Your Muscles Are Aging — And a Pomegranate Byproduct Might Slow It Down
Muscle loss starts earlier than most people realize. By age 40, the average person loses roughly 8% of their muscle mass per decade, and that rate accelerates after 70. The underlying problem is not just fewer muscle fibers — it is a breakdown in the mitochondria that power those fibers. As these organelles accumulate damage over time, muscle cells cannot produce the force or endurance they once could.
Urolithin A is a compound that targets this specific problem. It is not a vitamin, mineral, or herb — it is a postbiotic metabolite first identified in 2016 by researchers at the Swiss Federal Institute of Technology (EPFL) as a "first-in-class natural compound that induces mitophagy both in vitro and in vivo." In plain terms, urolithin A triggers the body's own cleanup system for damaged mitochondria, allowing healthy ones to take their place.
What makes urolithin A unusual is its origin. It does not come directly from any food. Instead, it is produced inside the gut when certain bacteria break down ellagitannins — polyphenols found in pomegranates, walnuts, raspberries, and strawberries. The catch: your body's ability to make urolithin A depends entirely on whether you harbor the right gut bacteria. And as we will see, most people do not.
The Gut Microbiome Bottleneck That Limits Urolithin A Production
Eating a pomegranate does not guarantee urolithin A will show up in your bloodstream. The conversion requires a specific sequence of microbial reactions in the colon, and research on ellagitannin metabolism has documented "a large interindividual variability" in how effectively people produce urolithins from dietary precursors.
The process works like this: ellagitannins from food are hydrolyzed in the gut into ellagic acid. From there, specialized colonic bacteria perform a series of chemical transformations — first removing one of two lactone rings, then progressively stripping hydroxyl groups — to produce a cascade of intermediate compounds. The end product of this cascade, and the most biologically active one, is urolithin A.
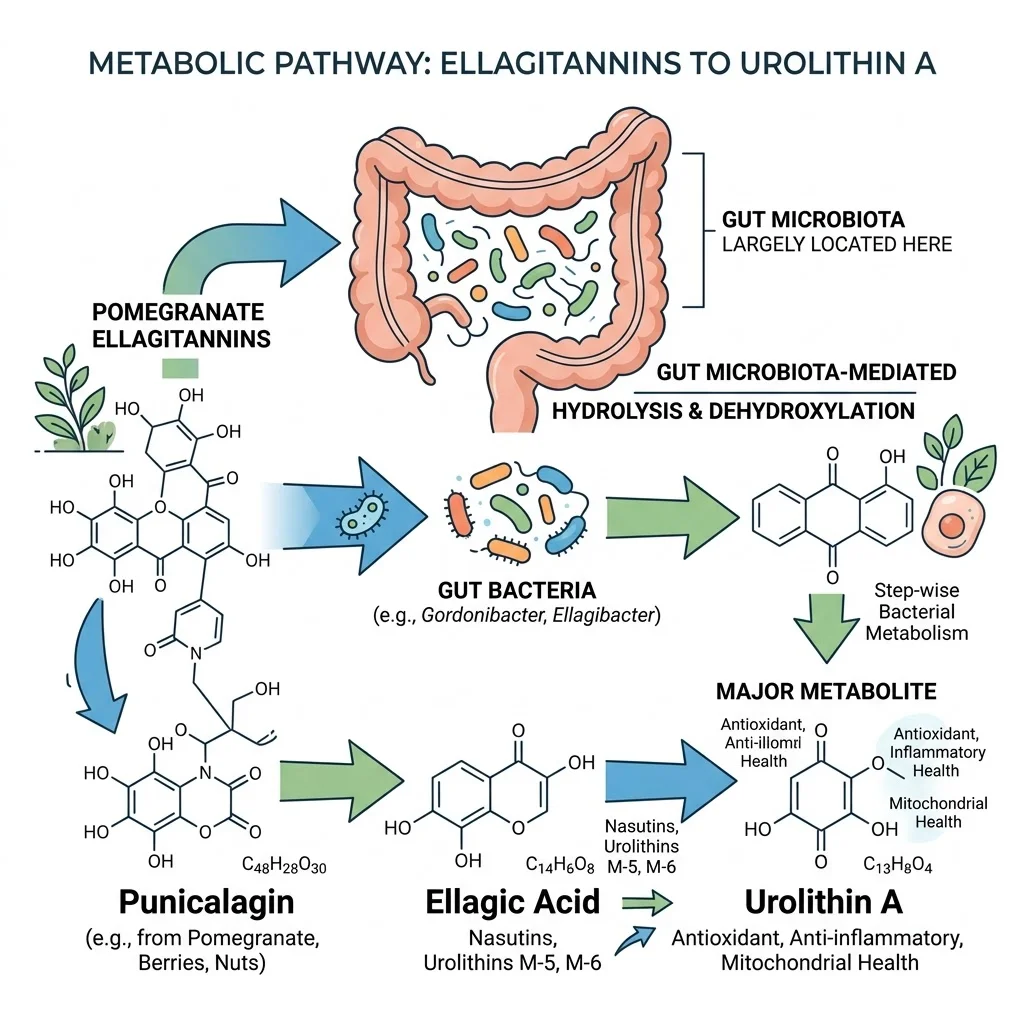
Population studies have classified people into three distinct urolithin metabotypes based on what their gut bacteria actually produce:
| Metabotype | What It Means | Estimated Prevalence |
|---|---|---|
| UM-A | Produces urolithin A as the primary metabolite | ~40% of population |
| UM-B | Produces urolithin B and isourolithin A instead | ~25-30% of population |
