Your cells have a backup copy of youth -- and scientists just figured out how to restore it
Every cell in your body carries the same DNA you were born with. What changes as you age is the way that DNA gets read. Chemical tags called methyl groups accumulate on your genes over decades, silencing some and activating others in patterns that slowly degrade cellular function. Histone proteins that spool your DNA get modified in ways that stiffen the chromatin structure, locking cells into an aged state. These epigenetic alterations -- changes to gene expression without changes to the DNA sequence itself -- are now understood to be a root driver of aging, not merely a consequence of it.
Think of it like a piano. The instrument itself stays the same, but over time someone keeps taping down keys and muting strings. The music gets worse -- not because the piano is broken, but because the instructions for playing it have degraded. Epigenetic reprogramming is the attempt to peel off that tape.
The technique traces back to a 2006 discovery by Professor Shinya Yamanaka, who identified four transcription factors -- OCT4, SOX2, KLF4, and c-MYC, collectively called OSKM -- that could reset any adult cell to an embryonic-like state. The discovery earned Yamanaka the Nobel Prize and launched an entirely new field. But there was a problem. Full reprogramming erased a cell's identity entirely. A heart cell, for instance, would forget it was a heart cell. Worse, in living animals, activating all four factors triggered rapid tumor formation.
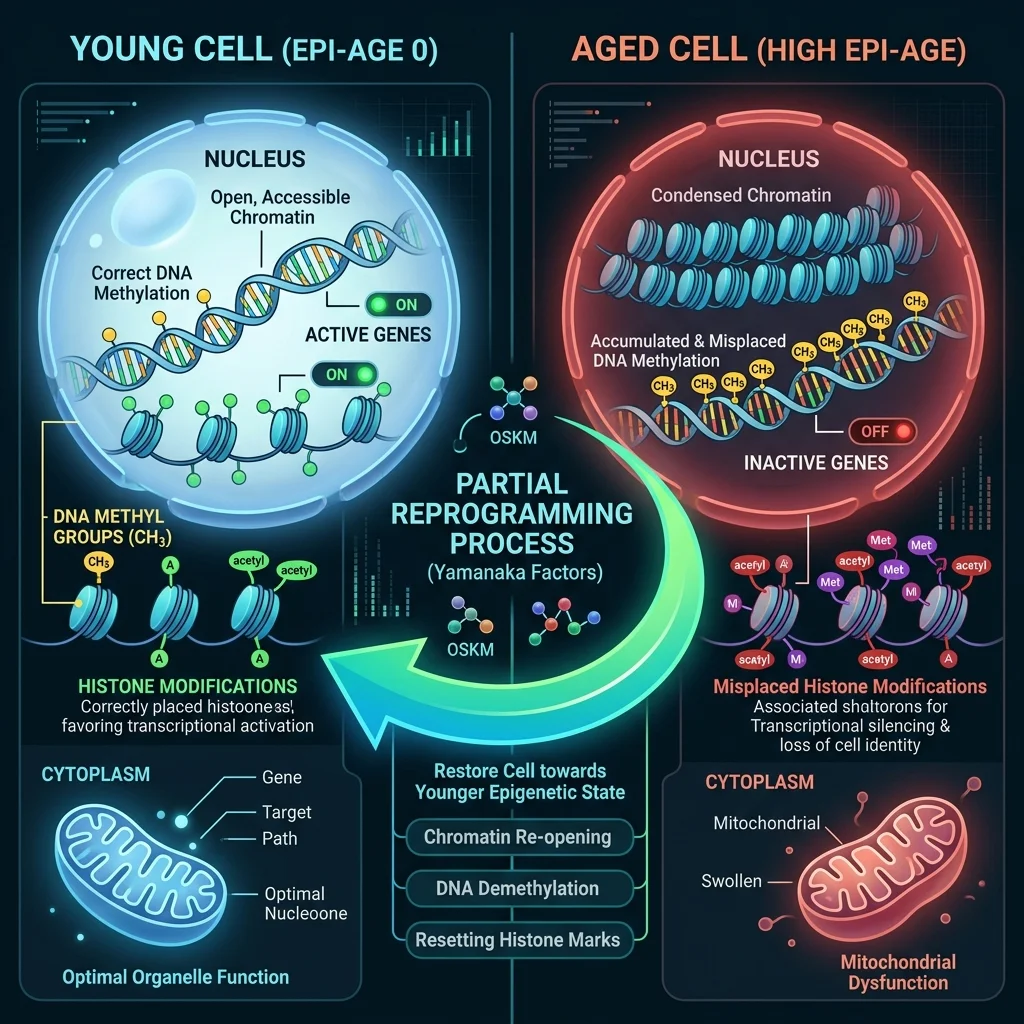
That barrier led researchers to a subtler idea: partial reprogramming. Instead of pushing cells all the way back to an embryonic state, you expose them briefly to a subset of Yamanaka factors, just long enough to strip away the accumulated epigenetic damage without erasing the cell's memory of what it is. A 2024 review in Nature Communications describes partial reprogramming as inducing a "shift in epigenetic management of nuclear DNA structure to a more youthful state, while avoiding any dedifferentiation." The cell gets younger. It stays a heart cell, or a neuron, or a retinal ganglion cell.
