22% Longer Life From a Diabetes Pill Nobody Was Watching
Rapamycin got the headlines. Metformin got a $75 million clinical trial. But while the longevity community argued about which of those two drugs would become the first FDA-approved anti-aging medicine, a quieter shift was happening in the data. Two classes of metabolic drugs — one that blocks sugar absorption in the gut, another that flushes glucose out through urine — were racking up results that neither rapamycin nor metformin could match in certain dimensions.
The NIA's Interventions Testing Program (ITP) has now validated 15 individual agents and 2 drug combinations that significantly extend median lifespan in genetically diverse mice. Acarbose and canagliflozin (an SGLT2 inhibitor) are both on that list. Rapamycin is too — but rapamycin comes with a problem that the other two largely avoid.
Rapamycin suppresses the immune system. That is its day job. It was approved to prevent organ transplant rejection, and its side-effect profile reflects that origin. A 2025 review in Frontiers in Aging by Roark and Iffland noted that the EXIST-3 trial documented stomatitis, infections, hyperlipidemia, and cytopenias as common adverse events with everolimus (a rapamycin analog). The long-term safety of chronic mTOR inhibition in otherwise healthy people remains unknown. Bryan Johnson, the tech entrepreneur who famously spent millions on his anti-aging protocol, publicly discontinued rapamycin after experiencing elevated blood glucose, increased susceptibility to infection, and impaired wound healing.
Metformin faces a different limitation. The Targeting Aging with Metformin (TAME) trial — a 6-year study of 3,000 non-diabetic adults aged 65 to 79 — was designed to test whether metformin slows aging beyond its glucose-lowering effects. But metformin does not actually reduce caloric intake. As Prattichizzo and colleagues pointed out in a 2022 review, none of the commonly studied calorie restriction mimetics produce a genuine decrease in calories. SGLT2 inhibitors are the exception.
SGLT2 inhibitors also carry something no other geroprotector candidate can match: the largest body of randomized controlled trial data in humans. A 2023 review by O'Keefe and colleagues described them as having "the most RCT data for safety and efficacy in humans" among all candidate geroprotective agents, including metformin and rapamycin.
How SGLT2 Inhibitors Starve Zombie Cells Into Silence
Senescent cells earned the nickname "zombie cells" because they stop dividing but refuse to die. They sit in tissues — heart, kidney, brain, vasculature — pumping out inflammatory molecules through what researchers call the senescence-associated secretory phenotype, or SASP. The SASP drives chronic inflammation, accelerates tissue deterioration, and contributes to cardiovascular disease, kidney failure, neurodegeneration, and cancer. Removing or silencing these cells is the goal of an entire drug class called senotherapeutics.
SGLT2 inhibitors were not designed for this purpose. They were built to lower blood sugar by blocking a protein called SGLT2 in the kidneys. Think of SGLT2 as a recycling pump: it normally grabs glucose from urine and sends it back into the bloodstream. SGLT2 handles 80 to 90 percent of all renal glucose reabsorption. When you inhibit that pump, glucose leaves the body through urine instead — roughly 60 to 100 grams per day, which translates to a caloric deficit of approximately 200 to 300 kilocalories daily.
What happened next was not on the label. The EMPA-REG trial of 7,020 patients found that empagliflozin reduced cardiovascular death by 38 percent. A meta-analysis pooling 21 randomized trials and over 70,000 patients showed a 14 percent reduction in all-cause mortality. In a real-world study of roughly 160,000 newly diagnosed diabetics, those prescribed an SGLT2 inhibitor had 45 percent lower all-cause mortality than those who were not. These benefits appeared within days of starting the drug, far too quickly to be explained by modest improvements in blood sugar or weight alone.
The senescence connection emerged from preclinical work. A 2025 review by Yesilyurt-Dirican and colleagues in npj Aging compiled every study testing SGLT2 inhibitors against cellular senescence. The list spans endothelial cells, cardiomyocytes, kidney tubular cells, brain tissue, liver, skeletal muscle, pancreatic beta cells, and bone marrow stem cells. In each model, SGLT2 inhibitors reduced markers of senescence — SA-beta-galactosidase, p16, p21, p53 — and dampened the SASP.