| UM-0 | Produces little to no urolithins at all | ~30-35% of population |
Research published in Clinical Nutrition found that these metabotypes are not just academic classifications — they correlate with real health differences. People in the UM-A group showed positive associations with apolipoprotein A-I (a marker of healthy cholesterol metabolism), while the UM-B pattern was linked to less favorable lipid profiles in overweight individuals.
The practical takeaway: roughly 60% of people either produce the wrong type of urolithin or produce none at all when they eat ellagitannin-rich foods. This is not a reflection of diet quality — it is a reflection of which bacterial species colonize your particular gut. The bacteria required for urolithin A production are specialized strains distinct from those that metabolize other polyphenol classes, and their presence varies based on factors including age, geography, antibiotic history, and overall microbiome composition.
The Cellular Recycling System That Keeps Muscles Young
To understand why urolithin A matters for aging muscles, you need to understand mitophagy — and why it fails as we get older.
Every muscle cell contains hundreds to thousands of mitochondria. These organelles are not permanent structures; they have a lifespan, and when they accumulate damage from normal metabolic activity, they need to be dismantled and replaced. The process of selectively tagging and removing dysfunctional mitochondria is called mitophagy. Think of it as a quality control system: when a mitochondrion starts leaking reactive oxygen species or can no longer produce ATP efficiently, the cell marks it for destruction and recycles its components to build fresh ones.
Key concept: Mitophagy is not the same as general autophagy. While autophagy recycles any damaged cellular component, mitophagy specifically targets mitochondria. Urolithin A activates mitophagy through the PINK1-Parkin signaling pathway — the same pathway that malfunctions in Parkinson's disease.
In young, healthy muscle tissue, this turnover happens continuously and efficiently. But with aging, the mitophagy machinery slows down. Damaged mitochondria accumulate, energy production drops, and the muscle cell's ability to contract forcefully and recover from exertion declines. This is not just a theory — biopsies from aging skeletal muscle consistently show higher proportions of dysfunctional mitochondria compared to younger tissue.