The mouse data is striking. When researchers applied cyclic doses of OSKM factors to progeric mice (which age rapidly), they observed a 33% increase in median lifespan. A separate study using only OSK factors -- dropping the cancer-linked c-MYC -- delivered via AAV9 gene therapy extended the remaining lifespan of 124-week-old wild-type mice by 109%. The treated mice showed rejuvenated transcriptomes, metabolomes, and lipidomes across multiple tissues. For the reader without a molecular biology background: the chemical profile of their cells looked younger by multiple measures, not just one.
What this means practically: aging may not be the irreversible decline we assumed. If the epigenetic code can be reset in cells that are still alive and functioning inside the body, then at least some aspects of biological aging are a software problem -- corrupted instructions that can be corrected -- rather than permanent hardware damage.
12 patients, one eye each: inside the trial that could change everything
On January 28, 2026, Life Biosciences announced that the FDA had cleared its Investigational New Drug application for ER-100, making it the first cellular rejuvenation therapy using epigenetic reprogramming to enter human clinical trials. The Phase 1 study, registered as NCT07290244, officially started on March 2, 2026.
Life Biosciences is a Boston-based startup cofounded by Harvard geneticist David Sinclair, who has spent years advancing the theory that epigenetic information loss is the primary cause of aging. The company has raised $158 million in total funding, including an $82 million Series C round in 2022. After an early strategy of launching multiple subsidiaries (which made limited progress), the company brought in CEO Jerry McLaughlin in 2021 and refocused entirely on Sinclair's mouse vision results and the push toward a human trial.
The trial targets two age-related eye conditions: open-angle glaucoma (OAG) and non-arteritic anterior ischemic optic neuropathy (NAION). Both involve irreversible death of retinal ganglion cells -- the neurons connecting the eye to the brain. Because these cells cannot naturally regenerate, their loss causes permanent vision impairment. NAION, sometimes called a "stroke of the eye," is the most common acute optic neuropathy in adults over fifty and currently has no approved treatments.
ER-100 trial at a glance: Up to 18 participants (12 OAG, 6 NAION), ages 40-85, single injection of modified AAV vector into one eye, doxycycline activation for 8 weeks, 5-year follow-up period. Primary endpoint: safety and tolerability.
The treatment works by injecting a modified adeno-associated virus (AAV) vector carrying the OSK genes directly into one eye of each patient. According to the trial registry, the genes are activated by systemic doxycycline administered for 56 days. The trial uses a dose-escalation design: the first "sentinel" patient at each dose level is monitored for 28 days before two additional patients receive the same dose. An independent Data Safety Monitoring Board oversees the entire process.
Sharon Rosenzweig-Lipson, Life Biosciences' Chief Scientific Officer, told Lifespan.io that enrollment began in March 2026 and that the company expects enough data by the end of the year to make a Phase 2 decision. "At this stage, success means demonstrating that ER-100 can be administered safely," she said. "Any signals of improvement in visual parameters would be highly encouraging."
