The 700-Species Neighborhood in Your Mouth
Your mouth is the second most microbially diverse environment in your entire body, topped only by your colon. Researchers have cataloged over 700 bacterial species in the human oral cavity, spanning nine major phyla — Firmicutes, Proteobacteria, Bacteroidetes, Actinobacteria, and Fusobacteria among the most abundant. Bacteria are not the only residents. Fungi (dominated by Candida, though they make up less than 0.1% of the total population), archaea, bacteriophages, and even eukaryotic viruses all share the space.
These organisms do not float around randomly. They organize themselves into structured biofilms — layered communities that coat the tongue dorsum, tooth enamel, gingival crevices, and the inner cheeks. The tongue alone hosts distinct microbial neighborhoods at its tip, sides, and back surface. A 2018 analysis in AIMS Microbiology described how the oral cavity is colonized within hours of birth, initially by aerobes like Streptococcus and Neisseria, and later — once teeth erupt and create oxygen-poor pockets — by anaerobes such as Prevotella and Fusobacterium.
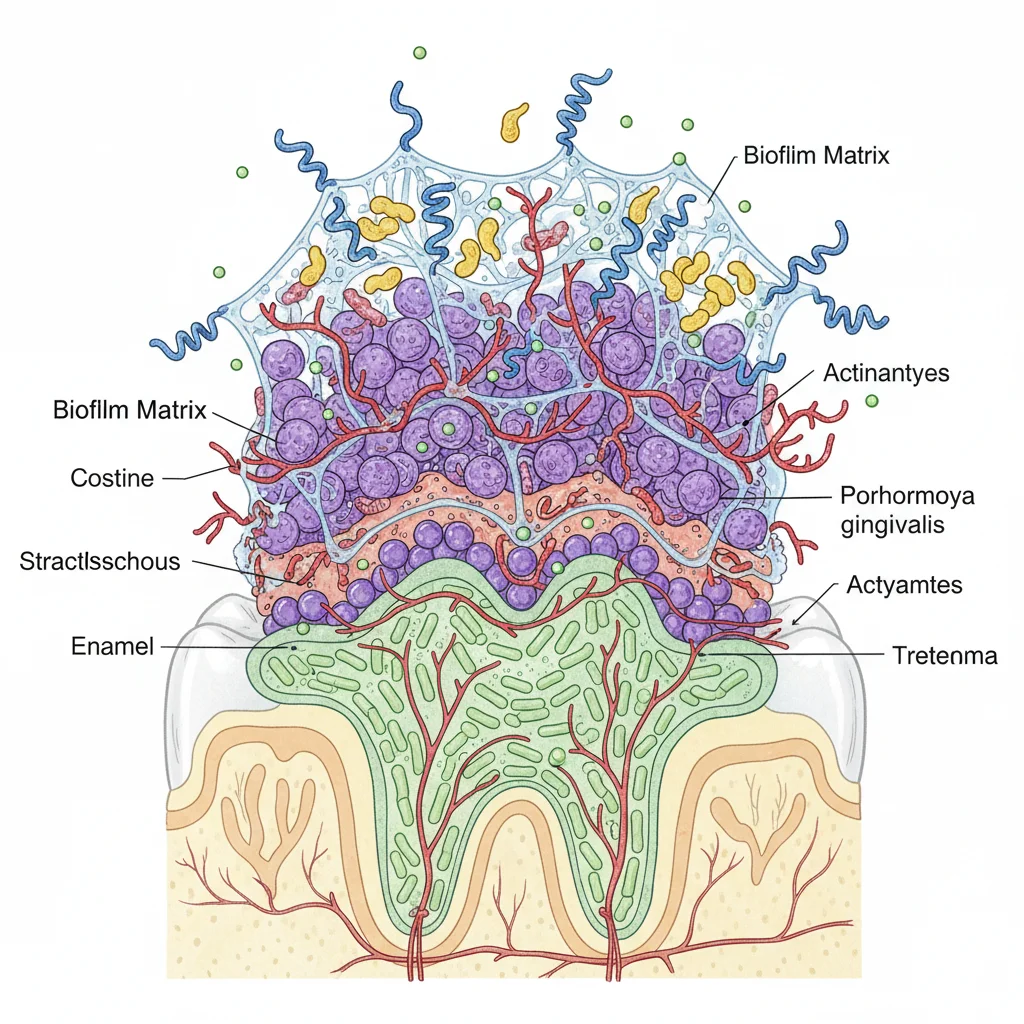
What keeps this ecosystem stable? Saliva is the primary regulator. It bathes every surface with immunoglobulin A (IgA), lysozyme, lactoferrin, and other antimicrobial compounds that selectively limit pathogenic growth while tolerating beneficial commensals. The constant flow physically washes away debris and unattached microbes, sending them to the stomach where acid destroys most of them. Medications that reduce saliva output — antihistamines, antidepressants, certain blood pressure drugs — can shift the balance toward dysbiosis by removing this natural flushing mechanism.
