40% of the Population Carries This Gene Variant and Most Have No Idea
Somewhere inside your cells, a single gene called MTHFR is quietly doing work that affects everything from how you process B vitamins to how your body repairs DNA. The full name is methylenetetrahydrofolate reductase, and no, you do not need to pronounce it. What it does is actually pretty simple once you strip away the jargon.
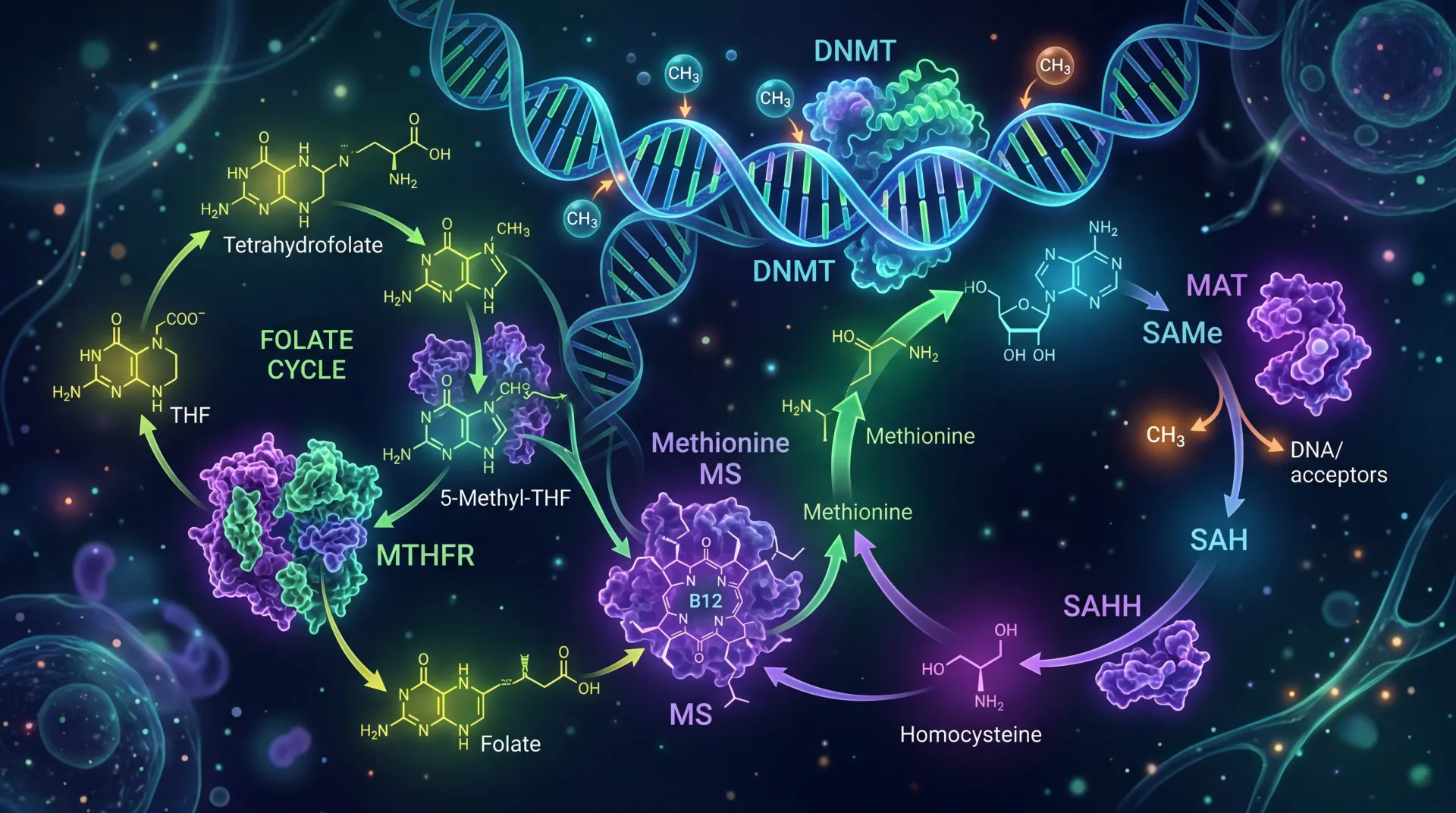
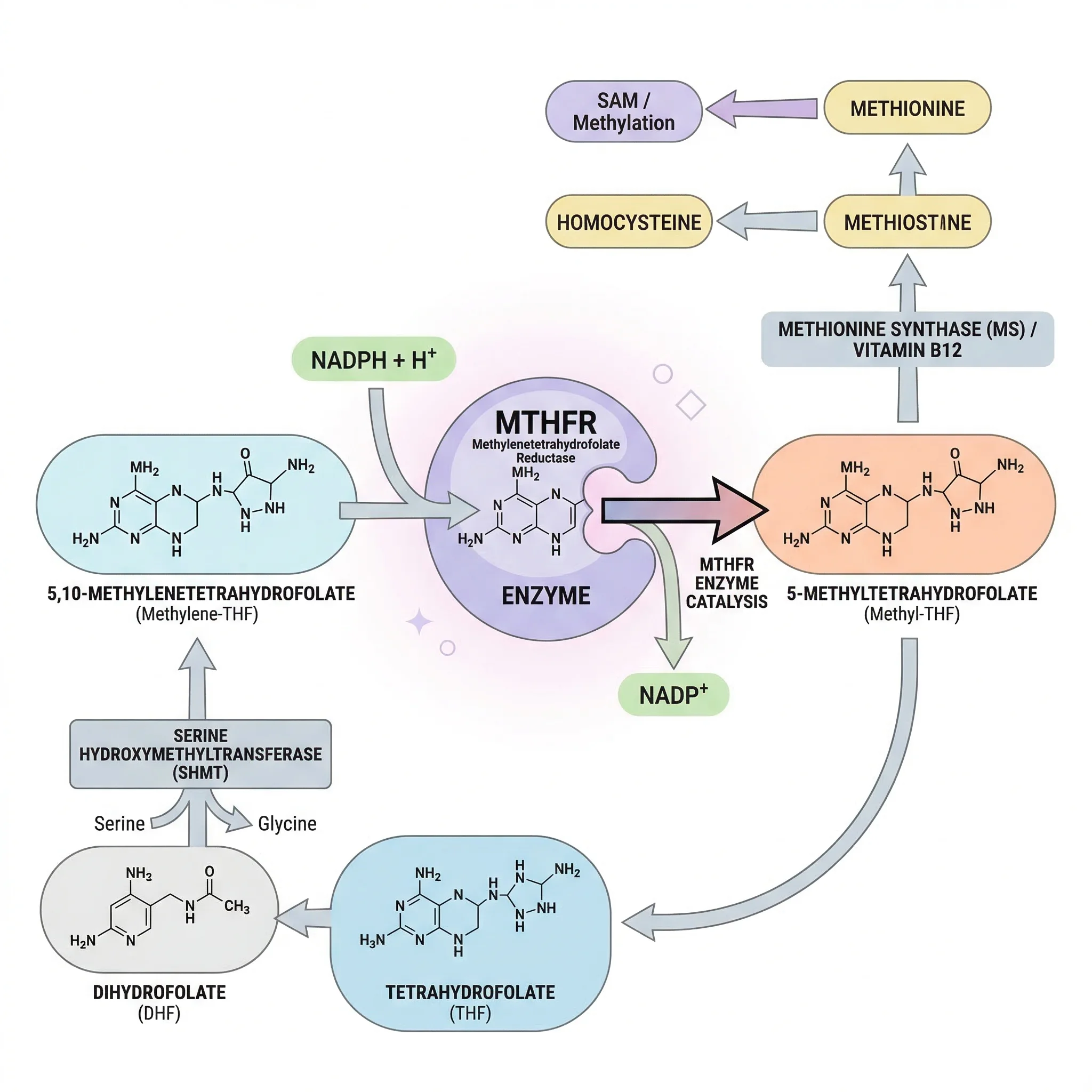
The MTHFR gene sits on chromosome 1 and provides instructions for producing the MTHFR enzyme. This enzyme has one critical job: it catalyzes the conversion of 5,10-methylenetetrahydrofolate into 5-methyltetrahydrofolate, the primary circulating form of folate in your blood. That converted folate then feeds directly into the methylation cycle, where it helps convert the amino acid homocysteine back into methionine. Methionine, in turn, gets transformed into S-adenosylmethionine (SAM), often called the body's universal methyl donor.