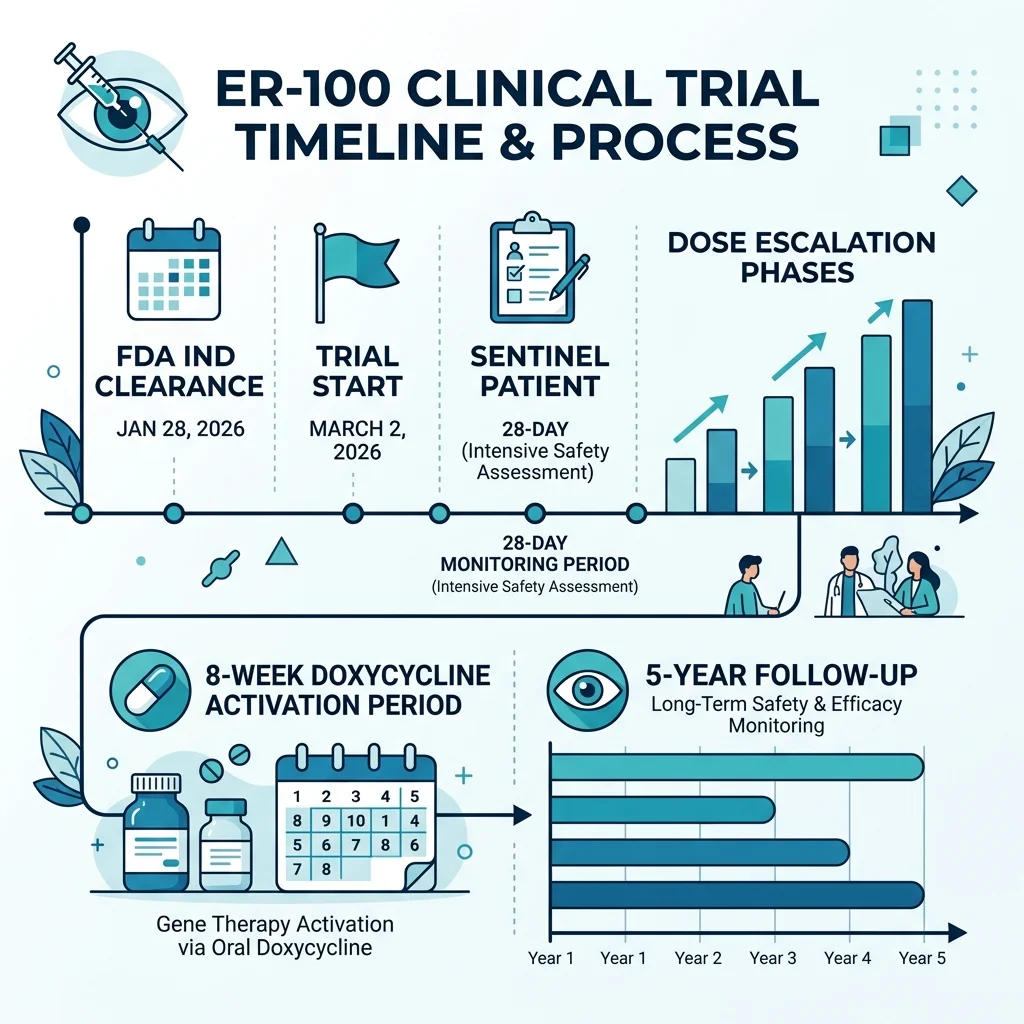
The company's preclinical work preceded this milestone. According to Life Biosciences' official announcement, nonhuman primate studies demonstrated controlled OSK expression, restoration of methylation patterns, and improved visual function. Rosenzweig-Lipson told Longevity Technology that these studies showed ER-100 was "well tolerated, with no systemic toxicities." But the company has not published these NHP results in a peer-reviewed journal -- a gap that skeptics have noticed.
David Sinclair called the moment meaningful. "It's incredibly meaningful to see this science reach clinical testing after more than 30 years of work," he told Lifespan.io. Not everyone in the field shares that level of enthusiasm about Sinclair specifically. A 2024 Wall Street Journal investigation dubbed him a "reverse-aging guru" whose companies "have not panned out," and critics note he has previously promoted sirtuins and resveratrol with similar conviction before those efforts stalled.
100+ genes that control cellular youth, and a Cambridge startup that found them
While Life Biosciences focused on getting OSK into human patients, a different question was being asked at the Milner Therapeutics Institute at the University of Cambridge: what if the three Yamanaka factors are not the best tools for rejuvenation? What if there are dozens -- or hundreds -- of genes that can safely reverse aging, and we just have not looked for them systematically?
That question drove clock.bio, a small Cambridge-based biotech founded by Mark Kotter (who also founded synthetic biology company bit.bio). The company launched out of stealth in 2023 and took an approach that, in scale, dwarfs what most reprogramming labs have attempted.
Clock.bio's team developed a proprietary model that forces human induced pluripotent stem cells (iPSCs) to age artificially. This matters because iPSCs have a built-in ability that somatic cells lack: when damaged, they can trigger a self-rejuvenation mechanism, repairing the hallmarks of aging and reverting to a young, healthy state. By forcing these cells to age and then watching which genes activated during self-repair, the company aimed to decode the complete rejuvenation playbook.
