A Diabetes Drug That Made Mice Live 14% Longer
In 2020, researchers at the National Institute on Aging's Interventions Testing Program gave canagliflozin — a drug originally designed to lower blood sugar in type 2 diabetes patients — to genetically diverse mice starting at seven months of age. The male mice lived 14% longer than untreated controls, with a 9% increase in 90th percentile survival. That finding would be notable for any compound. For a drug already taken daily by millions of people, it set off a very different kind of conversation.
SGLT2 inhibitors — sold under brand names like Jardiance (empagliflozin), Farxiga (dapagliflozin), and Invokana (canagliflozin) — belong to a class of medications that work by blocking a protein in the kidneys responsible for reabsorbing 80-90% of filtered glucose back into the bloodstream. Block that protein, and glucose exits through urine instead. The effect is straightforward: lower blood sugar without requiring more insulin.
What caught longevity researchers' attention wasn't the glucose lowering. It was everything else these drugs seemed to do. A 2023 review by Dr. James O'Keefe and colleagues at Saint Luke's Mid America Heart Institute catalogued an unusually wide range of benefits: reduced risks for heart failure, chronic kidney disease, atrial fibrillation, cancer, gout, dementia, fatty liver disease, and infections. When a single drug appears to protect against that many age-related conditions, the question shifts from "what disease does it treat?" to "is it affecting something more fundamental?"
The answer researchers have converged on: SGLT2 inhibitors may be mimicking caloric restriction — the only intervention consistently shown to extend lifespan across species from yeast to primates. By forcing the body to excrete glucose, these drugs create a mild energy deficit that triggers many of the same cellular defense pathways activated by fasting. Dr. Hoong and Dr. Chua, writing in Endocrinology, went further, calling SGLT2 inhibitors "the most promising" calorie restriction mimetic compared to metformin, rapamycin, resveratrol, and NAD+ precursors — because they regulate multiple longevity pathways simultaneously while already having extensive human safety data.
For anyone tracking the longevity pharmacology space, that combination of preclinical promise and clinical validation makes SGLT2 inhibitors harder to dismiss than most anti-aging candidates.
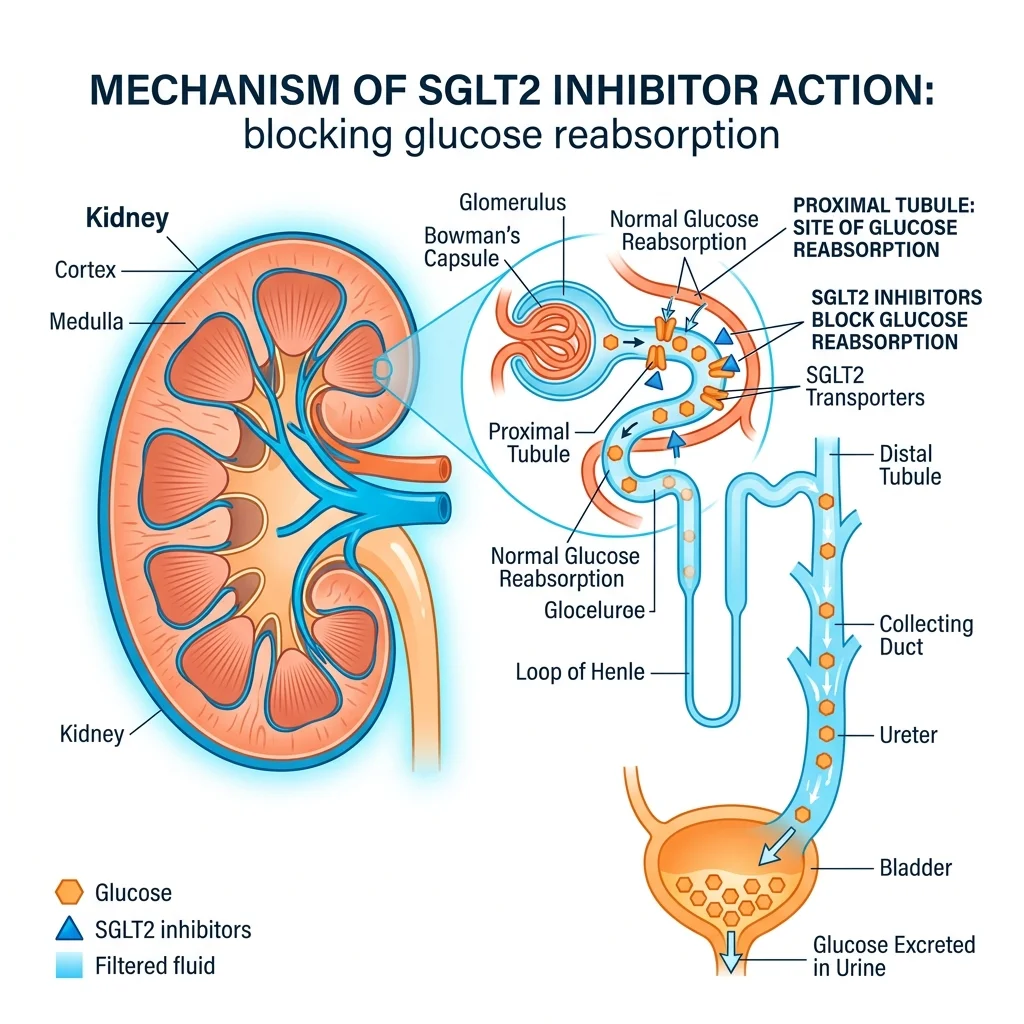
The 300-Calorie Trick Your Kidneys Didn't See Coming
Every day, your kidneys filter roughly 180 grams of glucose from the blood. Under normal circumstances, virtually all of it gets reabsorbed back into circulation through transport proteins in the proximal tubule. SGLT2 handles about 90% of that reabsorption; its cousin SGLT1 picks up the remaining 10%.