This is not just about lowering blood sugar. Prattichizzo's team showed that glimepiride, a sulfonylurea that lowers glucose through insulin secretion, could not reproduce the anti-senescence effects of SGLT2 inhibitors even at equivalent glycemic control. Separately, Katsuumi and colleagues found that canagliflozin eliminated senescent cells from visceral fat in obese mice — an effect that insulin treatment, despite normalizing glucose, did not achieve. The mechanism appears to work through metabolic reprogramming, not glucose reduction per se.
Why would blocking a kidney glucose transporter kill zombie cells? One hypothesis from Prattichizzo's group has a plausible answer: senescent cells are metabolically hyperactive. They depend heavily on glucose to fuel their inflammatory secretory program. Cutting their glucose supply — whether through the kidney or through recently discovered SGLT2 expression in non-renal tissues — may starve them into quiescence or trigger their elimination.
The Drug That Extended Male Mouse Lifespan Three Times Running
Acarbose has the most replicated lifespan extension result in the entire ITP catalog. The program tested it three separate times, across three independent sites each time, and the drug delivered every round.
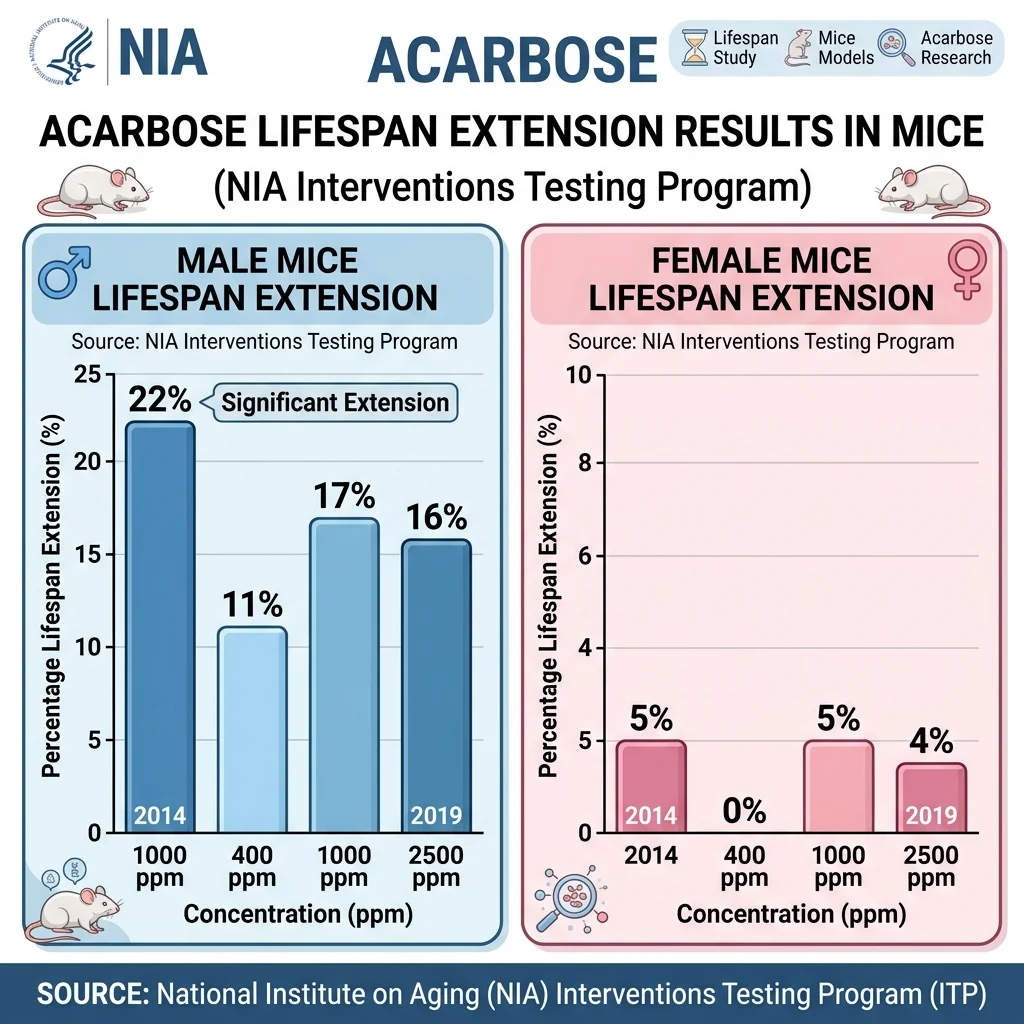
In the original 2014 study led by Dr. David Harrison at The Jackson Laboratory, acarbose at 1,000 parts per million increased male median lifespan by 22 percent (P less than 0.0001). Female median lifespan increased by 5 percent (P = 0.01). Maximum lifespan — measured at the 90th percentile — rose 11 percent in males and 9 percent in females.
