What the $400 Vial Actually Is
The first thing to know about "NAD+ peptide therapy" is awkward for the marketing: NAD+ is not a peptide. It is nicotinamide adenine dinucleotide, a coenzyme involved in redox reactions and cell signaling, while peptides are short chains of amino acids, as one clinic education page correctly notes while still using the search-friendly phrase "NAD peptide benefits". Think of a peptide as a message passed between cells, and NAD+ as part of the battery-and-wiring system that helps cells move energy around. They can appear in the same wellness menu, but they are not the same category of molecule.
So what does the vial buy? In one public clinic price example, $400 buys a one-month NAD+ supply that includes injection supplies and shipping. The same page describes a subcutaneous protocol of 30 units, or 30 mg, daily, with each vial providing a 33-day supply and a 90-day beyond-use date. That is a useful snapshot, not a recommendation. It tells you what a clinic can sell, not what has been proven to change aging.
WebMD describes NAD therapy as usually delivered by IV infusion, sometimes over several days and several hours at a time, and says plainly that NAD therapy is not proven or approved for any use. It also notes that NAD is considered a dietary supplement rather than a medicine, which is why clinics can offer it outside the normal drug-approval pathway. That regulatory detail matters because the phrase "therapy" makes the vial sound more like a prescription drug than the evidence supports.
The practical translation: a $400 vial may buy NAD+ plus supplies, instructions, and a clinic relationship. It does not buy FDA approval, standardized dosing, guaranteed potency, or proof that your cells will act younger.
| Marketing phrase | What it usually means | What the evidence can support |
|---|---|---|
| NAD+ peptide therapy | NAD+ sold alongside peptide protocols | NAD+ is a coenzyme, not a peptide |
| $400 vial | One clinic example of a one-month home injection supply | A price and dosing example, not proof of benefit |
| Cellular refill | A claim that shots or IVs directly restore intracellular NAD+ | An oversimplified mechanism that needs better human outcome data |
That is the frame for the rest of the article: NAD+ biology is real, but the vial is a commercial delivery format sitting on top of a much more complicated evidence base.
Why NAD+ Levels Became a Longevity Obsession
NAD+ earned attention because it sits near several processes people associate with aging. In their PubMed-indexed review, Nady Braidy and Yue Liu describe NAD+ as present in all living cells and involved in energy production, DNA repair, gene expression, calcium signaling, and immune regulation. Cleveland Clinic's Jacob Hill, ND, gives the simpler version: NAD has a primary role in energy production. If cells were a city, NAD+ would not be the mayor; it would be part of the power grid, repair crew, and traffic-control network at once.
The aging hook comes from a real observation. Braidy and Liu write that NAD+ levels have been reported to decline during aging and age-related diseases. Cleveland Clinic makes the same point for consumers: NAD levels decline with age, and cells may become less efficient at making energy and repairing damage as levels drop. This is why the idea feels intuitive. If the power grid weakens with age, topping it up sounds like common sense.
