The only drug proven to extend lifespan in mice — so why are people stopping it?
In 2009, a team led by David Harrison at the Jackson Laboratory made a finding that sent shockwaves through aging research: rapamycin, a drug originally approved to prevent organ transplant rejection, became the first compound unequivocally shown to extend maximum lifespan in genetically normal mice. Male mice lived about 9% longer. Females, 14%. The drug worked even when started in middle age — the equivalent of giving it to a 60-year-old human.
The mechanism centers on a protein complex called mTORC1, which functions like a cellular growth switch. When nutrients are plentiful, mTORC1 drives cells to grow, divide, and build new proteins. Think of it as the accelerator pedal on your metabolism. As we age, this pedal gets stuck — mTORC1 activity ratchets up in many tissues, pushing cells toward growth when they should be repairing and recycling damaged components instead.
Rapamycin pulls back on that accelerator. By inhibiting mTORC1, it triggers autophagy — a cellular housekeeping process where cells break down and recycle their own damaged parts. It mimics some of the same signaling changes that happen during caloric restriction, the oldest known lifespan-extending intervention, without requiring anyone to eat 30% fewer calories for decades.
That combination — robust animal data, a clear mechanism, and FDA approval since the 1990s — sparked a wave of off-label use among longevity enthusiasts. Physicians and biohackers began prescribing rapamycin for anti-aging despite it not being recognized as a treatment by the broader clinical community. But the excitement has collided with a stubborn reality: the side effect profile of this drug is more complicated than many early adopters anticipated, and the human evidence remains far thinner than the mouse data suggests.
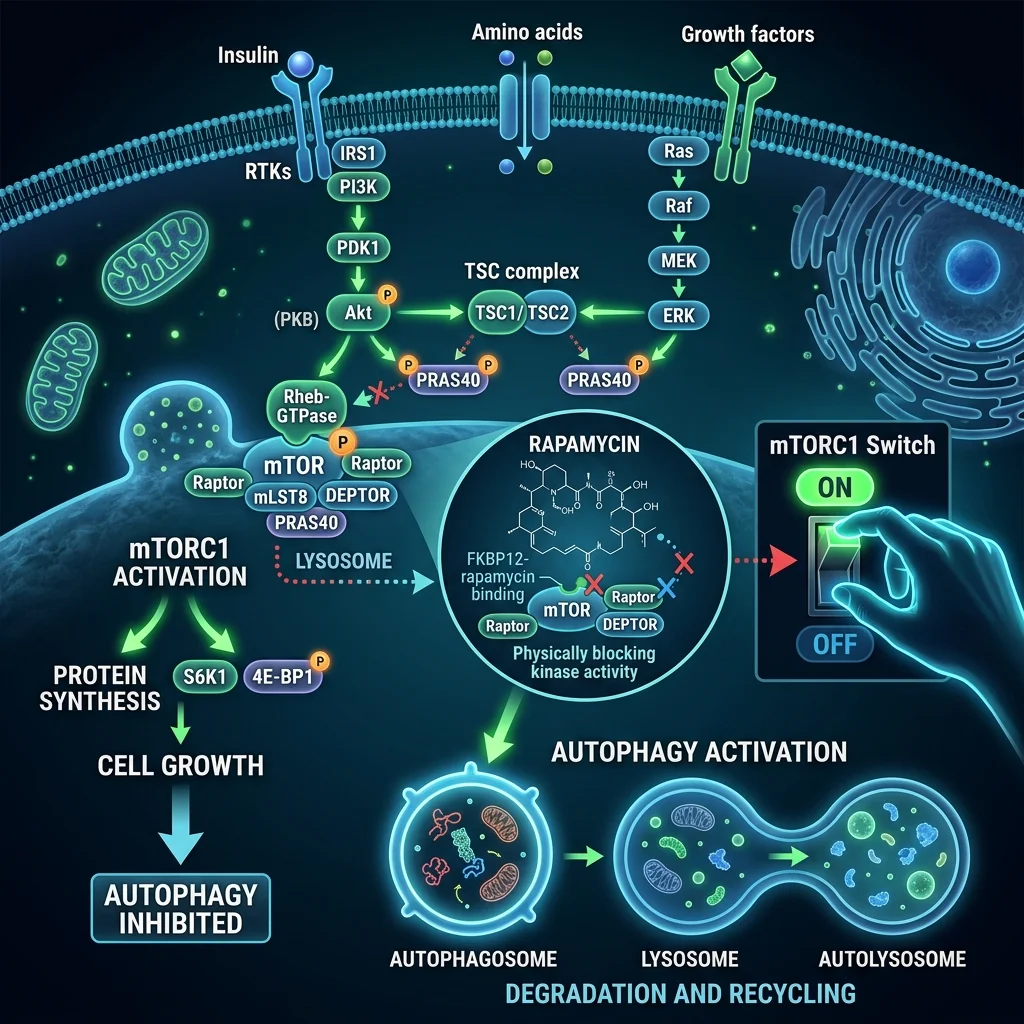
What happens to your blood sugar on rapamycin