The 2019 follow-up tested three doses: 400, 1,000, and 2,500 ppm. All three extended lifespan in both sexes, with males again showing the larger effect — 16 to 17 percent median increases at the two higher doses. There were no significant differences between doses, suggesting the drug hits a ceiling early.
Acarbose works nothing like rapamycin or metformin. It is an alpha-glucosidase inhibitor — it slows the breakdown of complex starches and sugars in the gut, flattening the postprandial glucose spike that follows every meal. It does not sequester glucose or block absorption entirely. It just delays digestion, spreading glucose entry into the bloodstream over a longer window.
The result resembles calorie restriction in some ways: both reduce body weight, body fat, and glucose dysregulation. But there is a telling difference. Mice on acarbose actually eat more food, not less. Calorie restriction extends lifespan equally in males and females; acarbose preferentially benefits males. These distinctions suggest acarbose is working through a mechanism that overlaps with but is not identical to simple caloric reduction.
The health effects extend beyond lifespan. In the 2019 ITP study, male mice on acarbose had roughly half the lung tumors of controls. Both sexes showed reduced liver degeneration. Female mice had less glomerulosclerosis. Aging females on acarbose performed better on the rotarod — a test of balance and coordination — after training, though males did not show this benefit.
One result stands out as genuinely puzzling: acarbose reduces body weight and fat more in females than in males, yet extends lifespan more in males. The original ITP paper explicitly stated that the sexual dimorphism in lifespan effect "could not be explained by differences in effects on weight." Blood glucose responses to refeeding were significantly reduced in males (P less than 0.002) but not females, suggesting the drug's metabolic effects are processed differently by sex in ways researchers have not yet untangled.
The ITP has also tested rapamycin combined with acarbose as one of its two validated combination regimens, a nod to the possibility that stacking mechanisms — mTOR inhibition plus postprandial glucose control — might yield additive benefits.
One Metabolic Switch Runs Through Both Drugs
Acarbose blunts glucose spikes from above. SGLT2 inhibitors drain glucose from below. The entry points differ, but both drugs converge on the same set of cellular energy sensors — and those sensors are the ones that longevity researchers have been targeting for decades.
When cells detect that glucose and nutrients are scarce, they activate a cascade of survival programs. AMPK (AMP-activated protein kinase) switches on, ramping up energy production and fat burning. SIRT1 and related sirtuins activate, promoting DNA repair and inflammation control. mTOR — the growth-promoting pathway that rapamycin inhibits — dials down. The net effect is a shift from "grow and divide" to "repair and conserve." This is, in broad strokes, what calorie restriction does, and it is why calorie-restricted animals live longer across dozens of species.
SGLT2 inhibitors trigger this cascade by forcing the body to lose approximately 200 kilocalories per day through urinary glucose excretion. The body compensates by burning fat stores and producing ketone bodies — primarily beta-hydroxybutyrate — in the liver. O'Keefe and colleagues documented that this metabolic shift simultaneously upregulates AMPK, SIRT1, SIRT3, SIRT6, and PGC-1alpha while downregulating mTOR signaling. In one striking finding, SGLT2 inhibitors were shown to bind mTOR at the same structural domain that rapamycin uses — raising the possibility that these diabetes drugs are, at the molecular level, doing part of what rapamycin does without the immunosuppression.
The mitochondrial effects are where this gets concrete. Aging cells accumulate damaged mitochondria — the organelles that produce cellular energy. Think of it as a factory running on malfunctioning generators: output drops, waste products (reactive oxygen species) accumulate, and the whole operation degrades. SGLT2 inhibitors appear to reset this machinery. Prattichizzo's group documented that these drugs restore the balance between mitochondrial fusion and fission proteins — normalizing levels of Mfn1, Mfn2, Opa1, and Drp1. Empagliflozin specifically suppressed excessive mitochondrial fission through an AMPK-dependent mechanism, preventing the fragmentation of mitochondrial networks that characterizes aged and diseased cells.