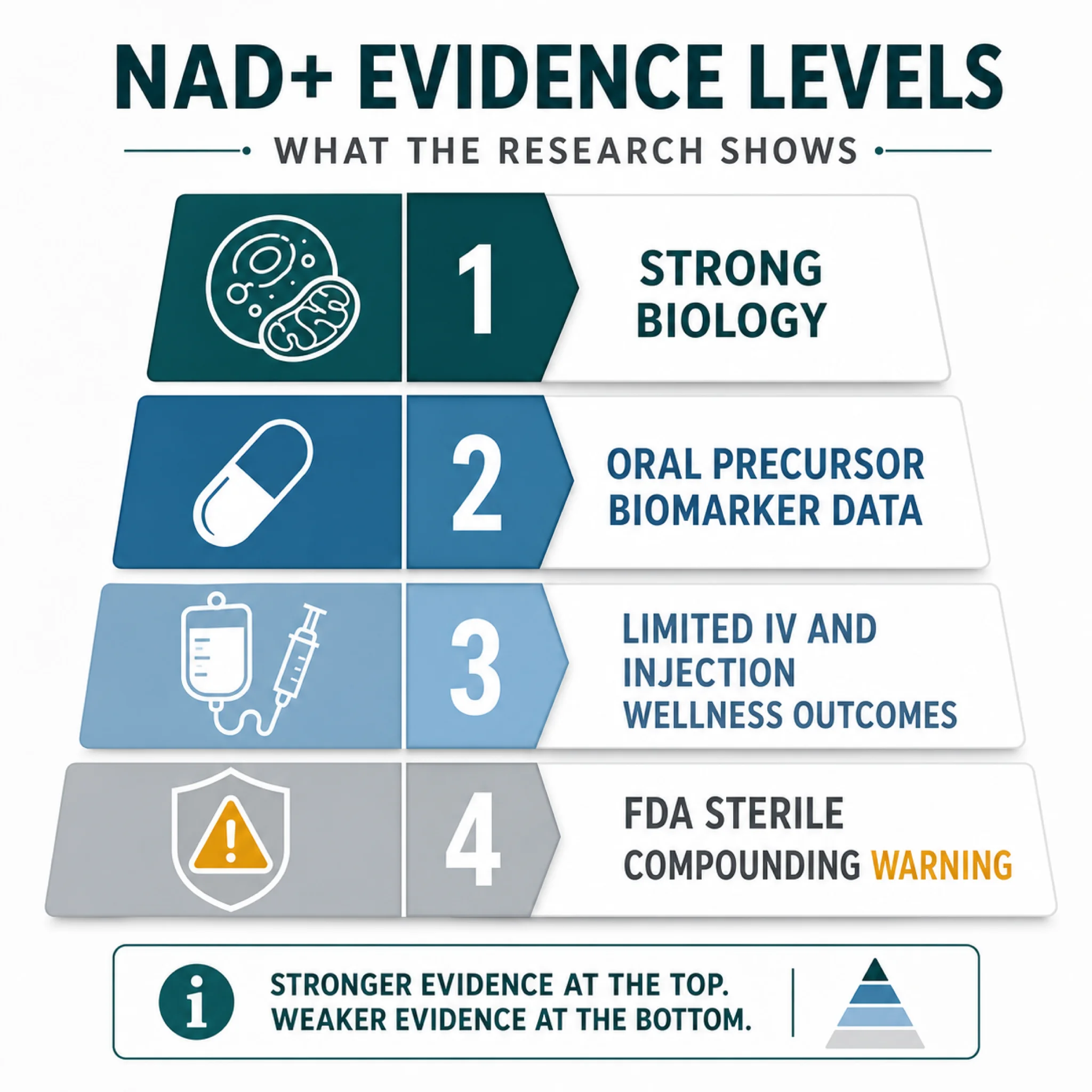
But "common sense" is not a clinical endpoint. Braidy and Liu's review identified 1,545 articles and included 147 studies, of which 113 were preclinical and 34 were clinical. Most studies suggested favorable outcomes for NAD+ precursors in age-related disorders, yet the same abstract says long-term human clinical trials were still nascent. That is the hinge: strong biological plausibility, promising early signals, and not enough durable human outcomes.
Hill's Cleveland Clinic caveat is worth keeping on the table. He says the current interest comes from potential anti-aging and longevity benefits, but long-term benefits and risks are unclear until more research is available. For readers, the takeaway is not that NAD+ is nonsense. It is that the biology is upstream from the marketing. A blood marker can move before your energy, sleep, skin, or lifespan meaningfully changes.
The Evidence Gap Between Molecules and Marketing
The best human evidence in this research package is about oral NAD+ precursors, not $400 injectable vials. A Nature Metabolism randomized, open-label, placebo-controlled study enrolled 65 healthy participants and compared nicotinamide, nicotinamide riboside, and nicotinamide mononucleotide. After 14 days, NR and NMN, but not nicotinamide, comparably increased circulatory NAD+ concentrations in healthy adults.