The metabolic story of rapamycin contains a genuine paradox. Give a single dose to cells in a lab dish, and insulin sensitivity improves within the first hour — rapamycin disrupts a feedback loop where overactive mTORC1 signaling jams insulin's ability to do its job. But keep those cells bathing in rapamycin for 24 to 48 hours, and the picture flips completely. Researchers at the University of Wisconsin found almost complete insulin resistance setting in by the second day of continuous exposure.
The culprit is a second protein complex called mTORC2. While mTORC1 (rapamycin's intended target) governs cell growth and autophagy, mTORC2 plays a different role — it sits in the insulin signaling chain and helps cells respond to glucose. A quick hit of rapamycin leaves mTORC2 alone. But with chronic exposure, rapamycin starts disrupting mTORC2 as well, and that is where the metabolic trouble begins.
In a 2010 study published in Diabetes, researchers gave rats rapamycin daily for 15 days and documented severe glucose intolerance and hyperlipidemia driven by increased hepatic gluconeogenesis — the liver cranking out more glucose than the body needed. The mechanism they uncovered was striking: the glucose dysregulation happened despite normal insulin signaling through the Akt pathway. The liver was producing excess glucose through a separate set of transcriptional regulators that rapamycin had activated.
Human data tells a more nuanced version of this story. In a trial by Kraig and colleagues, 25 healthy older adults (ages 70 to 95) took 1 mg of sirolimus daily for eight weeks. Their triglycerides went up, their HbA1c crept higher, and plasma albumin dropped — but fasting glucose and insulin sensitivity measured by standard tests did not change significantly. The researchers concluded that short-term rapamycin did not produce a frank diabetic picture, though the metabolic shifts were concerning.