When empagliflozin or dapagliflozin blocks SGLT2, the kidneys suddenly let 60 to 80 grams of glucose escape into urine each day — roughly 300 calories that would normally fuel cells. Think of it as a metabolic toll booth that's been shut down. Glucose that used to get waved through now exits the body, and the rest of the metabolic system has to adapt.
That adaptation looks remarkably similar to what happens during fasting. Insulin levels drop. The glucagon-to-insulin ratio shifts. The liver ramps up ketone body production — primarily beta-hydroxybutyrate — as the body pivots from glucose to fat as its preferred fuel source. La Grotta and colleagues documented that this metabolic reshaping occurs even when patients eat normally, because the calorie loss is involuntary. You're eating the same food but absorbing fewer calories. The body responds as if nutrients are scarce.
The downstream effects go well beyond the kidneys. SGLT2 inhibitors have been shown to transform white adipose tissue into metabolically active brown adipose tissue, which burns more calories and generates less inflammation. Blood pressure drops modestly (about 4/2 mmHg on average). Uric acid levels fall by around 13%, reducing gout risk. Weight comes down by roughly 2-3% of body weight.
The kidney and heart benefits of SGLT2 inhibitors are separate from their ability to lower blood sugar. Even people with chronic kidney disease who do not have diabetes may benefit. — National Kidney Foundation
Here's what makes less intuitive sense: the kidney benefits persist even as blood sugar-lowering effectiveness fades in patients with declining kidney function. Current guidelines recommend continuing SGLT2 inhibitors even in advanced chronic kidney disease, because the protective effect on the glomeruli — the tiny filtering units — operates through pressure reduction, not glucose control. The drug relaxes the kidney's filtration system, reducing the overwork that accelerates organ damage. This is why an initial small drop in estimated glomerular filtration rate after starting the drug is actually a reassuring sign, not a dangerous one.
The brain appears to benefit too. A meta-analysis of 10 observational studies involving 819,511 individuals with type 2 diabetes found that SGLT2 inhibitor users had a 38% lower risk of all-cause dementia (relative risk 0.62). Empagliflozin has been shown to increase brain-derived neurotrophic factor (BDNF), a protein that supports neuron growth, survival, and plasticity. Animal studies show reduced amyloid beta levels and senile plaque density — though translating rodent brain findings to humans remains uncertain.