That is interesting, but it is narrower than the sales pitch. The Nature Metabolism authors say a direct comparison of those prominent NAD+ boosters in humans had been lacking. They also found ex vivo evidence that NR and NMN can give rise to nicotinic acid and enhance microbial growth and metabolism. In plain English, the gut microbiome may be part of the story. The supplement does not simply teleport into a cell and flip a youth switch; it enters a messy chemical supply chain.
The injection claim has an even bigger gap. One clinic evidence review says it found no eligible outcomes trials evaluating IV or IM NAD+ itself for anti-aging or wellness indications. The same review argues that NAD+ is a large, highly polar dinucleotide that is not expected to freely enter cells intact, making the "direct cellular refill" pitch an oversimplification. Use that source cautiously because it is a clinic page, but the caution itself is biologically conservative and consistent with why delivery route matters.
The buyer's practical question is not "Can NAD+ matter?" It is "Does this delivery method, at this price, produce a meaningful outcome for me?" Right now, the strongest answer is: sometimes NAD-related biomarkers move, but the clinical claims around wellness injections run ahead of the evidence.
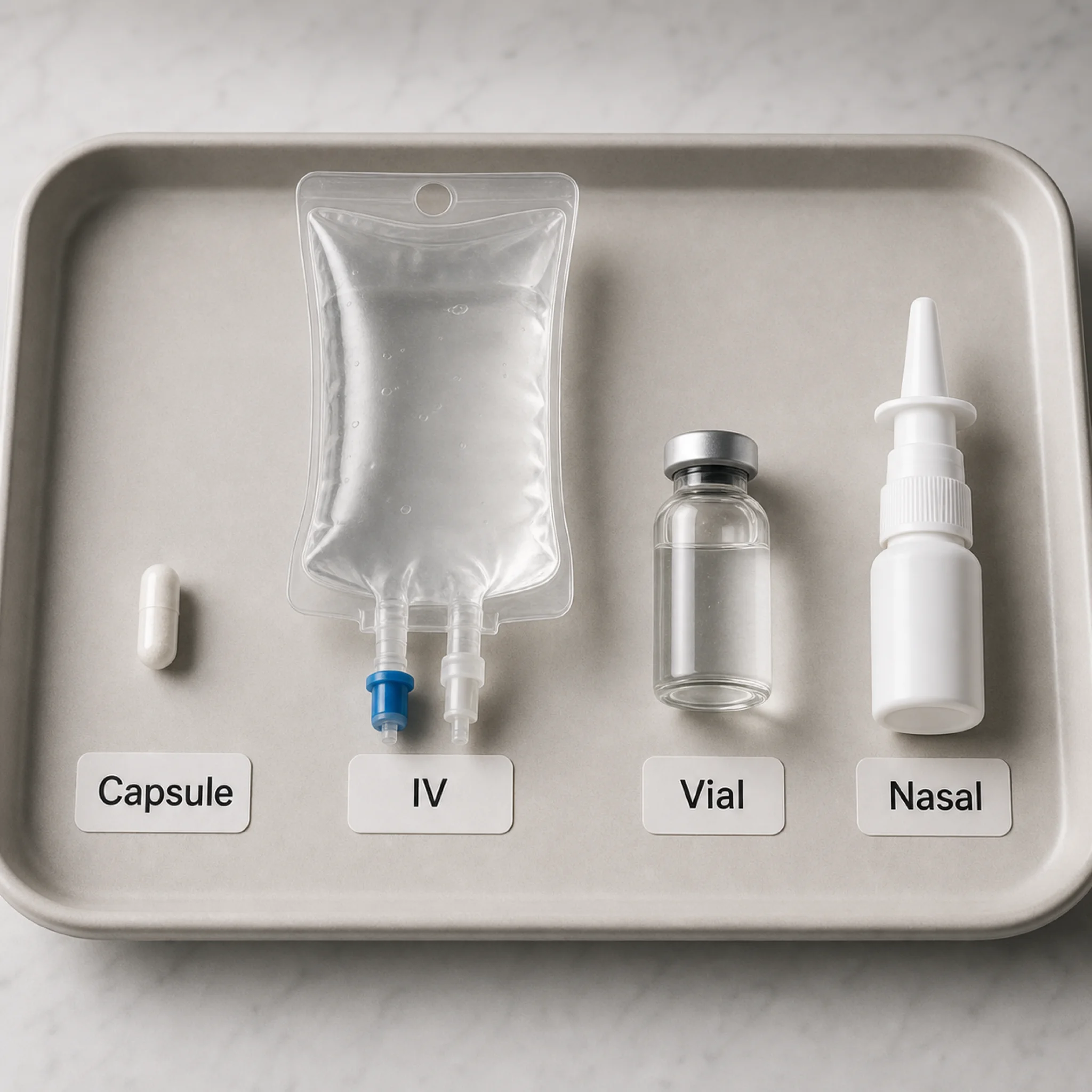
IVs, Injections, and Nasal Sprays Are Not the Same Thing
NAD+ products get grouped together online, but the route changes the question. WebMD says NAD therapy is usually IV, though people may also take pills containing NAD or its precursors. A separate clinic evidence review distinguishes infusions from shots: shots are usually intramuscular or subcutaneous, while infusions go through a vein. Those routes are not interchangeable. A nasal spray would add another absorption problem entirely, and this research package did not produce strong human outcome evidence for nasal NAD+ wellness claims.
IV data exists, but it is thin. WebMD describes one study in which 750 mg of NAD was infused over 6 hours, increasing levels without observed adverse effects, while the researchers still said more needs to be learned about what happens to NAD in the body. WebMD also describes more recent data in which people received 800 to 1,800 mg a day over 3 to 8 hours daily for at least a week, but says NAD therapy still has not been tested in enough people to say whether it works or how well.