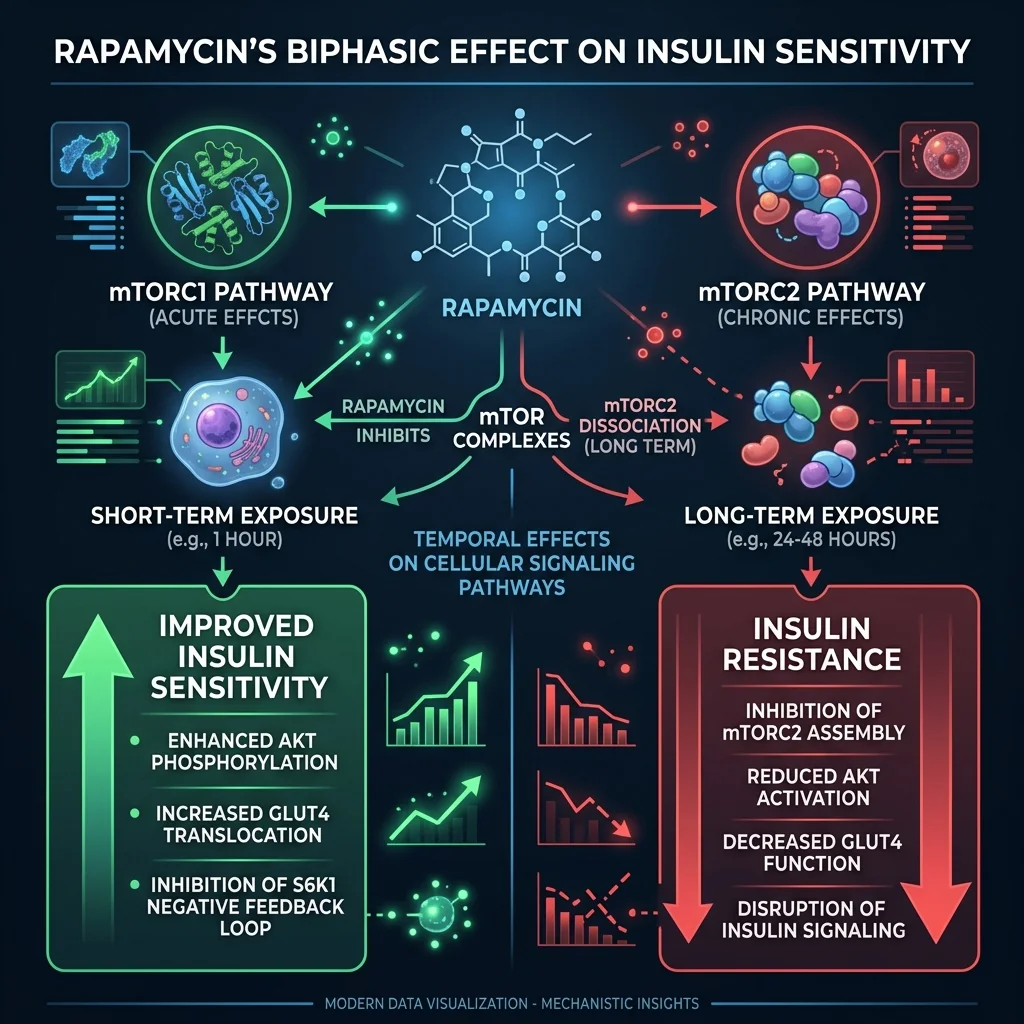
Some researchers frame these metabolic changes differently. Dr. Mikhail Blagosklonny, a vocal rapamycin advocate, has argued that what looks like diabetes is actually "starvation pseudo-diabetes" — a condition first described 170 years ago in fasted animals. During prolonged fasting, the body deliberately suppresses glucose uptake by non-brain tissues to preserve fuel for the brain. Rapamycin, as a partial starvation mimetic, may trigger this same metabolic shift. The distinction matters because starvation pseudo-diabetes is reversible and does not produce the organ damage that actual type 2 diabetes causes over time.
Where does that leave someone considering rapamycin? The metabolic effects are real and measurable. They appear dose-dependent and duration-dependent — the longer and higher the exposure, the worse the glucose picture gets. For someone already prediabetic or taking metformin, adding rapamycin could push metabolic markers in the wrong direction. The saving grace, based on current evidence, is that these changes seem to reverse when the drug is stopped.
Does rapamycin weaken or strengthen your immune system?
This is the question that generates the most heated disagreement among rapamycin researchers, and the honest answer is: it depends entirely on the dose.
The landmark study that shifted the conversation came from Dr. Joan Mannick and colleagues in 2014. They gave low-dose everolimus (a rapamycin analog) to 218 healthy older adults, then measured their immune response to a flu vaccine. The low-dose groups showed a 20% increase in antibody titers, and their levels of PD-1-positive T cells (a marker of immune exhaustion) dropped — suggesting their immune systems were actually getting younger, not weaker.