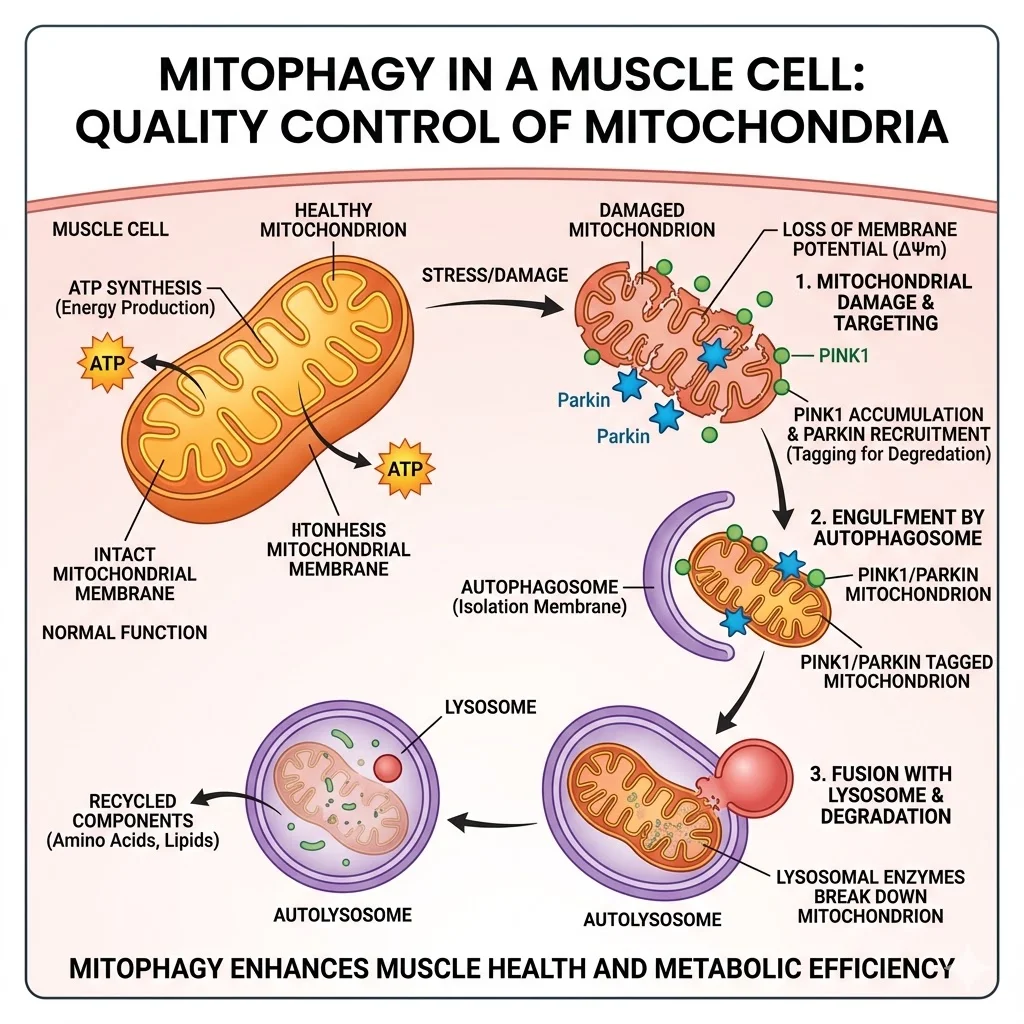
Urolithin A intervenes at precisely this point. The compound activates mitophagy through both PINK1-Parkin dependent and independent pathways. In the Parkin-dependent route, urolithin A increases phosphorylation of Parkin protein at serine 65 — a direct molecular switch that tags damaged mitochondria for removal. In clinical studies, muscle biopsies from participants taking urolithin A showed enrichment of Parkin-mediated ubiquitin and proteasomal systems, confirming that this pathway is activated in living human tissue, not just in laboratory cells.
The preclinical evidence for this mechanism is striking. In the original Nature Medicine study, worms given urolithin A showed prevented accumulation of dysfunctional mitochondria with age and extended lifespan while maintaining mobility. Aged mice (23 months old, equivalent to roughly 70 human years) showed a 9% increase in grip strength and a 57% increase in spontaneous physical activity — without any change in exercise or diet. Dystrophic mice, which model severe muscle wasting, also showed improved grip strength and running capacity.