Quick fact: The predominant bacterial genera in a healthy mouth include Streptococcus (~19%), Prevotella (~28%), Veillonella (~10%), Haemophilus (~5%), and Neisseria (~5-13%), based on 16S rRNA sequencing data from clinical studies.
Your Mouth Runs a Chemical Factory for Your Heart
The oral microbiome is not just a passive colony waiting to cause cavities. It actively participates in a physiological process that your cardiovascular system depends on: the production of nitric oxide.
When you eat nitrate-rich vegetables — beets, spinach, arugula, celery — your body absorbs the nitrate through your gut, and your salivary glands concentrate it into your saliva at levels 10 to 20 times higher than blood plasma. That nitrate-loaded saliva then reaches bacteria living on the back of your tongue, primarily species of Haemophilus, Neisseria, and Rothia. These organisms carry nitrate reductase enzymes that convert dietary nitrate into nitrite. The nitrite is then swallowed, absorbed, and converted to nitric oxide — a molecule that relaxes blood vessel walls and lowers blood pressure. This process is called the enterosalivary circulation of nitrate.
Humans lack nitrate reductase. Without oral bacteria performing this conversion step, a significant pathway for nitric oxide production does not function. A 2019 study at the University of Texas tracked 27 healthy adults who used chlorhexidine mouthwash (a potent antibacterial rinse) twice daily for seven days. After one week, their average systolic blood pressure rose from 113 to 115 mmHg — a small but statistically significant increase (p=0.017). When they stopped the mouthwash, nitrate-reducing bacteria repopulated their tongues, and blood pressure dropped back to baseline within three days. For a broader look at dietary strategies that support this pathway, see our guide to nitric oxide-boosting foods.
The same study found that tongue cleaning frequency predicted who was most affected. People who cleaned their tongue twice daily had cultivated a microbiome richer in nitrate-reducing species, so disrupting it with antiseptic rinse caused a larger blood pressure spike (Spearman R=0.45, p=0.03). Routine oral hygiene shapes vascular health, not just fresh breath.
| Oral Health Connection | Evidence Level | Mechanism |
|---|---|---|
| Cardiovascular disease | Multiple systematic reviews | Bacterial translocation, chronic inflammation, nitric oxide disruption |
| Endocarditis | Well-established clinical link | Oral bacteria enter bloodstream, colonize heart valves |
| Preterm birth / low birth weight | Observational studies | Periodontitis-associated systemic inflammation |
| Diabetes (bidirectional) | Strong epidemiological data | Periodontitis impairs glycemic control; diabetes worsens gum disease |
| Pneumonia | Clinical evidence | Aspiration of oral pathogens into lungs |
| Alzheimer's disease | Emerging research | P. gingivalis detected in brain tissue; neuroinflammation |
The Mayo Clinic describes oral health as "a window to your overall health", noting that the mouth serves as an entry point to both the digestive and respiratory tracts. When oral hygiene breaks down, bacteria can reach levels sufficient to trigger systemic infections — particularly in people whose immune function is compromised by conditions like diabetes or HIV.