42,000 Patients and a Pattern That Kept Repeating
The cardiovascular data for SGLT2 inhibitors is not speculative. It comes from some of the largest randomized controlled trials in cardiology, and the results have been consistent enough to reshape treatment guidelines worldwide.
The story starts with EMPA-REG OUTCOME in 2015. In a trial of 7,020 patients with type 2 diabetes and existing cardiovascular disease, empagliflozin reduced cardiovascular death by 38% compared to placebo (p<0.001). The magnitude of that reduction, and the speed with which the survival curves separated, surprised the cardiology community. This wasn't the gradual benefit you'd expect from better glucose control over years — it appeared within weeks.
Then came DAPA-HF, which changed the game entirely. Dr. Edgardo Kaplinsky's analysis describes how this trial enrolled 4,744 heart failure patients with reduced ejection fraction, with or without diabetes. Dapagliflozin 10mg daily reduced the primary composite endpoint by 26% (HR 0.74, p<0.001). Worsening heart failure dropped 30%. Cardiovascular mortality fell 18%. The finding that mattered most: the benefits were identical whether patients had diabetes or not. A diabetes drug was protecting hearts regardless of blood sugar status.

The pattern kept repeating. CANVAS showed canagliflozin produced a 14% relative risk reduction in major adverse cardiovascular events. DECLARE-TIMI 58, the largest of the group with over 17,000 patients, confirmed dapagliflozin significantly reduced the composite of cardiovascular death or heart failure hospitalization. CREDENCE demonstrated a 30% reduction in the primary renal composite endpoint with canagliflozin. EMPA-KIDNEY extended empagliflozin's benefits to chronic kidney disease patients regardless of diabetes status.
A meta-analysis pooling 21 randomized trials with 39,593 patients in the SGLT2 inhibitor arm found a 14% relative risk reduction in all-cause mortality (p<0.00001). A separate comprehensive meta-analysis of 17,000 heart failure patients reported reductions across the board: heart failure hospitalization down 29%, adverse renal outcomes down 37%, cardiovascular mortality down 13%, all-cause mortality down 11%.
Real-world data has been even more striking. A propensity-matched observational study of approximately 160,000 patients with newly diagnosed type 2 diabetes found that those prescribed SGLT2 inhibitors had 45% lower all-cause mortality compared to non-users. When a cohort study in JAMA Network Open compared dapagliflozin and empagliflozin head-to-head in 4,930 heart failure patients, the two drugs showed virtually identical outcomes (adjusted hazard ratio 0.99, p=0.95), suggesting this is a class effect rather than a property of any single molecule.