Injection clinics use their own protocols. One page markets subcutaneous NAD+ at 50 mg per injection, 5 injections per week. Another sells a home vial at 30 mg daily. Those numbers are not equivalent to the IV regimens above. Comparing them is like comparing a slow-release coffee habit with a hospital-grade caffeine drip; both involve caffeine, but timing, dose, monitoring, and risk are different.
| Route | Typical pitch | Evidence caution |
|---|---|---|
| Oral NR/NMN | Daily precursor support | Human biomarker data exists, but outcomes vary |
| IV NAD+ | Direct high-dose infusion | Time-consuming, costly, not proven for wellness outcomes |
| SubQ or IM NAD+ | At-home or clinic injection convenience | Dosing varies by clinic; not interchangeable with IV data |
| Nasal NAD+ | Fast, needle-free delivery | Not supported here by strong outcome evidence |
That is why route-specific questions beat brand promises. Ask what dose, what sterility controls, what endpoint, what monitoring, and what alternative would be tried first.
Where the Money Goes
The $400 is not just buying a molecule. It may be paying for compounding, sterile handling, supplies, shipping, clinic margin, a prescriber relationship, and the convenience of not sitting through an IV appointment. For IV programs, WebMD says NAD therapy can be costly, with reports suggesting several thousand dollars, and because NAD therapy is not an approved treatment for substance use disorder, insurance generally will not cover it.
Potency is the uncomfortable part of the cost story. A Niagen Bioscience market surveillance analysis tested 27 popular NAD+ products from Amazon. Only 6 of 27 products, or 22%, met or exceeded the label claim. Another 13 of 27 products, or 48%, had less than 1% of the claimed NAD+ or no detectable NAD+. The sponsor has a commercial stake in the NAD precursor market, so treat the report as market surveillance rather than neutral academic research. Still, the numbers are a useful warning: labels are not lab results.