When MTHFR works normally, this chain of reactions runs smoothly. Folate circulates, homocysteine stays within healthy ranges, methionine gets produced, and SAM hands off methyl groups to DNA, proteins, and neurotransmitters throughout the body. When the gene carries certain variants, the enzyme's efficiency drops, and problems start showing up in places you might not expect.
Key fact: According to the CDC, more people in the United States carry one or two copies of the most common MTHFR variant (C677T) than don't carry it. This is not a rare condition. It is a widespread genetic variation whose health impact depends largely on diet and folate intake.
The reason MTHFR has generated so much attention in recent years is this intersection between genetics and nutrition. Unlike many genetic variants that you can do nothing about, MTHFR variants respond to relatively simple dietary and supplemental interventions. The enzyme may work at reduced capacity, but the degree to which that affects your health is heavily influenced by factors within your control.
The Biochemical Engine That Keeps Every Cell Running
Methylation is a biochemical process that happens billions of times per second in your body. It involves transferring a methyl group -- one carbon atom bonded to three hydrogen atoms -- from one molecule to another. That simple transfer switches genes on and off, builds neurotransmitters like serotonin and dopamine, processes hormones, repairs DNA, and detoxifies chemicals in the liver. It is, in a sense, the body's way of labeling molecules with sticky notes that say "active" or "inactive."
The methylation cycle depends on several B vitamins working together. Folate (vitamin B9) enters the cycle as 5-methyltetrahydrofolate, the form produced by the MTHFR enzyme. This folate derivative donates its methyl group to homocysteine through a reaction that also requires vitamin B12 as a cofactor. The result is methionine, which the body then converts to SAM.
SAM is the molecule that actually performs methylation throughout your body. It donates methyl groups to over 100 different substrates, including DNA, RNA, proteins, and lipids. After SAM gives up its methyl group, it becomes S-adenosylhomocysteine (SAH), which gets converted back to homocysteine, restarting the cycle.
When MTHFR enzyme activity is reduced, less 5-methyltetrahydrofolate gets produced. This creates a bottleneck. Homocysteine does not get converted back to methionine as efficiently, so it accumulates in the blood. Methionine production drops, which means SAM production drops, and methylation reactions throughout the body slow down. Research from the NCBI Madame Curie Bioscience Database identifies three distinct pathways through which reduced MTHFR activity can influence disease:
- Elevated homocysteine: Homocysteine or its metabolites may have direct toxic effects on blood vessels and on embryo development during pregnancy.
- Disrupted SAM synthesis: Reduced methionine means less SAM, which means less methylation of DNA and other molecules. People with the TT genotype have demonstrated decreased methylation in lymphocytes, and since altered DNA methylation changes gene expression, this could influence developmental and even cancer-related processes.
- Redistributed folate metabolites: With less MTHFR activity, methyltetrahydrofolate decreases while other folate forms (like methylenetetrahydrofolate) increase. This redistribution can affect thymidine and purine synthesis, potentially impacting DNA synthesis and repair.