Tongue Scraping: What the Clinical Trials Found
Tongue scraping is not a wellness trend. It has roots in Ayurvedic medicine going back thousands of years, and the clinical data backing it up is straightforward: mechanical removal of the bacterial coating on the tongue dorsum reduces volatile sulfur compounds (VSCs), the molecules responsible for most cases of bad breath.

A 2006 Cochrane systematic review — the gold standard for evaluating medical interventions — examined randomized controlled trials comparing tongue scrapers to toothbrushes for halitosis reduction. Across 40 participants in two qualifying RCTs, tongue scrapers produced a statistically significant reduction in VSC levels compared to brushing the tongue with a toothbrush. The effect was described as "small but significant."
A larger 2021 RCT with 56 participants published in the International Journal of Environmental Research and Public Health expanded on this. Researchers divided subjects into three groups: toothbrush-only tongue cleaning, tongue scraper-only, and toothbrush plus scraper. All three methods significantly reduced both bad breath measurements and visible tongue coating. The tongue scraper group was the only one to show an immediate significant drop in hydrogen sulfide (H2S) gas — one of the two primary VSC components. However, the overall differences between groups were not statistically significant, leading researchers to conclude that consistent technique matters more than the specific tool.
| Method | VSC Reduction | Tongue Coating Reduction | H2S-Specific Reduction |
|---|---|---|---|
| Toothbrush only | Significant | Significant | Not significant immediately |
| Tongue scraper only | Significant | Significant | Significant immediately |
| Both combined | Significant | Significant | Not significant immediately |
The practical takeaway: a dedicated tongue scraper is slightly more efficient at reducing sulfur gas right after use, but the real variable is whether you clean your tongue at all, and whether you reach the posterior third — the area where anaerobic bacteria concentrate most heavily and produce the bulk of VSCs. The study authors specifically noted that "wiping accurately from the rear of the tongue to the front" mattered more than tool selection.
Beyond fresh breath, there is the cardiovascular dimension. The 2019 University of Texas study found that people who cleaned their tongue twice daily maintained a tongue microbiome enriched in beneficial nitrate-reducing bacteria (Haemophilus parainfluenzae accounted for 22% of tongue species, Neisseria subflava for 12%). Regular cleaning appears to manage the biofilm rather than sterilize it — removing dead cells and debris that feed anaerobic bacteria while preserving the aerobic species involved in nitric oxide generation.
Oil Pulling: Sorting Tradition from Trial Data
Oil pulling — swishing a tablespoon of oil through the teeth for 15 to 20 minutes — is an Ayurvedic practice with a history stretching back 3,000 to 5,000 years. Traditionally performed with sesame oil, it has gained modern popularity with coconut oil substituted as the oil of choice. The claims surrounding it range from plausible (reducing oral bacteria) to unfounded (curing systemic diseases through "toxin pulling"). What does the trial data actually show?
A 2022 meta-analysis published in Healthcare pooled data from nine randomized controlled trials involving 344 total subjects. The analysis found that oil pulling significantly reduced salivary bacterial colony counts compared to controls (mean difference: 17.55, 95% CI 2.56-32.55, p=0.02). Five studies used sesame oil, four used coconut oil, and controls were either chlorhexidine mouthwash or distilled water. However, the same meta-analysis found no significant difference in plaque index (p=0.42) or gingival index scores (p=0.41) between oil pulling and control groups.

A pilot study from Justus-Liebig-University in Germany provided a more granular picture. Researchers used 16S rRNA sequencing to analyze the oral microbiome of three healthy subjects before and after oil pulling with standardized sunflower oil. They found that oil pulling reduced the overall microbial burden by approximately 78-81% in two of the three subjects — a transient reduction, with bacterial counts returning to baseline within hours. Microscopy revealed that epithelial cells became ensheathed in oil droplets during swishing, physically trapping bacteria. The reduction was uniform across bacterial types — oil pulling did not selectively kill particular species but rather reduced the total load through mechanical action. The same study showed an 84% reduction in Candida albicans with oil pulling versus 57% with saline.