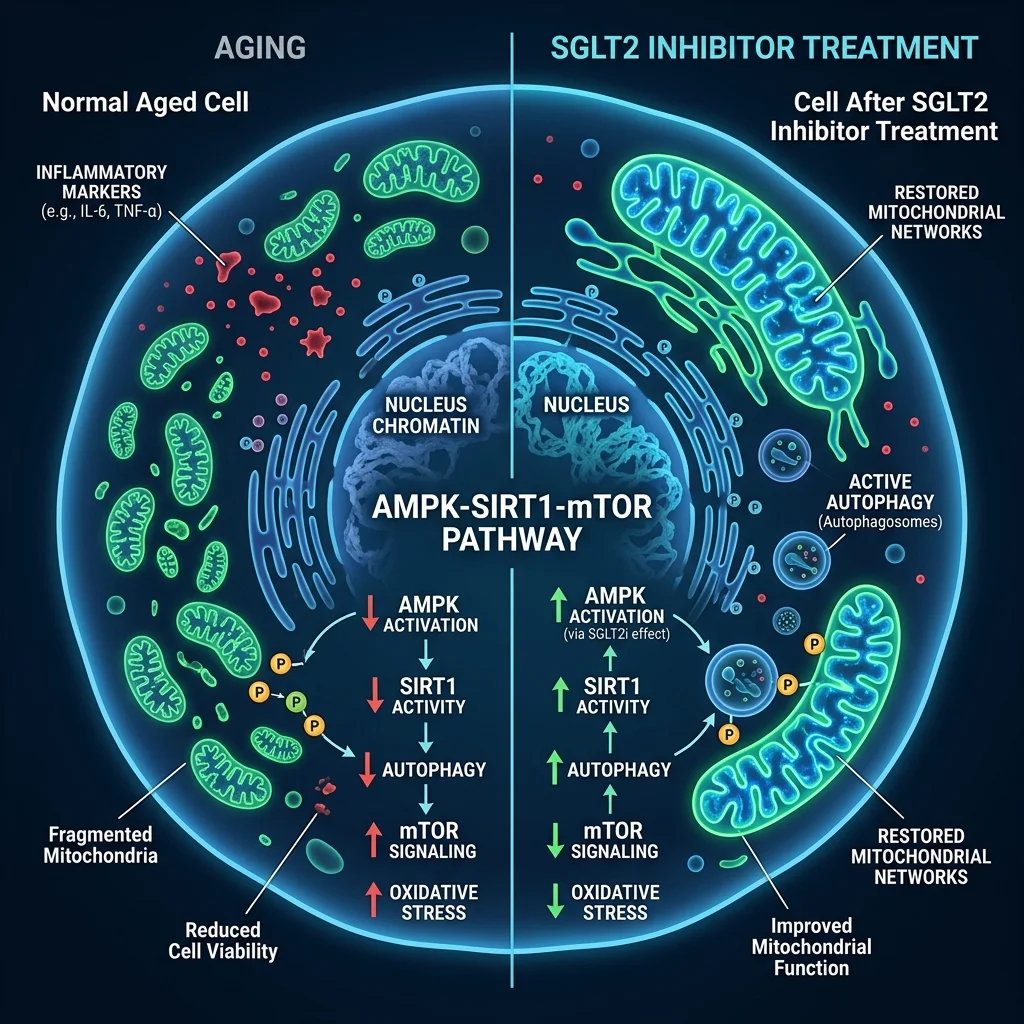
Ketones do more than serve as backup fuel. When SGLT2 inhibitors shift the body toward ketone production, the ketones themselves become signaling molecules. Beta-hydroxybutyrate directly deactivates the NLRP3 inflammasome, a molecular platform that drives the chronic, low-grade inflammation — called inflammaging — that accelerates every major age-related disease. In a pilot trial cited by Prattichizzo's team, macrophages collected from patients on SGLT2 inhibitors secreted less IL-6 and IL-1-beta than macrophages from patients on other glucose-lowering drugs at equivalent glycemic control. IL-6 has been called "the cytokine for gerontologists" because its circulating levels predict frailty, cardiovascular events, diabetes, and all-cause mortality in aging cohorts.
Acarbose likely converges on these same nutrient-sensing pathways through its own route: by flattening postprandial glucose spikes, it reduces the insulin surges that activate mTOR and suppresses the metabolic stress signals that drive inflammation. The ITP's validation of rapamycin plus acarbose as a combination suggests that targeting mTOR from multiple angles — direct inhibition and metabolic modulation — may compound the benefits.