So methylation problems are not one thing breaking in one place. When the MTHFR enzyme slows down, the effects spread across cardiovascular health, neurological function, reproductive health, and basic cellular maintenance. But here is the part that matters most: the slowdown responds to folate. The more folate available to the enzyme, the better it compensates for reduced efficiency.
Two Variants, Very Different Consequences
Researchers have identified multiple variants in the MTHFR gene, but two matter far more than the rest: C677T and A1298C. These are not diseases. They are polymorphisms, meaning they are common genetic variations found throughout the general population. The difference between them lies in how much they reduce enzyme function and what that means for homocysteine levels.
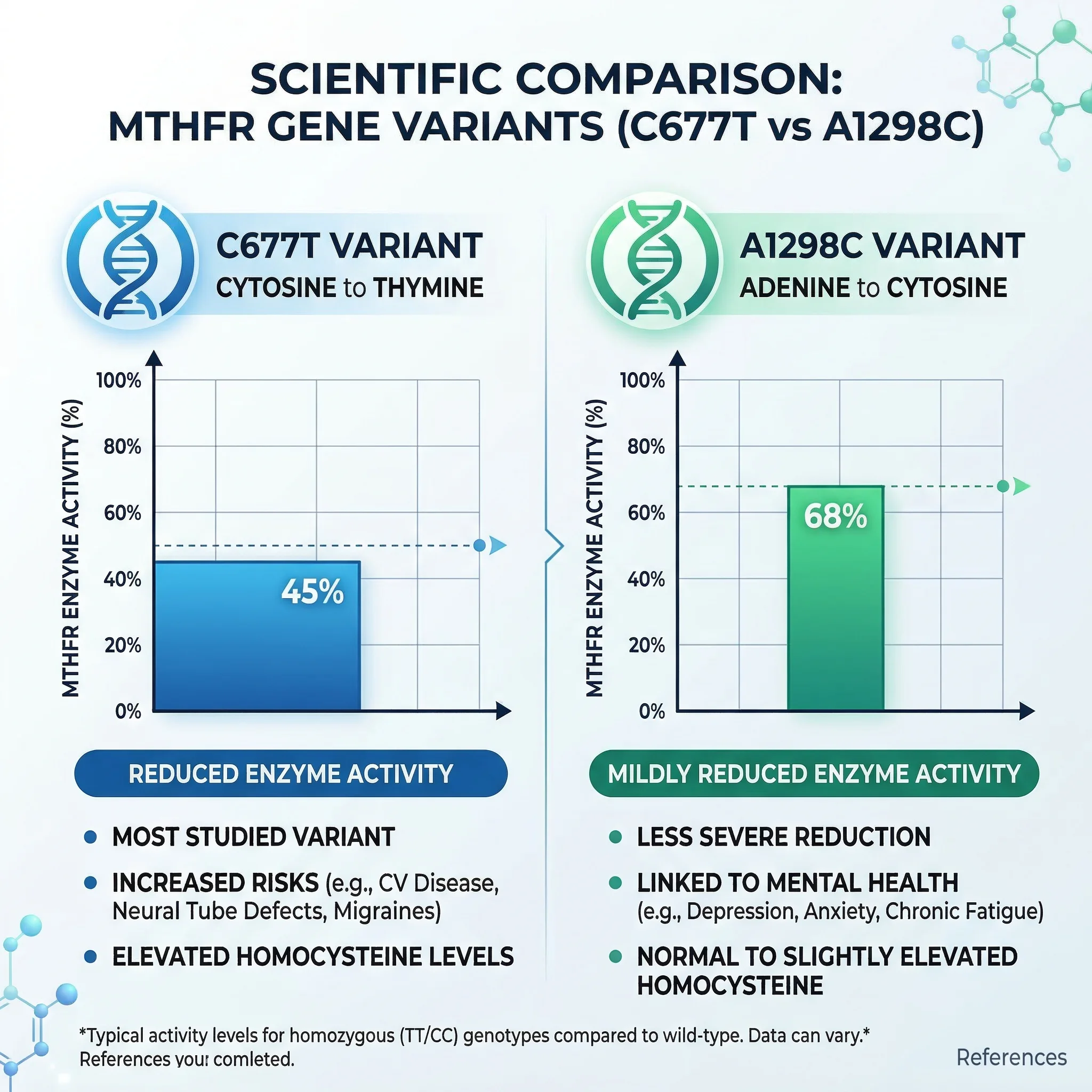
The C677T Variant
The C677T variant involves a single nucleotide change at position 677 of the MTHFR gene, where cytosine (C) is replaced by thymine (T). This substitution changes the amino acid at position 222 from alanine to valine, producing an enzyme that is thermolabile (less stable at body temperature) and functions at roughly 45% of normal activity when both copies are affected (homozygous TT). Heterozygous carriers (CT) have intermediate enzyme activity.