Standard risk factors — blood sugar, blood pressure, weight, cholesterol — explain only a fraction of this benefit. Something else is driving the cardiovascular protection. The longevity research community thinks that "something else" may be the drug's effects on the fundamental biology of aging.
Zombie Cells, Inflammaging, and the Pathways That Drive Aging
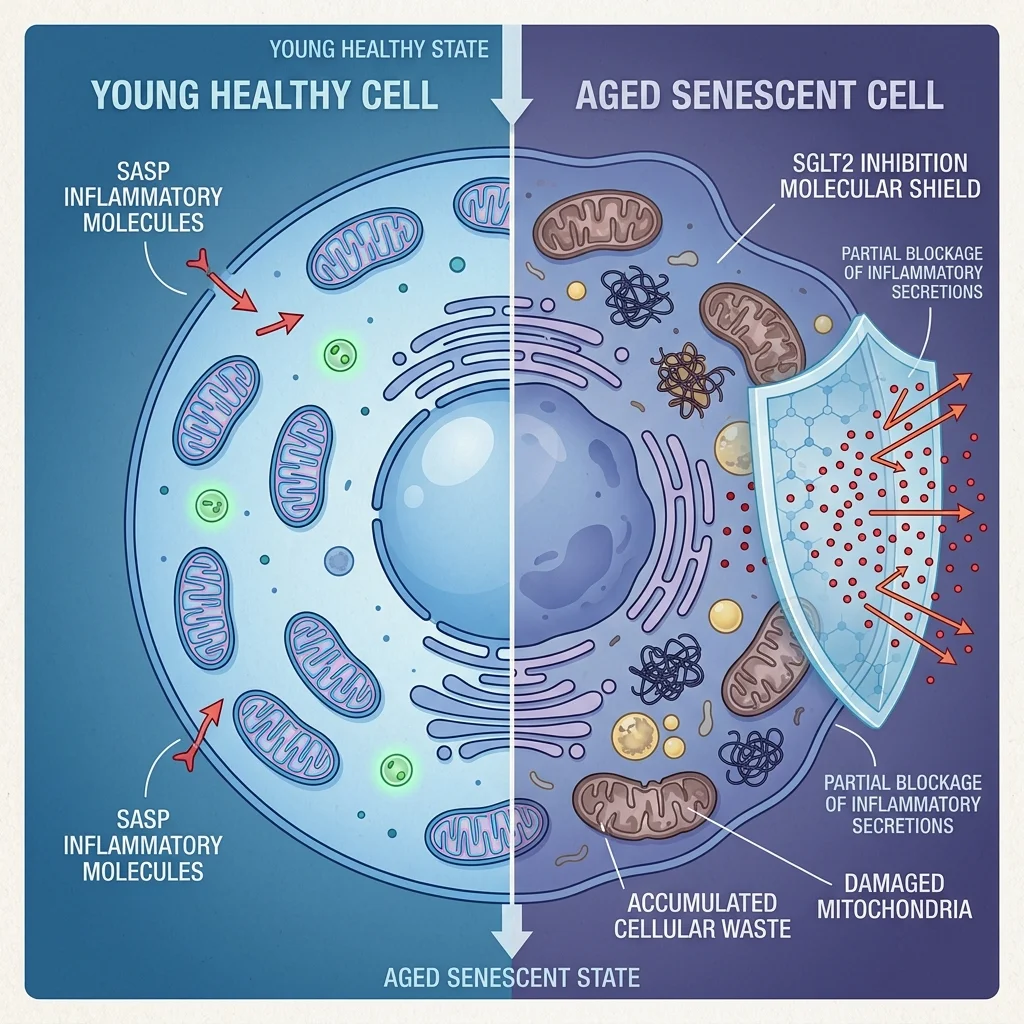
To understand why a diabetes drug might affect aging itself, you need to understand senescent cells — what researchers have taken to calling "zombie cells." These are cells that have permanently stopped dividing but refuse to die. They accumulate in tissues with age, and they're not quiet about it. Each one pumps out a cocktail of inflammatory molecules, growth factors, and tissue-degrading enzymes known as the senescence-associated secretory phenotype, or SASP. Think of them as retired employees who won't leave the office and spend all day sending hostile emails that poison the workplace culture.
This slow, systemic inflammation driven by senescent cell accumulation has a name: inflammaging. It's increasingly recognized as a key driver of virtually all age-related diseases, from cardiovascular disease to neurodegeneration. Among the inflammatory mediators that rise with age, interleukin-6 has been so consistently linked to frailty, diabetes, cardiovascular events, and all-cause mortality that researchers call it "the cytokine for gerontologists."
SGLT2 inhibitors appear to hit this system from multiple angles simultaneously.
First, the senescent cells themselves. Multiple studies in diabetic mouse models show that SGLT2 inhibitors decrease the rate of cellular senescence in kidneys, hearts, and other organs. The mechanism may be direct: senescent cells are metabolically hyperactive and depend heavily on glucose to fuel their inflammatory secretions. Cut their glucose supply via SGLT2 inhibition, and you may starve them into quieter behavior — or make them unable to sustain themselves at all. In cell culture, both dapagliflozin and empagliflozin counteracted pro-senescence effects induced by angiotensin II and by the chemotherapy drug ponatinib in endothelial cells. Researchers now classify SGLT2 inhibitors as "senomorphic" drugs — compounds that don't kill senescent cells outright but dampen their destructive output.