What Longevity Doctors Are Actually Writing on Prescription Pads
The gap between research and clinical practice in longevity medicine is narrowing faster than regulators can keep up. Metformin and rapamycin were the first drugs to cross from laboratory curiosity to off-label longevity prescriptions. O'Keefe's review noted that both are already being prescribed off-label to slow aging "despite a paucity of evidence showing safety and efficacy" for that purpose. SGLT2 inhibitors and acarbose are now joining them.
Telehealth longevity platforms have accelerated this trend. AgelessRx, a longevity-focused telehealth provider, currently offers canagliflozin (Invokana) and bexagliflozin (Brenzavvy, FDA-approved in 2023) to patients seeking metabolic optimization for aging. Their model — a free medical review with a licensed prescriber, followed by ongoing monitoring — represents the expanding infrastructure of longevity medicine that operates outside traditional diabetes care.
The regulatory landscape for these drugs favors access. All major SGLT2 inhibitors — empagliflozin (Jardiance), dapagliflozin (Farxiga), canagliflozin (Invokana) — are FDA-approved, widely available, and covered by most insurance plans for their on-label indications in diabetes, heart failure, and chronic kidney disease. Acarbose (Precose) has been on the market since the 1990s and is available as a low-cost generic. Any physician can legally prescribe these drugs off-label for longevity purposes, though the patient typically bears the cost when the prescription does not match an approved indication.
| Drug | Class | FDA-Approved For | ITP Lifespan Result | Off-Label Longevity Use |
|---|---|---|---|---|
| Canagliflozin (Invokana) | SGLT2 inhibitor | T2D, CKD | 14% male median extension | Growing — telehealth platforms |
| Empagliflozin (Jardiance) | SGLT2 inhibitor | T2D, HF, CKD | Not tested in ITP | Most prescribed SGLT2i; strong CV data |
| Dapagliflozin (Farxiga) | SGLT2 inhibitor | T2D, HF, CKD | Not tested in ITP | Broad indications; renal focus |
| Acarbose (Precose) | Alpha-glucosidase inhibitor | T2D | 22% male median extension (highest) | Niche — longevity physicians |
| Rapamycin (Sirolimus) | mTOR inhibitor | Transplant rejection, TSC | Extended both sexes | Established but controversial |
| Metformin | Biguanide | T2D | Not a standalone ITP success | Most widely used; TAME trial pending |
What is missing is a dedicated clinical trial. The TAME trial for metformin was designed specifically to test anti-aging properties in non-diabetic individuals, and it has been held up as a template for how longevity medicine could earn mainstream credibility. No equivalent trial exists for SGLT2 inhibitors or acarbose in a purely aging-focused context. The existing human evidence for SGLT2 inhibitors comes from cardiovascular and renal outcome trials that happened to measure mortality — and the mortality reductions were so large and consistent that longevity researchers took notice.
The Fine Print: Side Effects, Sex Gaps, and Missing Human Data
SGLT2 inhibitors are well-tolerated as diabetes drugs go, but "well-tolerated for diabetes management" and "safe enough for healthy people to take for decades" are different bars.
The most common side effect is genital yeast infections. A large meta-analysis found a 3.3-fold increased risk — 6.3 percent of SGLT2 inhibitor users versus 1.7 percent of controls. These infections are generally mild and treatable, but they are a recurring nuisance that drives some patients to discontinue the drug.
The serious risks concentrate in diabetic populations. StatPearls reports a nearly 3-fold increased risk of diabetic ketoacidosis, with canagliflozin carrying the highest risk followed by empagliflozin and dapagliflozin. Euglycemic DKA — ketoacidosis with normal-looking blood sugar — is a particular concern because it can be missed. For non-diabetics, however, the risk profile changes dramatically: only a single starvation ketoacidosis event was reported in non-diabetic individuals across approximately 30,000 patient-years of trial follow-up. Discontinuation rates in trials are similar to placebo.
For nondiabetic individuals, SGLT2 inhibitor therapy virtually never causes ketoacidosis, lower limb amputation, or Fournier's gangrene. — O'Keefe et al., Progress in Cardiovascular Diseases, 2023
The amputation concern with canagliflozin made headlines but has since been downgraded: the FDA removed its boxed warning in 2020 based on updated safety data, and a large meta-analysis showed no class-wide amputation risk. Bone fracture risk from the CANVAS trial was not consistently observed with empagliflozin or dapagliflozin.