This variant is remarkably common. Population studies cited in the NCBI GeneReviews show homozygosity (TT) rates of 6 to 14 percent in White European populations, less than 2 percent in individuals of African descent, and as high as 21 percent in some Hispanic populations in California. The 677T allele is thought to have arisen from a single founder event, and its persistence at such high frequencies across diverse populations suggests it may have once conferred a selective advantage.
| Population | TT Homozygosity Rate | Notable Finding |
|---|---|---|
| White European | 6-14% | Moderate prevalence across populations |
| Hispanic (California) | ~21% | Highest documented rates |
| Hispanic (Colombia) | ~18% | Consistent with Mediterranean ancestry |
| African (continental) | <2% | Lowest documented rates |
| African American | <2% | Similar to continental African populations |
| Southern Mediterranean | High | Likely ancestral population for Hispanic prevalence |
The A1298C Variant
The A1298C variant changes adenine to cytosine at position 1298, converting glutamate to alanine at amino acid position 429. This variant sits in the regulatory domain of the enzyme rather than the catalytic domain. The result is a milder reduction in enzyme activity, down to about 68% of normal activity in homozygotes. The A1298C enzyme is not thermolabile, meaning it does not lose additional function at body temperature the way the C677T variant does.
A1298C homozygotes (CC) make up approximately 8 percent of tested European populations and, on their own, do not appear to have significantly higher homocysteine levels than people with the normal genotype. The CDC notes there is not enough evidence that the A1298C variant alone significantly affects how the body processes folate.
Compound Heterozygotes: When Both Variants Collide
The combination that does warrant attention is compound heterozygosity, where someone carries one copy of C677T and one copy of A1298C. Research published in Atherosclerosis found that compound heterozygotes tend to have a biochemical profile closer to that of C677T homozygotes (TT), with increased serum homocysteine levels. The two variants almost always occur on separate chromosomes (in trans), since they arose independently, and recombination between the two positions has been too infrequent to place them on the same chromosome.
| Genotype | Enzyme Activity | Homocysteine Impact | Clinical Significance |
|---|---|---|---|
| 677 CC / 1298 AA (normal) | 100% | Baseline | No concerns |
| 677 CT (heterozygous) | ~65-70% | Minimal increase | Generally not clinically significant |
| 677 TT (homozygous) | ~45% | Moderate increase (folate-dependent) | Clinically relevant if folate is low |
| 1298 CC (homozygous) | ~68% | No significant increase | Minimal clinical significance alone |
| 677 CT + 1298 AC (compound) | ~50-55% | Similar to 677 TT | Clinically relevant if folate is low |
When a Gene Variant Becomes a Health Problem
Carrying an MTHFR variant is not the same as having a disease. Most carriers live their entire lives without symptoms. The variant becomes a health concern only when reduced enzyme activity combines with poor folate intake, certain medications, or other genetic and environmental factors that push homocysteine levels high enough to cause problems.