This mechanism differs from how most supplements claim to work. Urolithin A does not provide extra energy or stimulate growth hormones — it restores a cellular maintenance process that deteriorates with age. The muscle strengthens because its power generation infrastructure improves. This is similar in principle to other autophagy-activating compounds like spermidine, though the molecular targets differ.
What the Human Trials Actually Show
The human evidence for urolithin A now spans five published clinical trials covering different populations and endpoints. The results are promising but nuanced — this is not a story where everything worked perfectly.
The First-in-Human Safety Trial (2019)
The first trial in healthy elderly volunteers, published in Nature Metabolism, established that oral urolithin A at 500 mg and 1,000 mg doses was safe, well-absorbed, and produced measurable molecular changes. After four weeks of supplementation, participants showed altered plasma acylcarnitine profiles and changes in skeletal muscle mitochondrial gene expression — a "molecular signature of improved mitochondrial and cellular health," as the authors put it. The compound peaked in plasma 6-8 hours after ingestion and had a half-life of 17-22 hours, meaning once-daily dosing maintained steady levels.
The ATLAS Trial: Middle-Aged Adults (2022)
The ATLAS trial, published in Cell Reports Medicine, randomized 88 untrained, overweight adults aged 40-64 to receive placebo, 500 mg, or 1,000 mg of urolithin A daily for four months. This trial produced the strongest functional results to date.
| Outcome | 500 mg/day | 1,000 mg/day | Placebo |
|---|---|---|---|
| Hamstring strength change | +12% (p=0.027) | +9.8% (p=0.029) | No significant change |
| Peak VO2 change | Not significant | +10% | No significant change |
| 6-minute walk distance | Improved | +33 meters | No significant change |
| Phospho-Parkin (mitophagy marker) | Significantly increased | Significantly increased | No change |
The molecular data from ATLAS were particularly convincing. Muscle biopsies showed that urolithin A increased levels of mitochondrial respiratory chain proteins (NDUFB8, SDHB) at the 1,000 mg dose and activated the Parkin-mediated mitophagy pathway at 500 mg. This provided direct tissue-level evidence that the proposed mechanism was actually operating in human skeletal muscle.
The ENERGIZE Trial: Older Adults (2022)
The ENERGIZE trial, published in JAMA Network Open, tested 1,000 mg daily in 66 adults aged 65-90 for four months. Here the results were more mixed. The primary endpoints — six-minute walk distance and maximal ATP production measured by magnetic resonance spectroscopy — did not reach statistical significance versus placebo. Both the treatment and placebo groups improved more than expected on the walk test (60.8 meters vs. 42.5 meters), which the authors attributed to a stronger-than-anticipated placebo effect in this motivated elderly population.
However, secondary outcomes told a different story. At the two-month mark, muscle endurance measured by contractions until fatigue was dramatically better in the urolithin A group:
| Muscle | Urolithin A (contractions gained) | Placebo (contractions gained) |
|---|---|---|
| Hand (first dorsal interosseous) | +95.3 | +11.6 |
| Leg (tibialis anterior) | +41.4 | +5.7 |
Biomarker changes were also favorable: C-reactive protein decreased from 2.14 to 2.07 mg/L in the urolithin A group while increasing from 2.17 to 2.65 mg/L in the placebo group. Plasma ceramides (lipids associated with cardiovascular risk) dropped significantly, as did long-chain acylcarnitines, suggesting improved mitochondrial fat-burning efficiency. The inflammation reduction was noteworthy because chronic low-grade inflammation is itself a driver of age-related muscle loss.
Athletic Populations: Younger and Fitter
Two recent trials extended the research beyond elderly and sedentary populations. A study of 42 competitive distance runners (average age 27.2, VO2max 66.4 mL/kg/min) found that four weeks of 1,000 mg daily urolithin A increased VO2max by 5.4% compared to 3.6% in the placebo group (p=0.009) during altitude training. Post-race creatine kinase levels — a marker of muscle damage — were also significantly lower in the treatment group. However, actual race times in a 3,000-meter time trial did not differ significantly.