Three mechanisms have been proposed to explain how oil pulling works:
| Mechanism | How It Works | Evidence Status |
|---|---|---|
| Saponification | Saliva's bicarbonate ions cause alkali hydrolysis of the oil's fatty acids, creating soap-like compounds that increase the oil's cleansing surface area | Theoretical, consistent with chemistry |
| Viscosity-based trapping | The oil's thick consistency inhibits bacterial adhesion to surfaces and physically traps microbes in the oil/saliva emulsion | Supported by microscopy data |
| Antioxidant action | Compounds in sesame oil (sesamin, sesamolin) and coconut oil (lauric acid → monolaurin) have antimicrobial and antioxidant properties | Demonstrated in vitro; limited in vivo data |
Coconut oil deserves separate mention because its fatty acid profile — 92% saturated fat, roughly half of which is lauric acid — gives it properties the other oils lack. Lauric acid converts to monolaurin in the body, a compound with demonstrated antimicrobial activity against Streptococcus mutans (the primary cavity-causing bacterium) and several Candida species. For more on coconut oil's broader health applications, we have a separate guide.
Bottom line: oil pulling can temporarily reduce bacterial counts in the mouth and may serve as a useful supplement to standard oral hygiene — particularly for people in settings where commercial mouthwashes are unavailable or unaffordable. It is not a replacement for brushing, flossing, or professional dental care. The American Dental Association has not endorsed it, and the meta-analytic data shows no benefit for plaque or gingival inflammation beyond what conventional care provides.
Diet, Habits, and the Balance of Your Oral Ecosystem
The composition of your oral microbiome responds to what you eat, what you drink, and what you expose your mouth to — sometimes within hours.
Fermentable carbohydrates (sugars and refined starches) feed acid-producing bacteria, particularly Streptococcus mutans, driving the acidic pH drops that dissolve tooth enamel and initiate cavities. This relationship is not new — ancient Egyptian mummies show remarkably low rates of dental caries compared to modern populations, primarily because their diet lacked the refined sugars that became widespread in the last few centuries. The modern oral microbiome has shifted toward acid-tolerant species precisely because of dietary changes.
Nitrate-rich vegetables produce the opposite effect. A diet high in leafy greens, beets, and root vegetables promotes the growth of nitrate-reducing bacteria like Rothia and Neisseria, while populations of Prevotella and Veillonella decline. This shift supports the enterosalivary nitric oxide pathway discussed earlier and contributes to better blood pressure regulation.
Smoking creates particularly hostile conditions for oral microbial balance. Cigarette smoke introduces anaerobic conditions, suppresses immune responses, alters salivary pH, and delivers toxic compounds that selectively promote pathogenic biofilm formation. Nicotine itself has been shown to increase the virulence of oral microorganisms by stimulating the expression of virulence-related genes. The connection between smoking and periodontal disease is one of the strongest risk-factor associations in dentistry.
Alcohol is another disruptor. It reduces saliva production (contributing to dry mouth), shifts the pH of the oral environment, and has been associated with changes in microbial diversity that favor pathogenic species. Chronic heavy drinking compounds these effects over time.
Understanding your gut microbiome through testing can provide parallel insights, since the oral and gut microbiomes communicate along the oral-gut axis — swallowed oral bacteria can colonize the gut and influence digestive health.
Practical tips for a balanced oral microbiome: Eat a varied diet rich in fiber and leafy greens. Limit sugary snacks and acidic beverages between meals. Stay hydrated — water supports saliva production. Clean your tongue daily. If you use mouthwash, consider alcohol-free formulas that are less disruptive to beneficial bacteria. Support your body's own defenses with probiotic-rich foods like yogurt and fermented vegetables.
Red Flags: When Your Oral Microbiome Turns Against You
A healthy oral microbiome maintains homeostasis — beneficial and potentially harmful species coexist in a managed balance. Dysbiosis occurs when that balance tips. Here are the warning signs that something has shifted, and what each one means.