Second, the inflammatory machinery. A pilot trial comparing macrophages from SGLT2 inhibitor-treated patients versus sulphonylurea-treated patients found reduced IL-1beta secretion in the SGLT2 inhibitor group, an effect mediated by elevated beta-hydroxybutyrate levels. The ketone bodies produced by the metabolic shift don't just serve as fuel — they actively suppress the NLRP3 inflammasome, a molecular alarm system that drives much of age-related inflammation. Three separate studies have shown that SGLT2 inhibitors reduce circulating IL-6 levels compared to patients on other diabetes therapies matched for glycemic control, suggesting the anti-inflammatory effect is independent of blood sugar improvement.
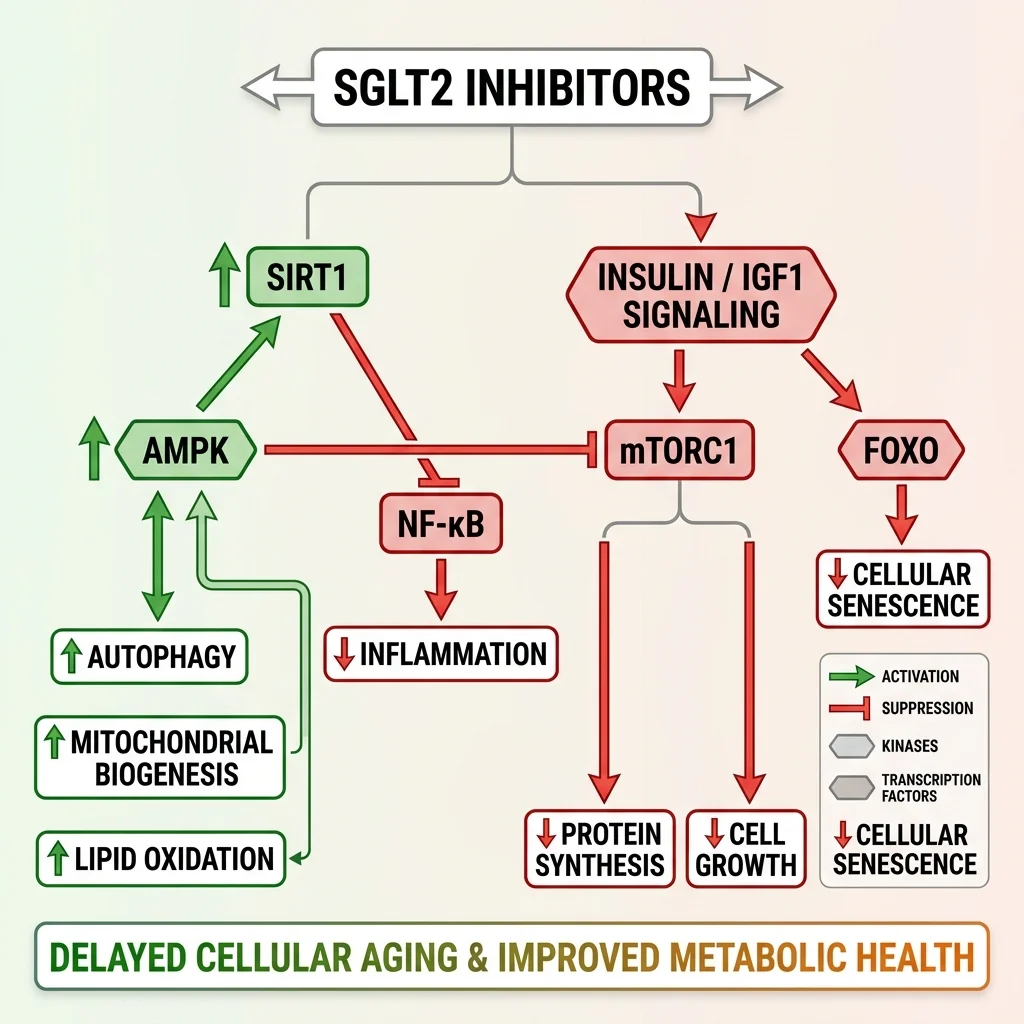
Third, the nutrient-sensing pathways. SGLT2 inhibitors simultaneously upregulate AMPK and SIRT1 (energy deprivation sensors) while suppressing mTOR and insulin/IGF1 signaling (nutrient surplus pathways). This is the same pattern produced by caloric restriction, fasting, and vigorous exercise. AMPK activation restores mitochondrial health and triggers autophagy — the cellular housekeeping process that clears damaged proteins and organelles. SIRT1, activated by rising NAD+ levels, switches on genes involved in stress resistance and mitochondrial biogenesis through transcription factors like PGC-1alpha and NRF-2. Even FOXO3, a transcription factor linked to human longevity in centenarian studies, gets activated through this network.
