gastric bypass accounts for about 60 percent of cases, followed by inflammatory bowel disease (20 percent), celiac disease (8 percent), and chronic pancreatitis (4 percent). A meta-analysis of bariatric surgery outcomes found a pooled relative risk of 1.79 for kidney stone formation after Roux-en-Y procedures. Sleeve gastrectomy, which is a restrictive rather than malabsorptive procedure, rarely causes hyperoxaluria — an important distinction for patients weighing surgical options.
Dietary Hyperoxaluria
The most common form results simply from eating too many oxalate-rich foods, especially when calcium intake is low. Reducing dietary calcium from 1,000 to 400 mg per day on a moderate-oxalate diet increases urinary oxalate excretion by 20 to 50 percent depending on gut bacterial colonization status. This interaction explains a counterintuitive clinical finding: low-calcium diets actually raise kidney stone risk rather than lowering it.
80% of Kidney Stones Contain Calcium Oxalate
The relationship between oxalate and kidney stones is not theoretical — calcium oxalate dominates the global kidney stone landscape. Roughly four out of five stones contain it, either as the sole component or mixed with calcium phosphate. And among people who form one calcium oxalate stone, 60 percent will form another within ten years if they do not change their approach to prevention.
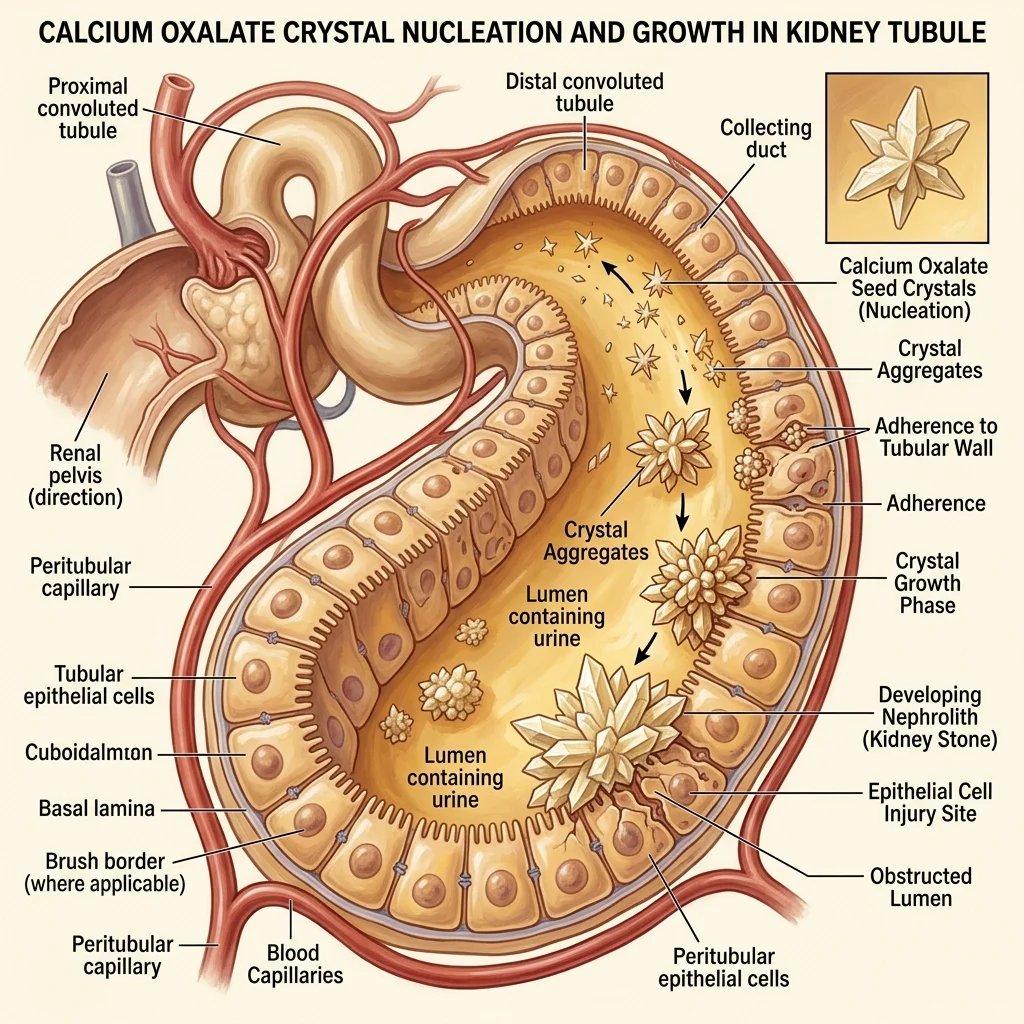
Stone formation follows a defined sequence. First, urinary supersaturation with calcium oxalate allows crystal nucleation — the initial seed of a future stone. Crystals then grow and aggregate, particularly at points where urine flow slows: the bend of the loop of Henle, the tip of the renal papilla, and the openings of collecting ducts. Natural inhibitors like citrate, magnesium, and certain proteins work to prevent aggregation, but when oxalate concentrations overwhelm these defenses, stones form.