That finding upended the assumption that rapamycin was purely immunosuppressive. Blagosklonny has gone further, arguing that at anti-aging doses, rapamycin "eliminates hyperimmunity rather than suppresses immunity" — essentially dialing down an overactive aging immune system rather than shutting it off. He points to data showing rapamycin reduces CMV infection risk in transplant patients and protects aged mice against pneumonia.
But this picture has limits. When a follow-up study tested whether mTOR inhibition could reduce actual respiratory tract infections (not just vaccine responses), the results were mixed. A phase 2b trial showed promise, but the larger phase 3 trial failed to replicate those results — though the endpoint was changed from lab-confirmed infections to patient-reported infections, muddying the comparison.
The dose-dependent threshold appears to be real. At immunosuppressive doses used in transplant medicine, rapamycin clearly dampens immune function — that is its approved purpose. At the lower doses longevity practitioners use (typically 3-10 mg weekly rather than daily), the immune effects may reverse direction. But "may" is doing heavy lifting in that sentence. The largest trials have not definitively proven that low-dose rapamycin protects against real-world infections in healthy people.
Bryan Johnson's experience sits uncomfortably in this gap. He reported intermittent skin and soft tissue infections during his rapamycin use. His doses ranged from 5 mg to 13 mg weekly — within the range that some studies suggest should enhance rather than suppress immunity. Whether his infections were caused by rapamycin, by his complex cocktail of 50+ other supplements and medications, or by something unrelated remains genuinely unclear. Rapamycin's effect on natural killer cells is one documented concern that could theoretically impair cancer surveillance and pathogen defense even at moderate doses.
Mouth sores, slow healing, and the side effects nobody warned about
If you spend time in rapamycin user communities, mouth sores come up constantly. There is good reason for that — they are among the most common and most disruptive side effects of mTOR inhibition, and they have a formal medical name: mTOR inhibitor-associated stomatitis, or mIAS.
A comprehensive review by Dr. Douglas Peterson and colleagues analyzed data from 2,822 patients on mTOR inhibitors and found an all-grade stomatitis incidence of 52.9%. That is more than half of all users developing mouth lesions. The incidence range across different trials was 2% to 78%, with grade 3 or 4 severity (meaning painful enough to affect eating or requiring medical intervention) occurring in up to 9% of patients.
What makes mIAS distinctive is the mismatch between size and pain. The lesions are typically small — less than half a centimeter in diameter — and appear on the tongue, floor of the mouth, or inner cheeks. But the pain they produce is disproportionate to their size, often more severe than what patients or clinicians expect from such small ulcerations. They tend to appear within the first few weeks of treatment and usually resolve within one to two weeks, though they can recur with continued dosing.
The silver lining: mIAS responds to steroids. Topical, intralesional, or systemic corticosteroids can help manage the ulcers, which distinguishes them from chemotherapy-induced oral mucositis that does not respond well to steroids. Many longevity practitioners advise swallowing rapamycin quickly rather than letting it dissolve in the mouth, as direct mucosal contact appears to worsen the problem.
| Side Effect | Transplant Doses (daily) | Low-Dose Longevity (weekly) |
|---|---|---|
| Mouth sores (mIAS) | 2-78% incidence | More common than placebo (Mannick 2014) |
| Hyperlipidemia | Common, well-documented | Increased triglycerides in some trials |
| Glucose elevation | Dose-dependent, can be severe | HbA1c creep observed, fasting glucose often stable |
| Wound healing delay | Well-documented surgical concern | Limited data at low doses |
| Infections | Increased risk (immunosuppression) | Mixed evidence; some data shows protection |
| GI discomfort | Common | Most common minor side effect (PEARL) |
Wound healing impairment is another established concern, particularly from the transplant and surgical literature. Long-term mTOR inhibition is associated with impaired wound healing and delayed tissue repair across multiple clinical contexts. This effect makes biological sense — mTOR drives the cell proliferation needed for wound closure, so inhibiting it slows that process. For someone taking rapamycin who needs surgery or sustains an injury, this is a practical concern that requires coordination with their physician.