The sex gap is the elephant in the room for both drugs. Acarbose extends male mouse lifespan by 16 to 22 percent but only 4 to 5 percent in females — a disparity that weight differences cannot explain. Canagliflozin extended male mouse lifespan by 14 percent but, in a more recent study, actually reduced female lifespan. The researchers attributed this to sex-specific metabolic differences, but the mechanism remains unclear. Yesilyurt-Dirican and colleagues flagged the predominant focus on male mice in SGLT2 inhibitor aging research as a significant limitation that raises questions about generalizability to human studies.
| Risk | SGLT2 Inhibitors | Rapamycin | Acarbose |
|---|---|---|---|
| Immunosuppression | None | Primary concern | None |
| Genital yeast infections | 3.3x increased risk | Not applicable | Not applicable |
| DKA (diabetics) | ~3x increased risk | Not applicable | Not applicable |
| DKA (non-diabetics) | Extremely rare (1 case/30,000 pt-years) | Not applicable | Not applicable |
| GI side effects | Minimal | Stomatitis common | Flatulence, bloating (common) |
| Female-specific lifespan concern | Reduced female lifespan in one mouse study | Benefits both sexes | Minimal female benefit |
| Human RCT safety data | Extensive (70,000+ patients) | Limited in healthy adults | Extensive for diabetes |
No one has run a randomized controlled trial testing whether SGLT2 inhibitors or acarbose slow aging in healthy humans. The human data that exists comes from cardiovascular and renal trials in sick populations. Extrapolating from diabetics with heart failure to healthy 50-year-olds seeking to slow aging requires assumptions that have not been tested. The preclinical evidence is strong, the mechanistic rationale is coherent, and the safety profile in non-diabetics looks favorable — but until someone runs the trial, the clinical case remains built on inference rather than direct proof.
Frequently Asked Questions
Are SGLT2 inhibitors approved for anti-aging use?
No. SGLT2 inhibitors are FDA-approved for type 2 diabetes, heart failure, and chronic kidney disease. Any use for longevity or anti-aging purposes is off-label. Physicians can legally prescribe them off-label, but patients should understand that no clinical trial has tested these drugs specifically for slowing aging in healthy people. The safety profile in non-diabetic populations appears favorable based on subgroup analyses from existing trials, but long-term data in healthy individuals is lacking.
Why does acarbose extend lifespan more in male mice than in females?
Researchers do not have a clear answer. The sex difference cannot be explained by weight loss, since acarbose actually reduces body weight and fat more in females. Blood glucose responses to refeeding were significantly reduced only in males, suggesting the drug's metabolic effects are processed differently by sex. The ITP researchers have noted this pattern across multiple studies, and it remains one of the open questions in the field. Similar sex-specific patterns appear with SGLT2 inhibitors, where canagliflozin extended male mouse lifespan but reduced female lifespan in one study.
Can you take acarbose and an SGLT2 inhibitor together?
The NIA's Interventions Testing Program has tested rapamycin plus acarbose as a combination and validated it as one of only two drug combinations that extend lifespan. No ITP study has yet tested acarbose combined with an SGLT2 inhibitor. Some longevity physicians do prescribe both, but there is no published clinical data on this specific combination for aging endpoints. Both drugs affect glucose metabolism, so combining them requires medical oversight to monitor for hypoglycemia and other metabolic effects.
What is the difference between canagliflozin and empagliflozin for longevity?
Canagliflozin is the only SGLT2 inhibitor tested and validated in the ITP for lifespan extension. It inhibits both SGLT2 and SGLT1 (an intestinal glucose transporter), which may contribute to its broader metabolic effects. Empagliflozin is highly selective for SGLT2 with minimal SGLT1 activity, and it has the strongest cardiovascular outcome data from the EMPA-REG trial. Neither has been tested head-to-head for longevity endpoints in humans.
Is there a human clinical trial for acarbose and aging?
Not specifically for aging as a primary endpoint. Acarbose has been used clinically for type 2 diabetes since the 1990s and has extensive human safety data. The Targeting Aging with Metformin (TAME) trial is the closest model for what an aging-focused trial might look like, but TAME tests metformin, not acarbose. Given acarbose's stronger ITP results compared to metformin, some researchers have argued it deserves its own aging-focused human trial, but none has been announced.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.