Data from three large prospective cohorts following over 240,000 participants revealed that people in the highest quintile of dietary oxalate intake had modestly elevated stone risk (relative risk around 1.2). That sounds reassuring until you look at urinary oxalate data from the same investigators: a three- to four-fold increase in stone risk separated the highest and lowest urinary oxalate groups. The disconnect suggests that what matters most is not how much oxalate you eat but how much actually reaches your urine — which depends on calcium intake, gut absorption efficiency, and microbial degradation.
Stone formers appear biologically different from non-formers in one key respect: their intestines absorb more oxalate. A controlled study using isotope-labeled oxalate measured gut uptake of 9.2 percent in stone-forming patients versus 6.8 percent in healthy controls. That roughly 35 percent higher absorption rate, compounded over years of meals, creates a persistent biochemical disadvantage that dietary modifications can help offset.
Beyond Stones: Oxalate Nephropathy
Kidney stones get most of the attention, but oxalate can damage kidneys without ever forming a visible stone. Oxalate nephropathy — calcium oxalate crystal deposition directly within kidney tissue — was identified in 4 percent of kidney biopsies in a New York City metropolitan study, and roughly half of those patients progressed to end-stage kidney disease. The inflammatory mechanism involves the NLRP3 inflammasome — the same immune pathway implicated in gout and atherosclerosis. When calcium oxalate crystals deposit in tubular cells, they trigger interleukin-1 beta release, attracting macrophages that drive progressive fibrosis.
This connection between oxalate and chronic inflammation extends beyond the kidneys. Mouse models of dietary oxalate-induced chronic kidney disease show cardiac fibrosis and severe arterial hypertension, and human data from the CRIC study (3,123 participants with CKD) found that patients in the highest quintile of urinary oxalate excretion had a 33 percent greater risk of CKD progression and a 45 percent greater risk of reaching end-stage kidney disease compared to the lowest quintile.
Oxalate Myths vs. What the Research Actually Shows
| Common Belief | What the Evidence Says |
|---|---|
| You should avoid all calcium to prevent kidney stones | The opposite is true. Higher dietary calcium binds oxalate in the gut, reducing absorption. The National Kidney Foundation recommends 1,000-1,200 mg of calcium daily from food sources. |
| Spinach is always dangerous | Spinach is the single largest dietary oxalate source (658 mg/100g), but risk depends on how much you eat, what you eat it with, and your individual absorption rate. Pairing it with calcium-rich foods reduces the impact. |
| Cooking eliminates oxalates | Boiling can leach some soluble oxalate into cooking water (reducing content by 30-50% for some vegetables), but it does not eliminate oxalate entirely. Steaming has minimal effect. |
| Oxalate problems are rare and genetic | Primary hyperoxaluria is rare (1-3 per million), but secondary and dietary hyperoxaluria affect far more people. Between 25 and 45 percent of recurrent calcium stone formers have elevated urinary oxalate. |
| Vitamin C is safe at any dose | The body converts excess vitamin C to oxalate. Doses above 1,000 mg per day measurably increase urinary oxalate. The National Kidney Foundation suggests limiting supplemental C to 60 mg daily for stone-prone individuals. |
| If you do not have kidney stones, oxalate does not affect you | Elevated urinary oxalate independently predicts CKD progression and ESKD risk, even in people who never form a visible stone. Crystal deposition can silently damage kidney tissue. |
Practical Strategies to Lower Your Oxalate Load
Managing oxalate does not require eliminating every plant food from your diet. The research points to a handful of high-impact strategies that meaningfully reduce urinary oxalate without stripping nutrition from your meals.
Pair Calcium With Oxalate-Rich Meals
Calcium pairing is the most effective dietary strategy available. When calcium and oxalate meet in the intestinal lumen, they form insoluble complexes that pass through your stool instead of entering your bloodstream. The National Kidney Foundation recommends aiming for 1,000 to 1,200 mg of dietary calcium daily, distributed across meals. A practical target: 300 to 400 mg of calcium with each meal, ideally from dairy or fortified foods eaten alongside any oxalate-containing items.
In one controlled study, reducing dietary calcium from 1,000 to 400 mg per day on a moderate-oxalate diet increased urinary oxalate excretion by 50.3 percent in people lacking O. formigenes colonization. Adequate mineral intake — including both calcium and magnesium — is one of the strongest modifiable factors in oxalate management.