The internet has turned MTHFR into a catch-all explanation for everything from chronic fatigue and depression to autoimmune disorders and cancer. The actual evidence is narrower than that. Here is what research has consistently linked to MTHFR variants, particularly the C677T homozygous (TT) genotype:
Elevated homocysteine (hyperhomocysteinemia): This is the most well-established consequence. The 677TT genotype was identified as the most common genetic cause of hyperhomocysteinemia by Frosst et al. in their landmark 1995 study in Nature Genetics. High homocysteine is recognized as an independent risk factor for cardiovascular disease, though the magnitude of that risk remains debated.
Neural tube defects: The MTHFR 677C→T variant was reported as the first genetic risk factor for neural tube defects shortly after its identification. Families with neural tube defects had elevated plasma homocysteine levels. This finding reinforced the importance of adequate folate intake before and during pregnancy, regardless of MTHFR status.
Cardiovascular disease: The connection between homocysteine and vascular damage was first proposed in the 1960s, and the MTHFR variant became a candidate for cardiovascular risk modification. Early studies supported the connection, but subsequent large-scale trials have produced mixed results. The current scientific consensus is that the C677T variant is a modest, not dramatic, cardiovascular risk factor, and that its impact is heavily modulated by folate status.
Pregnancy complications: Beyond neural tube defects, some studies have associated the TT genotype with recurrent pregnancy loss and other obstetric complications, though the strength of these associations varies across populations.
Severe MTHFR deficiency, caused by rare deleterious mutations (not the common C677T or A1298C polymorphisms), produces a different clinical picture entirely. About 50 patients worldwide have been diagnosed with severe deficiency. Their symptoms include developmental delay, seizures, thromboses, and vascular lesions. This condition is the most common inborn error of folate metabolism, yet it remains extremely rare compared to the common polymorphisms that most discussions of MTHFR focus on.
A Test That Tells You Less Than You Think
MTHFR genetic testing is widely available through direct-to-consumer companies and medical laboratories. A simple blood draw or saliva sample can identify whether you carry the C677T variant, the A1298C variant, or both. The test itself is accurate and inexpensive. The real question is whether knowing your result changes anything you would actually do.
Multiple medical organizations, including the American College of Medical Genetics and Genomics (ACMG), have recommended against routine MTHFR testing for most clinical scenarios. Their reasoning: knowing your MTHFR status rarely changes the recommended course of action. If your homocysteine is elevated, you should optimize folate intake whether or not you carry an MTHFR variant. If your homocysteine is normal, an MTHFR variant is clinically irrelevant.
The more useful test, in most cases, is a serum homocysteine level. If it is elevated (generally above 15 micromoles per liter), the treatment is the same regardless of the underlying cause: ensure adequate intake of folate, vitamin B12, and vitamin B6. If homocysteine is normal, an MTHFR variant is unlikely to be causing health problems.
That said, MTHFR testing can be informative in specific situations:
- Evaluating unexplained hyperhomocysteinemia that does not respond to standard B vitamin supplementation
- Investigating recurrent pregnancy loss in conjunction with other thrombophilia testing
- Providing context for family planning when there is a history of neural tube defects
- Understanding personal pharmacogenomics (certain drugs like methotrexate interact with folate metabolism)
| Test | What It Measures | When It Is Useful |
|---|---|---|
| MTHFR genotyping | Specific gene variants (C677T, A1298C) | Family planning, unexplained hyperhomocysteinemia, pharmacogenomics |
| Serum homocysteine | Total homocysteine level in blood | Most clinically actionable test for assessing methylation status |
| Serum folate | Circulating folate level | Evaluating nutritional status, especially if homocysteine is elevated |
| Red blood cell folate | Long-term folate stores | More accurate than serum folate for assessing true folate status |
| Vitamin B12 | B12 level (cofactor in homocysteine metabolism) | Should always be checked alongside homocysteine |
What Actually Helps: Food, Folate, and the Folic Acid Debate
If you take one thing from this article, make it this: eat enough folate. That is the single biggest lever you have. Research from the NHLBI Family Heart Study, involving 365 individuals, demonstrated that the 677TT genotype was associated with higher homocysteine only in individuals whose plasma folate was below the median. When folate levels were above the median, the genotype had no measurable effect on homocysteine. Folate effectively neutralized the genetic disadvantage.