The PEARL trial, the longest study of rapamycin in healthy aging adults, found a somewhat reassuring picture at low doses. Among 114 participants taking 5 mg or 10 mg weekly for 48 weeks, the most frequent minor issue was mild gastrointestinal discomfort, and serious adverse events occurred at similar rates across all groups including placebo. That suggests the side effect burden drops considerably when moving from immunosuppressive doses to the lower longevity-oriented protocols — though the trial was not large enough to detect rare events.
Why Bryan Johnson quit rapamycin after five years
On September 28, 2024, Bryan Johnson stopped taking rapamycin. He announced the decision in a January 2025 blog post, making him the most high-profile person to publicly reverse course on one of longevity medicine's most discussed interventions.
Johnson had spent nearly five years testing various protocols: weekly doses at 5, 6, and 10 mg; biweekly at 13 mg; and alternating weekly schedules switching between 6 mg and 13 mg. His team's conclusion was blunt — "the benefits of lifelong dosing of Rapamycin do not justify the hefty side-effects." The specific problems he listed: intermittent skin and soft tissue infections, lipid abnormalities, glucose elevations, and increased resting heart rate.
What made his decision particularly significant was the timing. An October 2024 pre-print had analyzed 51 longevity interventional studies using 16 different epigenetic aging clocks — and rapamycin appeared among a handful of interventions that actually accelerated aging markers rather than slowing them. Johnson noted that most prior rapamycin studies had cherry-picked favorable clocks, and this broader analysis painted a different picture.
But Johnson's case comes with significant caveats that his critics have been quick to point out. He was simultaneously taking more than 50 supplements and medications, including 2,000 mg of metformin and 400 mg of acarbose daily — both of which affect glucose metabolism independently. He also practiced strict caloric restriction. Stacking multiple interventions that suppress mTOR and alter metabolism makes it genuinely difficult to isolate which one caused which side effect.
The Frontiers in Aging review that discussed Johnson's case framed it as highlighting "the risks of bypassing peer-reviewed science in favor of anecdotal biohacking culture." Clinical literature had documented every side effect Johnson reported long before he experienced them. His experience confirmed what transplant medicine already knew — but it did so in a way that reached millions of people who had never read a clinical pharmacology paper.
Dr. Matt Kaeberlein, a prominent rapamycin researcher, responded publicly by noting that Johnson's protocols used higher and more frequent doses than what most longevity-focused clinicians recommend. The growing consensus among researchers who remain enthusiastic about rapamycin is that intermittent, pulsed dosing — hitting mTORC1 briefly then allowing washout — may capture benefits while avoiding the mTORC2 disruption that causes metabolic and immune problems. Johnson's experience, in this reading, may reflect the consequences of chronic high-dose exposure rather than an indictment of rapamycin itself.
What your doctor should check before prescribing rapamycin off-label
The clinical evidence for rapamycin as an anti-aging therapy is thin. A 2025 review in the journal Aging put it plainly: "the data in humans have yet to establish that rapamycin, or its analogues, is a proven seno-therapeutic that can delay aging in healthy older adults." That does not mean it is useless — it means the evidence is not there yet, and anyone considering it is participating in an experiment whether they frame it that way or not.
The PEARL trial provides the strongest safety signal so far. Over 48 weeks with 114 participants, low-dose weekly rapamycin was safe and well-tolerated, with serious adverse events occurring at rates no higher than placebo. Women taking 10 mg weekly showed significant gains in lean muscle mass and reported less pain. The 5 mg group reported improvements in emotional well-being. These are encouraging signals, but from a single, relatively small trial with a health-conscious participant pool.
If you are discussing rapamycin with a physician, several factors should shape that conversation.
Start with your metabolic baseline. Someone with prediabetes, insulin resistance, or elevated triglycerides is starting from a worse position for rapamycin's metabolic effects. Fasting glucose, HbA1c, lipid panels, and fasting insulin should all be tracked before and during treatment.