A pilot trial in 20 academy soccer players (mean age 17.5) showed more pronounced performance effects: six weeks of 1,000 mg daily urolithin A improved aerobic endurance by 239 meters on the Yo-Yo intermittent recovery test (p=0.048) and increased countermovement jump height by 3.33 cm (p=0.020).
What Urolithin A Does Outside of Skeletal Muscle
While the muscle aging story dominates the research, mitochondria are not unique to muscle cells. Every tissue in the body depends on mitochondrial function, and urolithin A's mitophagy-activating effects appear to extend beyond the musculoskeletal system.
Immune System Rejuvenation
A randomized trial of 50 middle-aged adults (ages 45-70) tested whether just 28 days of 1,000 mg daily urolithin A could influence immune cell aging. The results were notable: urolithin A expanded naive-like CD8+ T cells — the immune cells that respond to new threats — while reducing markers of terminal exhaustion. Single-cell RNA sequencing confirmed that the compound upregulated stemness genes (TCF7, LEF1) in immune cells while suppressing exhaustion-associated genes.
Perhaps more relevant to daily health, monocytes in the treatment group showed improved bacterial phagocytosis — their ability to engulf and destroy pathogens. The enhanced fatty acid oxidation in CD8+ cells (a treatment difference of 14.72 percentage points, p=0.006) suggests that urolithin A may help immune cells maintain their metabolic fitness as we age.
Joint and Cartilage Protection
Preclinical research has shown that urolithin A reduces pro-inflammatory cytokines in human chondrocytes (cartilage cells) while simultaneously increasing their mitochondrial respiration and ATP production. For anyone dealing with age-related joint stiffness, the dual action of reducing inflammation while improving cellular energy in cartilage tissue is a compelling combination. This overlaps with strategies like consuming collagen-rich foods for joint support, though the mechanisms are distinct — urolithin A works at the mitochondrial level rather than providing structural raw materials.
Bone Density
Urolithin A appears to influence bone remodeling through a dual mechanism: promoting osteoblast differentiation (the cells that build new bone) while inhibiting osteoclast-mediated bone resorption (the cells that break bone down). This has particular relevance for aging populations where bone density loss and sarcopenia often co-occur — addressing both simultaneously would be more efficient than treating each independently.
Cardiovascular Markers
Across multiple trials, urolithin A has consistently reduced circulating ceramides — lipid molecules increasingly recognized as independent cardiovascular risk factors. The ENERGIZE trial documented significant reductions in C16 and C18 ceramide species, and the metabotype research found that natural urolithin A producers have more favorable apolipoprotein profiles. While no trial has yet tested cardiovascular events directly, these biomarker shifts point in a promising direction.
The Practical Question: How to Actually Get Enough Urolithin A
Given that most people cannot produce adequate urolithin A from food alone, there are two distinct strategies — and they are not equally effective for everyone.
The Dietary Approach: Ellagitannin-Rich Foods
Foods highest in the precursor ellagitannins include:
- Pomegranates — the richest dietary source, particularly the juice and arils
- Walnuts — contain significant ellagitannins along with omega-3 fats
- Raspberries — moderate ellagitannin content
- Strawberries — lower but still meaningful levels
The limitation is straightforward: if you are in the ~60% of people whose gut bacteria cannot efficiently convert these polyphenols into urolithin A, eating more pomegranates will not solve the problem. There is currently no reliable consumer test to determine your metabotype, though researchers have used urine analysis after pomegranate consumption in study settings.
Direct Supplementation
Synthetic urolithin A supplements bypass the gut microbiome bottleneck entirely. The compound used in all major clinical trials is Mitopure (manufactured by Amazentis SA), which delivers pure urolithin A without requiring bacterial conversion. This is the approach that has been tested in the trials described above, and it is the only method where specific dosages and outcomes have been validated.