There's also a structural clue that this isn't just about metabolism. SGLT2 inhibitors bind directly to mTOR in the same structural domain used by rapamycin, the most-studied longevity drug in existence. Cell culture studies show protective effects even in isolated cells without any systemic metabolic changes, which means the drugs may be doing something at the molecular level that has nothing to do with glucose or calories.
From Research Papers to Reddit Threads
The longevity biohacking community has noticed all of this. SGLT2 inhibitors have become a regular topic on Reddit longevity forums, longevity-focused podcasts, and in the prescribing patterns of anti-aging clinicians. The appeal is obvious: unlike rapamycin, which requires careful dosing and has immunosuppressive effects, or metformin, whose anti-aging evidence in non-diabetic humans is weak, SGLT2 inhibitors come with a safety profile validated across tens of thousands of patients in randomized controlled trials.
But there's a gap between what the data shows and what the biohacking community wants it to show. Every cardiovascular and mortality benefit demonstrated in trials has come from patients who already had type 2 diabetes, heart failure, or chronic kidney disease. No randomized trial has tested whether SGLT2 inhibitors slow aging in otherwise healthy people. The mouse lifespan data is promising, but the sex-specific finding — benefits only in males — hasn't been explained, and mouse longevity studies frequently fail to translate to humans.
That gap is about to get its first serious investigation. In February 2026, the Advanced Research Projects Agency for Health (ARPA-H) announced a contract of up to $38 million to the Sam and Ann Barshop Institute at UT Health San Antonio for a trial called VITAL-H (Validation and Intervention Testing for Aging, Longevity and Healthspan). Led by Dr. Elena Volpi, the study will test dapagliflozin alongside rapamycin and semaglutide in generally healthy adults aged 60 to 65 — measuring functional decline rather than disease endpoints.
As Dr. Volpi framed it: "Over the past 50 years, global life expectancy has increased substantially, yet the age of onset of age-related diseases and disabilities has remained largely unchanged." The VITAL-H trial is designed to test whether that trajectory can be bent with drugs that already exist.
Until VITAL-H and similar trials produce results, anyone taking SGLT2 inhibitors off-label for longevity is making a bet — an educated one, backed by strong biological rationale and extensive safety data, but a bet nonetheless. The difference between "reduces cardiovascular death in sick people" and "slows aging in healthy people" is not a small one.
The Side Effects You Need to Weigh
SGLT2 inhibitors have a good safety profile as prescription medications go. In trials, discontinuation rates are similar to placebo. A network meta-analysis of 47,000 patients found no significant increases in risk for hypoglycemia, urinary tract infections, bone fractures, or volume depletion compared to control groups — with one exception.
Genital yeast infections. The same meta-analysis reported a 3.3-fold increased risk of genital fungal infections (6.3% of SGLT2 inhibitor users versus 1.7% of controls). Glucose in the urine creates conditions favorable for fungal growth. For most people this is a manageable nuisance — treatable with antifungals — but for some it's a recurring problem significant enough to stop the medication.
| Risk | Severity | Who's Most Affected |
|---|---|---|
| Genital yeast infections | Common (6.3%) | Women more than men; higher risk with uncontrolled blood sugar |
| Increased urination | Common, mild | Everyone; worse in first weeks |
| Dehydration / low blood pressure | Moderate | Elderly; those on diuretics |
| Euglycemic ketoacidosis | Rare but serious | Diabetic patients during illness, surgery, or fasting |
| Lower limb amputation | Rare, possibly canagliflozin-specific | Patients with peripheral vascular disease |
| Fournier's gangrene | Extremely rare | 12 cases reported over 5 years across all SGLT2i |
The ketoacidosis question deserves particular attention for anyone considering off-label use. In diabetic patients, SGLT2 inhibitors can trigger a dangerous form of ketoacidosis where blood sugar appears normal but ketone levels rise to toxic levels — euglycemic diabetic ketoacidosis. It's most likely during serious illness, surgery, or prolonged fasting. However, in non-diabetic individuals, this risk appears vanishingly small: only a single starvation ketoacidosis event has been reported across approximately 30,000 patient-years of trial follow-up in people without diabetes.