The same report says all 10 softgel products tested had NAD+ below 1% of label claim. That does not automatically indict every vial from every compounding pharmacy. It does show why a buyer should ask for a certificate of analysis, sterility documentation, expiration or beyond-use dating, and exact active ingredient form.
| What you might be paying for | Question to ask |
|---|---|
| Active ingredient | What form is it, and is there a current certificate of analysis? |
| Sterile compounding | Which pharmacy made it, and what sterility testing is done? |
| Clinical supervision | Who reviews contraindications, cancer history, medications, kidney disease, and reactions? |
| Convenience | Is there a cheaper oral precursor or lifestyle step that fits the same goal? |
The most practical way to think about cost is not "Is $400 expensive?" It is "What part of this $400 reduces uncertainty?" If the answer is mostly branding, convenience, and hope, the price is doing more psychological work than clinical work.

Risk, Legality, and Smarter Questions to Ask
The FDA warning is the line that should slow everyone down. The agency says it is aware of compounders using food-grade NAD+ sold by repackagers to make intravenous products. FDA states that food-grade ingredients are not suitable for sterile drug compounding without appropriate processing because of the risk of microbes and endotoxins. It also says adverse event reports after NAD+ injectable drugs included severe chills, shaking, vomiting, and fatigue, reactions consistent with excessive endotoxins.
There is also the deeper biological concern: NAD+ supports cell energy. Cleveland Clinic's Jacob Hill warns that because cancer cells use a lot of energy to grow, trying to increase energy production with NAD supplements could potentially support cancer-cell growth, though this is not conclusive. Braidy and Liu's review similarly lists toxic metabolites, tumorigenesis, and promotion of cellular senescence as potential risks of raising NAD+ in clinical disorders.
None of this means a healthy adult who tries a supervised NAD+ protocol is guaranteed to be harmed. It means the burden of proof belongs on the clinic, not the customer. A glossy promise of "cellular repair" should not outrank sterility, contraindication screening, and honest discussion of uncertainty.
- Ask whether the product is NAD+, NR, NMN, NADH, or another precursor.
- Ask whether the product is FDA-approved for your intended use. For anti-aging and wellness injections, the answer should not be yes.
- Ask which compounding pharmacy made it, what grade of ingredient was used, and whether sterility and endotoxin testing are documented.
- Ask what clinical endpoint will define success before you pay. Better energy is too vague unless you track it.
- Ask whether cancer history, pregnancy, kidney disease, heart disease, medications, and supplement interactions change the risk calculation.
- Ask what cheaper, lower-risk option should be tried first.
Cleveland Clinic's Hill suggests starting with food and healthy lifestyle habits as a safer first step for supporting NAD levels. That advice is less exciting than a vial in a padded mailer, but it is also harder to upsell. For most readers, the smartest move is to separate the molecule from the business model: respect NAD+ biology, question NAD+ therapy claims, and make any paid protocol prove why it is better than the lower-risk alternatives.

Frequently Asked Questions
Is NAD+ actually a peptide?
No. NAD+ is a coenzyme involved in cellular energy and signaling, while peptides are short chains of amino acids. The phrase "NAD+ peptide therapy" usually means NAD+ is being marketed in the same clinic ecosystem as injectable peptides.
Does a $400 NAD+ vial prove the treatment works?
No. A $400 vial can represent a one-month clinic supply, supplies, shipping, and dosing instructions, but price is not proof of clinical benefit. The strongest human evidence in this package is for NAD+ precursors affecting biomarkers, not for injectable NAD+ reversing aging.
Are NAD+ injections FDA-approved for anti-aging?
No. NAD+ infusions and injections are not FDA-approved treatments for anti-aging or wellness outcomes, and WebMD notes that NAD therapy is not proven or approved for any use. Dietary supplements are regulated differently from medicines.
What is the biggest safety concern with injectable NAD+?
Sterility and suitability of ingredients are major concerns. FDA has warned that food-grade NAD+ is not suitable for sterile compounding without appropriate processing and has linked reported chills, shaking, vomiting, and fatigue to possible endotoxin exposure.
What should I ask a clinic before paying?
Ask what exact ingredient is used, who compounded it, whether sterility and endotoxin testing are documented, what dose and route are proposed, what medical conditions would make it risky, and what measurable endpoint would justify continuing.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.