Liver function determines how much drug actually reaches your bloodstream. Rapamycin is metabolized by the CYP3A4 enzyme system, and variations in CYP3A4 activity mean two people taking identical doses can end up with very different blood levels. Liver conditions, medications that inhibit CYP3A4 (including CBD), and genetic polymorphisms all alter how long the drug stays active.
Dosing schedule matters more than most people realize. The emerging clinical consensus favors once-weekly or less frequent dosing over daily regimens. Research shows that daily or frequent high-dose rapamycin triggers persistent mTORC2 inhibition, the mechanism behind insulin resistance and metabolic disruption. Pulsed dosing aims to inhibit mTORC1 briefly and allow recovery before the drug reaches mTORC2.
Drug interactions deserve more scrutiny than they typically get. Anyone taking metformin, acarbose, statins, or CBD products alongside rapamycin is creating interaction potential that has not been studied in combination for anti-aging purposes. Only about 30% of off-label prescribing is backed by adequate scientific evidence according to one analysis, and rapamycin-for-longevity falls squarely in the other 70%.
Then there is the access problem. Online longevity clinics offer rapamycin with varying levels of medical oversight. Costs range from $64 per month for the drug itself to $124 to over $700 per month when membership fees are included. Researchers have raised concerns about diverting limited drug supply from transplant patients and others with approved medical needs, echoing similar tensions with GLP-1 agonists like semaglutide.
Rapamycin remains the most promising pharmacological candidate for human longevity, backed by stronger animal data than any competitor. But the gap between mouse lifespan extension and proven human benefit has not been bridged. The side effects are real, dose-dependent, and manageable for most people who are being properly monitored. The operative phrase there is "properly monitored" — and that requires more than a subscription to an online clinic.
Frequently Asked Questions
Is rapamycin the same drug used in organ transplant patients?
Yes. Rapamycin (brand name Rapamune, generic name sirolimus) is FDA-approved for preventing organ transplant rejection. Transplant patients take it at higher daily doses for immunosuppression. The longevity community uses the same drug at lower doses, typically once per week, aiming for different biological effects — specifically mTORC1 inhibition to promote autophagy rather than broad immune suppression.
Can rapamycin cause diabetes?
Rapamycin can raise blood glucose and HbA1c levels, especially with chronic high-dose use. Whether this constitutes true diabetes or a reversible metabolic shift (what some researchers call "starvation pseudo-diabetes") is debated. The metabolic changes documented in human trials have been mild to moderate and appear to reverse when the drug is discontinued. People with pre-existing insulin resistance face higher risk from these effects.
How much does off-label rapamycin cost?
The drug itself can cost as little as $64 per month through online longevity clinics. However, most clinics charge additional membership or consultation fees ranging from $124 to over $700 per six-month cycle. Lab monitoring (lipid panels, glucose tests, CBC) adds further costs that vary by provider and insurance coverage.
Did Bryan Johnson's side effects prove rapamycin is dangerous?
Not conclusively. Johnson reported real side effects — infections, glucose elevation, lipid changes, increased resting heart rate — that are consistent with known rapamycin risks. However, he was simultaneously taking over 50 supplements and medications, making it impossible to isolate rapamycin as the sole cause. His experience represents a single case study, not clinical proof of danger. It does underscore the importance of medical monitoring and cautious dosing.
What is the safest rapamycin dosing protocol for longevity?
No protocol has been proven "safe" for long-term anti-aging use because the trials have not run long enough. Current clinical opinion favors intermittent dosing (once weekly or less) at 5 to 10 mg rather than daily use. The PEARL trial used 5 mg and 10 mg weekly for 48 weeks with a favorable safety profile. Pulsed dosing aims to inhibit mTORC1 briefly while avoiding chronic disruption of mTORC2, which drives metabolic side effects.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.