Persistent bad breath that brushing does not fix. Chronic halitosis that returns within hours of brushing points to a problem beyond surface debris. It usually indicates an overgrowth of anaerobic bacteria in the gingival pockets or on the posterior tongue — producing hydrogen sulfide and methyl mercaptan at levels that overwhelm normal saliva clearance. This accounts for roughly 85-90% of persistent bad breath cases.
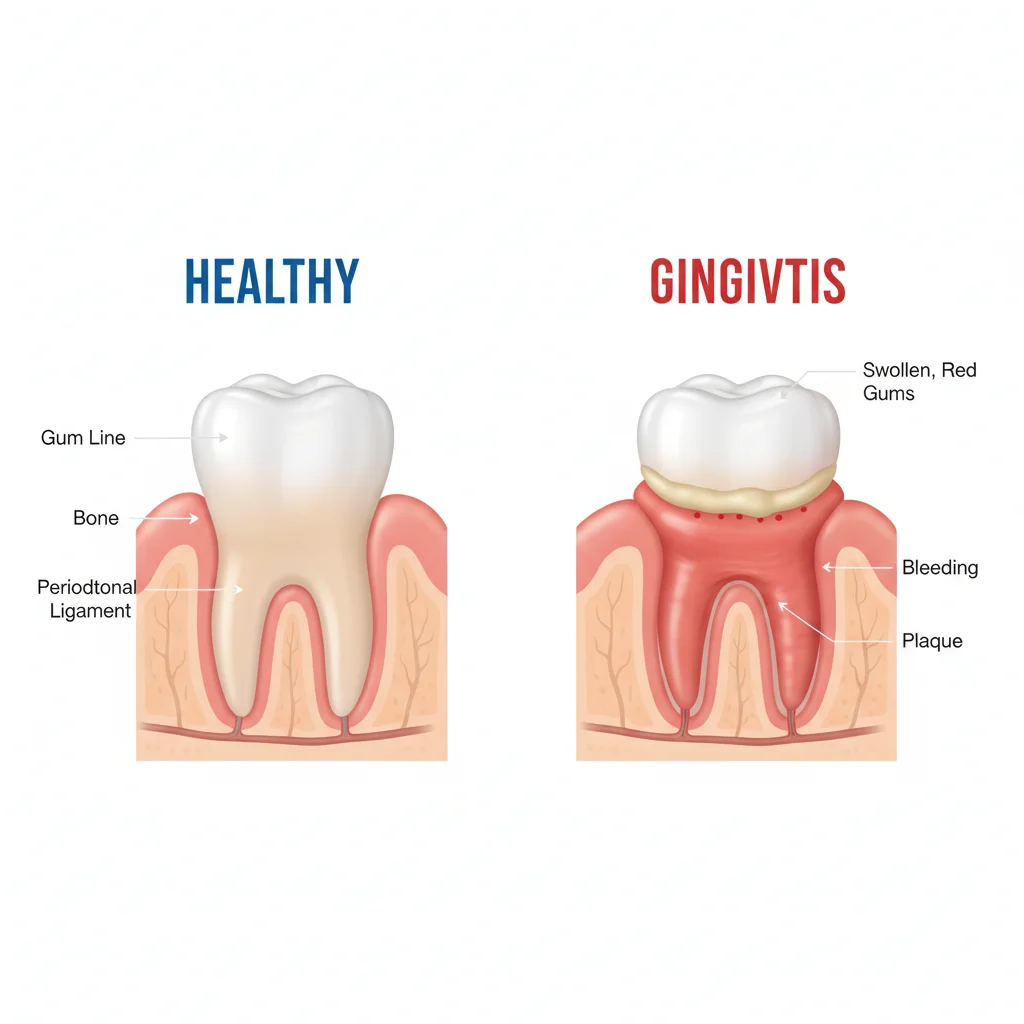
Bleeding gums during brushing or flossing. Occasional minor bleeding from aggressive flossing can be normal. But gums that bleed regularly indicate gingivitis — the earliest stage of periodontal disease — driven by bacterial biofilm irritating the gum tissue. Left unchecked, gingivitis progresses to periodontitis, where the inflammation destroys the bone supporting your teeth.