This brings us to one of the most contentious debates in the MTHFR conversation: folic acid versus methylfolate (5-MTHF). Supplement companies and social media influencers widely promote the idea that people with MTHFR variants cannot process folic acid and must take methylfolate instead. The CDC directly contradicts this claim. Their official position, updated in 2025, states unequivocally: "People with an MTHFR gene variant can process all types of folate, including folic acid."
The data behind this position shows that even people with the most affected genotype (677 TT) have blood folate levels only about 16 percent lower than those with the normal genotype when consuming the same amount of folic acid. Folic acid remains the only form of folate clinically proven to prevent neural tube defects, and the CDC recommends 400 micrograms daily for anyone who could become pregnant, regardless of MTHFR status.
That said, some clinicians and researchers offer a more nuanced view. While folic acid clearly works, methylfolate bypasses the MTHFR enzyme entirely, entering the methylation cycle as the already-active form. For individuals with confirmed hyperhomocysteinemia who have not responded adequately to folic acid alone, methylfolate supplementation may be worth discussing with a healthcare provider.
Beyond folate, several other nutrients play supporting roles in methylation:
- Riboflavin (vitamin B2): This is the precursor of FAD, the cofactor that stabilizes the MTHFR enzyme. Research on the MTHFR enzyme showed that FAD could protect the mutant enzyme from destabilization, suggesting riboflavin supplementation may help maintain enzyme function in carriers of the 677T variant. Riboflavin gets almost no attention in the MTHFR conversation, and it probably should.
- Vitamin B12: Essential as a cofactor for the methionine synthase reaction that converts homocysteine to methionine. Without adequate B12, folate supplementation alone will not normalize homocysteine. For more on this nutrient's broader role, see our guide to iron and nutrient deficiency testing.
- Vitamin B6: Supports the transsulfuration pathway, an alternate route for homocysteine disposal that converts it to cysteine instead of recycling it back to methionine.
- Betaine (trimethylglycine): Acts as an alternative methyl donor, converting homocysteine to methionine through a pathway that does not depend on MTHFR or folate at all. Animal studies in MTHFR-deficient mice showed that betaine supplementation reduced plasma homocysteine across all genotypes and prevented liver damage in the most severely affected animals.
Food sources of natural folate include dark leafy greens (spinach, kale, collards), legumes (lentils, chickpeas, black beans), asparagus, broccoli, and citrus fruits. The body absorbs about 50 percent of naturally occurring food folate versus about 85 percent of folic acid from fortified foods, so people relying solely on whole foods may need to eat more folate-rich meals to maintain adequate levels. You can find dietary strategies that complement methylation support in our electrolytes and mineral nutrition guide.
Separating MTHFR Facts from Internet Myths
| Myth | Fact |
|---|---|
| MTHFR mutations are rare genetic diseases | The common C677T and A1298C variants are polymorphisms found in a large portion of the population. The CDC states that more Americans carry C677T than don't. Rare, severe MTHFR deficiency (about 50 cases worldwide) is a different condition entirely. |
| People with MTHFR variants cannot process folic acid | The CDC explicitly states this is false. Even 677TT homozygotes process folic acid, achieving blood folate levels only about 16% lower than people with the normal genotype. |
| You need methylfolate supplements if you have MTHFR | Folic acid is the only folate form proven to prevent neural tube defects. Methylfolate is an option, not a requirement. Many people with MTHFR variants do fine on standard folic acid at recommended doses. |
| MTHFR causes anxiety, depression, chronic fatigue, and autoimmune disease | While methylation influences neurotransmitter production, direct causal links between common MTHFR variants and psychiatric or autoimmune conditions have not been established in controlled studies. Correlation in small studies does not equal causation. |
| Everyone should get MTHFR testing | Major medical organizations recommend against routine MTHFR testing. A homocysteine level test is more clinically useful and actionable. |
| The MTHFR gene mutation means you are a poor detoxifier | MTHFR affects one specific enzyme in the folate-homocysteine cycle. It does not broadly impair the body's detoxification capacity, which involves hundreds of different enzymes across multiple organ systems. |
Frequently Asked Questions
Is MTHFR a serious health condition?