Target the Highest-Oxalate Foods First
Rather than adopting a blanket low-oxalate diet, focus on the foods that deliver the largest doses. A reasonable approach:
| Action | Foods | Rationale |
|---|---|---|
| Avoid or minimize | Spinach, Swiss chard, rhubarb, star fruit, bran cereals | Extremely high oxalate; 50-100g of spinach alone delivers 500-1,000 mg |
| Limit portions | Almonds, peanuts, beets, potatoes, chocolate, soy products | Moderate-to-high; manageable in small amounts especially with calcium |
| Eat freely | Most fruits, cauliflower, broccoli, cabbage, mushrooms, cucumbers | Low oxalate; not clinically significant sources |
Stay Hydrated
Dilution is the simplest defense against crystal formation. Urologists consistently recommend producing at least 2.5 liters of urine daily — which typically requires drinking 3 liters of fluid. Higher urine volume reduces the concentration of both calcium and oxalate, lowering supersaturation levels below the threshold for crystal nucleation.
Monitor Vitamin C Intake
Your body metabolizes excess ascorbic acid into oxalate. While normal dietary vitamin C from fruits and vegetables is not a concern, supplemental doses exceeding 1,000 mg per day reliably increase urinary oxalate. For people with a history of calcium oxalate stones or known hyperoxaluria, keeping vitamin C supplements below 60 mg daily — or eliminating them entirely in favor of food sources — reduces risk without requiring major lifestyle changes.
Support Your Gut Bacteria
Given the role of O. formigenes and other oxalate-degrading bacteria, anything that preserves microbial diversity supports oxalate homeostasis. Avoiding unnecessary antibiotics is the most impactful step. Dietary fiber from varied plant sources feeds the broader microbial ecosystem that supports oxalate-degrading species. Fermented foods and gut-healing dietary choices help maintain the intestinal environment these bacteria need.
Get Tested If Risk Factors Apply
A 24-hour urine collection remains the standard tool for measuring oxalate excretion. Optimal levels sit below 25 mg/day; anything above 40 mg/day warrants investigation. People who should consider testing include recurrent kidney stone formers, anyone who has had Roux-en-Y gastric bypass or other malabsorptive surgery, individuals with inflammatory bowel disease, and those with a family history of primary hyperoxaluria.
Frequently Asked Questions
Can you eat too many oxalates without knowing it?
Yes. Many people consume high amounts of dietary oxalate without recognizing the cumulative load. Green smoothies made with multiple cups of raw spinach, daily almond-heavy snacking, and regular dark chocolate consumption can push dietary oxalate well above 200 mg per day. Because oxalate-related kidney damage can progress silently — without symptoms or visible stones — elevated exposure may go undetected for years until kidney function tests reveal a problem.
Does cooking reduce oxalate in food?
Boiling is the most effective cooking method for reducing soluble oxalate, since it leaches into the cooking water (which you then discard). Depending on the food, boiling can reduce soluble oxalate content by 30 to 50 percent. Steaming and baking are much less effective. Importantly, no cooking method eliminates oxalate entirely — the insoluble crystalline portion remains largely unaffected by heat.
Should people with kidney stones avoid all high-oxalate foods?
Not necessarily. Current evidence supports limiting the very highest sources — spinach, rhubarb, Swiss chard, and star fruit — while managing moderate sources through portion control and calcium pairing. The critical factor is not eliminating oxalate from the diet entirely but preventing excessive amounts from reaching the urine. Eating calcium-rich foods at the same meal neutralizes much of the dietary oxalate before it can be absorbed.
How are antibiotics connected to oxalate problems?
Several antibiotic classes — especially tetracyclines, macrolides, and fluoroquinolones — kill Oxalobacter formigenes and other oxalate-degrading gut bacteria. Loss of these organisms removes a natural defense that can degrade dietary oxalate before absorption. People without O. formigenes colonization show approximately 40 percent higher urinary oxalate, and one study found elevated kidney stone risk beginning three or more months after antibiotic exposure.
Is hyperoxaluria the same as having kidney stones?
No. Hyperoxaluria is a biochemical condition — elevated urinary oxalate above 40 to 45 mg per day — that significantly raises the risk for kidney stones but does not guarantee them. Some people with hyperoxaluria never form visible stones but still develop oxalate nephropathy, a condition where calcium oxalate crystals deposit directly in kidney tissue and cause progressive damage through inflammation and fibrosis.
Related Articles
- Electrolytes: Sodium, Potassium, Magnesium, and Hydration — How mineral balance affects kidney function and stone prevention.
- The Gut-Brain Axis: How Your Microbiome Affects Mood and Cognition — A deeper look at the gut bacteria that play a role in oxalate metabolism.
- Inflammation, Chronic Disease, and Anti-Inflammatory Living — Understanding the NLRP3 inflammasome pathway that oxalate activates.
- Leaky Gut Syndrome: Symptoms, Causes, and Evidence-Based Healing — How intestinal permeability influences oxalate absorption.
- Bone Broth Benefits: Gut Healing, Joints, and Skin — Gut-supportive nutrition that can complement oxalate management strategies.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.