White patches on the tongue or inner cheeks. A thick white coating on the tongue can signal overgrowth of Candida species (oral thrush), common in people using inhaled corticosteroids, taking antibiotics, or with weakened immune function. The oral cavity normally hosts small amounts of Candida — problems arise when the bacterial balance shifts enough to let it proliferate unchecked.
Dry mouth that persists. Chronic xerostomia (dry mouth) is not just uncomfortable — it removes one of the oral microbiome's primary regulators. Without adequate saliva flow, bacterial populations shift toward acid-producing and pathogenic species. More than 500 medications list dry mouth as a side effect, including common antidepressants, antihistamines, and diuretics.
Recurrent cavities despite good brushing habits. If you are developing new cavities despite consistent brushing and flossing, the microbial composition of your biofilm may have shifted toward highly cariogenic (cavity-causing) species. This can happen after antibiotic use, during periods of high sugar consumption, or as a result of reduced saliva flow.
Any of these signs warrant a dental evaluation. Periodontal disease in particular has documented associations with cardiovascular disease, diabetic complications, adverse pregnancy outcomes, and respiratory infections — making it a systemic health concern, not just a dental one.
Frequently Asked Questions
How often should you scrape your tongue?
Once or twice daily is sufficient for most people. The clinical evidence supports daily tongue cleaning as the minimum frequency for maintaining reduced VSC levels and managing the tongue biofilm. The best time is during your morning oral hygiene routine, before eating, using gentle strokes from back to front. If you notice significant tongue coating, a second cleaning in the evening may help.
Can oil pulling replace brushing and flossing?
No. Oil pulling can temporarily reduce the total bacterial count in your mouth, but meta-analytic data shows it does not significantly improve plaque or gingival index scores compared to standard oral care. It works best as a supplement — not a substitute — for brushing, flossing, and professional cleanings. The American Dental Association has not endorsed oil pulling as a standalone oral hygiene method.
Does mouthwash kill good bacteria along with bad?
Yes, particularly antiseptic mouthwashes containing chlorhexidine or high alcohol concentrations. Research shows that twice-daily chlorhexidine use for just one week significantly reduced nitrate-reducing bacteria on the tongue and raised systolic blood pressure in healthy adults. Alcohol-free mouthwashes and those formulated with cetylpyridinium chloride tend to be less disruptive to the overall microbial community.
What type of oil works best for oil pulling?
Clinical trials have used sesame oil, coconut oil, and sunflower oil. Coconut oil has the added benefit of containing lauric acid, which converts to monolaurin — a compound with demonstrated antimicrobial activity against S. mutans and Candida species. Sesame oil contains antioxidant lignans. Both are reasonable choices. The key variables are duration (15-20 minutes) and consistency rather than oil type.
Is there a connection between oral health and heart disease?
Multiple lines of evidence support this connection. Oral bacteria can enter the bloodstream through inflamed gums, potentially contributing to endocarditis and atherosclerosis. The oral microbiome's role in nitric oxide production directly affects vascular function. And chronic periodontal inflammation triggers systemic inflammatory markers associated with cardiovascular risk. The Mayo Clinic lists cardiovascular disease, endocarditis, and pregnancy complications among the conditions linked to oral health.
Related Articles
- Nitric Oxide Foods: The Complete Guide to Eating for Better Blood Flow and Lower Blood Pressure — Learn which vegetables fuel the nitrate-nitrite-NO pathway that your oral bacteria help drive.
- Bad Breath (Halitosis): Causes, Remedies, and What Actually Works — A deeper look at the volatile sulfur compounds behind chronic halitosis and evidence-based treatments.
- Health Benefits of Coconut Oil — Explore the broader applications of coconut oil, including its lauric acid content and antimicrobial properties.
- Health Benefits of Probiotics: An Evidence-Based Ultimate Guide — How probiotic strains support immune function and microbial balance throughout the body.
- Gut Health Personalization and Microbiome Testing Guide — Understanding the oral-gut axis and how microbiome testing can reveal connections between your mouth and digestive health.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.