The amputation signal from the CANVAS trial — where canagliflozin doubled amputation incidence (6.3 vs 3.4 per 1,000 patient-years) — initially alarmed clinicians. Subsequent large meta-analyses showed no increased amputation risk with other drugs in the class, suggesting this may have been specific to canagliflozin or the trial population rather than a class-wide effect.
There's also an unresolved biological puzzle. The mouse lifespan extension was observed only in males, and a COVID-19 trial of acute SGLT2 inhibitor administration showed protective effects in men but not women. Whether this sex dimorphism applies to anti-aging effects in humans is unknown. Estrogen's protective effects against senescence may mean women already have partial coverage of the pathways SGLT2 inhibitors activate — or it may mean nothing at all. The VITAL-H trial should help clarify this.
The most honest limitation is the simplest: all the human evidence for cardiovascular and mortality benefits comes from sick populations. Extrapolating "reduces death in heart failure patients" to "slows aging in healthy 45-year-olds" requires a logical leap that biology doesn't always support. The caloric restriction mimicry framework is biologically plausible. The animal data is encouraging. The safety profile is reassuring. But the actual question — does this drug slow human aging? — hasn't been answered yet.
Frequently Asked Questions
Can healthy people without diabetes take SGLT2 inhibitors for longevity?
Technically, a physician can prescribe SGLT2 inhibitors off-label. Some longevity-focused clinicians already do. However, no randomized controlled trial has tested these drugs specifically for anti-aging benefits in healthy people. The ARPA-H-funded VITAL-H trial at UT Health San Antonio will be the first to evaluate dapagliflozin in healthy adults aged 60-65 for age-related functional decline. Until that data exists, off-label use for longevity is based on biological plausibility rather than direct clinical evidence.
What is the difference between empagliflozin and dapagliflozin?
Both are SGLT2 inhibitors with nearly identical cardiovascular and renal benefits. A JAMA Network Open study of 4,930 heart failure patients found no significant difference in outcomes between the two drugs (adjusted hazard ratio 0.99). Empagliflozin (Jardiance) is typically dosed at 10-25mg; dapagliflozin (Farxiga) at 5-10mg. Both are taken once daily. The choice between them usually comes down to insurance coverage and specific FDA-approved indications rather than efficacy differences.
How do SGLT2 inhibitors compare to metformin or rapamycin for anti-aging?
All three drugs are being studied as potential geroprotective agents. Metformin is the subject of the long-running TAME (Targeting Aging with Metformin) trial, while rapamycin has the strongest preclinical longevity data. SGLT2 inhibitors have the advantage of regulating multiple longevity pathways simultaneously — AMPK, SIRT1, mTOR, and autophagy — while having far more clinical safety data than rapamycin. The VITAL-H trial will test dapagliflozin alongside rapamycin and semaglutide head-to-head.
Are SGLT2 inhibitors safe for long-term use?
Clinical trial data extending several years shows a favorable safety profile, with discontinuation rates similar to placebo. The main side effect is genital yeast infections (about 6% of users). Serious adverse events like ketoacidosis are rare and primarily affect diabetic patients during acute illness. However, these drugs have been on the market for only about a decade, which limits long-term data on effects like cancer incidence that require decades of follow-up.
Do SGLT2 inhibitors work differently in men and women?
Possibly. Mouse lifespan extension was observed only in males, and a COVID-19 trial showed protective effects in men but not women. The cardiovascular benefits in clinical trials appear consistent across sexes, but the potential anti-aging effects haven't been specifically studied by sex in humans. Estrogen's own anti-senescence effects may overlap with some of the pathways SGLT2 inhibitors activate, which could explain the sex difference seen in animal models.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.