Clinical data points on dosing:
| Dose | Evidence Level | Key Findings |
|---|---|---|
| 250 mg/day | Tested, minimal effects | Below therapeutic threshold in most studies |
| 500 mg/day | Effective | Significant hamstring strength gains, mitophagy activation in biopsies |
| 1,000 mg/day | Most tested | Consistent across trials for strength, endurance, and biomarker improvements |
| 2,000 mg/day | Safety tested | Safe but no additional benefit demonstrated over 1,000 mg |
The pharmacokinetics favor once-daily dosing. Urolithin A reaches peak plasma concentration at approximately 6 hours and has a half-life of 17-22 hours, meaning a single morning dose maintains effective levels throughout the day. Bioavailability is unaffected by food co-consumption, so timing relative to meals is not critical.
One important note about the supplement market: urolithin A received Generally Recognized as Safe (GRAS) status from the FDA, but this does not mean all products labeled "urolithin A" have been through clinical testing. The specific formulation used in published trials (Mitopure) is distinct from generic urolithin A products, and bioequivalence between brands has not been established. As with any supplement, the gap between what clinical trials demonstrate and what the consumer market offers can be significant.

Frequently Asked Questions
Is urolithin A the same as pomegranate extract?
No. Pomegranate extract contains ellagitannins and ellagic acid — the precursors to urolithin A. Whether those precursors get converted into urolithin A depends on your gut bacteria. Urolithin A supplements provide the final active compound directly, skipping the microbial conversion step. Only about 40% of people efficiently produce urolithin A from pomegranate or its extract.
How long does it take for urolithin A to produce noticeable effects?
Molecular changes (mitophagy biomarkers, acylcarnitine profiles) appear within the first four weeks of supplementation at 500-1,000 mg daily. Functional improvements in muscle endurance have been measured as early as two months. The immune cell remodeling study showed changes after just 28 days, though the clinical significance of short-term immune shifts requires further study.
Are there side effects from urolithin A supplementation?
Across all published clinical trials (totaling over 250 participants), urolithin A at doses up to 2,000 mg daily showed a safety profile comparable to placebo. No serious adverse events have been attributed to the compound. Minor adverse events (mostly upper respiratory infections) occurred at similar rates in both treatment and placebo groups. Kidney and liver function markers remained within normal ranges throughout all studies.
Can urolithin A replace exercise for maintaining muscle health?
No. While urolithin A improved muscle strength and endurance in sedentary participants without requiring exercise, the magnitude of those improvements (10-12% strength gains) is smaller than what regular resistance training achieves. The compound addresses mitochondrial quality — one component of muscle health — but does not stimulate the mechanical loading, neuromuscular adaptation, or hormonal responses that exercise provides. Think of it as complementary, not a substitute.
Should I take urolithin A if I already eat pomegranates regularly?
That depends on your gut microbiome — specifically, whether you fall into the UM-A metabotype that efficiently converts dietary ellagitannins to urolithin A. Since there is no widely available consumer test for this, direct supplementation is the only guaranteed route to consistent urolithin A levels. Eating pomegranates and walnuts remains beneficial for other nutritional reasons (fiber, other polyphenols, healthy fats) regardless of your metabotype.
Related Articles
- Spermidine: The Longevity Compound Hidden in Wheat Germ — Another autophagy activator with overlapping longevity benefits and a different molecular mechanism.
- The Gut-Brain Axis: How Your Microbiome Affects Mood and Cognition — A deeper look at how gut bacteria influence health far beyond digestion.
- Inflammation, Chronic Disease, and Anti-Inflammatory Living — Understanding the low-grade inflammation that drives both muscle aging and cardiovascular risk.
- Bone Broth Benefits for Gut Healing, Joints, and Skin — Complementary joint and gut support strategies from a dietary perspective.
- Rapamycin: The Longevity Anti-Aging Drug — Another compound targeting cellular aging through a different longevity pathway.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.