The common MTHFR variants (C677T and A1298C) are not diseases. They are genetic polymorphisms carried by a large portion of the population. For most people, they cause no symptoms and require no treatment. The variants become clinically relevant primarily when folate intake is inadequate and homocysteine levels rise. Severe MTHFR deficiency, caused by rare mutations, is a distinct and serious metabolic disorder, but it affects fewer than 50 known patients worldwide.
Should I take methylfolate instead of folic acid if I have an MTHFR variant?
Not necessarily. The CDC states that people with MTHFR gene variants can process all types of folate, including folic acid. Folic acid is the only form proven to prevent neural tube defects and is recommended at 400 mcg daily regardless of MTHFR status. Methylfolate (5-MTHF) is an alternative that bypasses the MTHFR enzyme, and some clinicians recommend it for individuals with confirmed hyperhomocysteinemia who do not respond to folic acid. Discuss options with a healthcare provider rather than self-prescribing based on genetic test results alone.
Can diet alone manage MTHFR-related homocysteine elevation?
For many people, yes. Adequate dietary folate from dark leafy greens, legumes, and fortified foods, combined with sufficient B12 and B6, can maintain normal homocysteine levels even with reduced MTHFR enzyme activity. The NHLBI Family Heart Study showed that 677TT individuals with folate levels above the population median had no elevation in homocysteine. However, people with significantly elevated homocysteine may benefit from targeted supplementation with folate, B12, B6, riboflavin, and possibly betaine.
Does having an MTHFR variant affect pregnancy?
The C677T variant was identified as the first genetic risk factor for neural tube defects, which reinforces the importance of folic acid supplementation before and during pregnancy. The CDC recommends 400 mcg of folic acid daily for anyone who could become pregnant, with or without an MTHFR variant. Some practitioners recommend higher doses or methylfolate for women with the TT genotype, though standard folic acid remains the evidence-based recommendation. Discuss your specific situation with a prenatal care provider.
Why does riboflavin matter for MTHFR?
Riboflavin (vitamin B2) is the precursor of FAD, the cofactor that the MTHFR enzyme needs to function. Research has shown that FAD can protect the C677T mutant enzyme from losing its structural stability. Ensuring adequate riboflavin intake may help maintain higher levels of MTHFR enzyme activity in carriers of the variant. Good dietary sources include dairy products, eggs, lean meats, and fortified cereals. Most people do not think about riboflavin when they think about metabolic health, but for MTHFR carriers it may be one of the most practical interventions available.
Related Articles
- Gut-Brain Axis: How Your Microbiome Affects Mood and Cognition -- Explore how gut health influences the same neurotransmitter pathways affected by methylation.
- Thyroid Health and Hashimoto's: Symptoms, Diet, and Natural Support -- Thyroid function and MTHFR share overlapping nutritional cofactors including B vitamins and selenium.
- Iron Deficiency in Women: Symptoms, Testing, and Recovery -- Nutrient testing strategies that apply equally to evaluating folate and B12 status alongside iron.
- Spinach Health Benefits and Nutritional Facts -- One of the richest natural sources of dietary folate, the nutrient most critical for MTHFR variant carriers.
- Electrolytes Explained: Sodium, Potassium, Magnesium for Hydration -- Mineral balance supports the same metabolic pathways that methylation depends on.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.