The tool they used was a genome-wide CRISPR screen -- systematically knocking out individual genes across the entire human genome to see which ones are required for the rejuvenation process. Combined with single-cell RNA-sequencing of more than 3 million cells, generating 20 terabytes of data, the project identified more than 100 genes that together form what clock.bio calls an "Atlas of Rejuvenation Factors."

"Running CRISPR screens across the full human genome constitutes an unbiased, comprehensive methodology, which now gives us more than one shot on target," said clock.bio CEO Markus Gstottner in the company's funding announcement. The company raised $5.3 million in seed funding led by LocalGlobe, with additional backing from BlueYard Capital and Onsight Ventures.
The practical implication is significant. If the Atlas holds up to validation in somatic cells (the next step clock.bio is pursuing), it could open the door to repurposing existing drugs that target these rejuvenation pathways -- bypassing the need for gene therapy entirely. Instead of injecting viral vectors carrying OSK, a patient might take a pill that activates one of these 100+ rejuvenation genes through a conventional pharmaceutical mechanism. That remains hypothetical, but if even a small fraction of the 100+ genes pan out, the anti-aging field would have a far larger toolkit than the three Yamanaka factors everyone has been working with.
One caveat: clock.bio's Atlas has not been published as a peer-reviewed paper. The company has disclosed its methodology and headline numbers through press releases and conference presentations, but the full dataset and independent validation remain pending. That does not mean the work is unreliable -- it means it has not yet faced the scrutiny that peer review provides.
The cancer problem, and why dropping one gene changed everything
The fear that keeps every reprogramming researcher awake at night is teratomas -- tumors that form when cells lose their identity and start proliferating uncontrollably. When Yamanaka first demonstrated full reprogramming with all four factors (OSKM), the c-MYC gene was the primary troublemaker. It is a known oncogene, and when activated alongside the other three factors in living animals, it can cause an eruption of tumors.
Life Biosciences' solution was straightforward: drop c-MYC from the cocktail entirely. Rosenzweig-Lipson described the logic to Lifespan.io in blunt terms: "Taking the M out makes it impossible to go all the way back. You just can't push the system hard enough." Without c-MYC, the three remaining factors (OSK) reset the epigenetic code of aging cells without triggering the runaway dedifferentiation that leads to cancer. Cell identity is maintained.
But dropping c-MYC is only half the safety architecture. The other half is the doxycycline-inducible switch. ER-100's OSK genes are designed to remain dormant until the patient takes a course of the common antibiotic doxycycline. This gives clinicians a kill switch: stop the antibiotic, and gene expression stops. Life Biosciences says its mouse data supports continuous OSK activation for three months or beyond with both good reprogramming and good safety.
| Safety Concern | How ER-100 Addresses It |
|---|---|
| Tumor formation from full reprogramming | Uses only OSK (no c-MYC oncogene) |
| Loss of cell identity (dedifferentiation) | OSK resets epigenetic code without reaching pluripotency |
| Uncontrolled gene expression | Doxycycline-inducible switch -- stop the antibiotic, stop the genes |
| Systemic toxicity | Local injection into one eye only; NHP studies showed no systemic toxicities |
| Long-term unknown effects | 5-year monitoring with independent Data Safety Monitoring Board |
The risks are not zero. Noah Davidsohn, who helped Sinclair implement the original technique and is now chief scientist at Rejuvenate Bio, raised concerns about the doxycycline switch itself. The switch is built from gene components derived from E. coli and the herpes virus, and it has never been tested in humans. There is a real possibility that the human immune system could mount a reaction against these foreign genetic components.
Beyond the eye, the challenges multiply. A 2026 review in Trends in Molecular Medicine identified several major obstacles for whole-body reprogramming. Systemic doxycycline delivery produces unequal gene activation across tissues. The liver and intestines are especially vulnerable because they absorb doxycycline rapidly and have high intrinsic epigenetic flexibility -- meaning they reprogram first and most aggressively, potentially causing malabsorption and toxicity before other organs benefit. And reprogramming remains stochastic: only a fraction of cells respond as intended, making outcomes inherently unpredictable.
This is precisely why the eye was chosen as the first target. It is a contained compartment -- accessible, monitorable, and isolated from the rest of the body. Daniel Ives, CEO of Shift Bioscience, put it with characteristic directness when discussing Life Biosciences' approach with MIT Technology Review: "They have found a route forward in the eye, which is a nice self-contained system. If it goes wrong, you've still got one left."
$4.5 billion and counting: who's funding the cellular time machine
The amount of money flowing into epigenetic reprogramming is staggering for a field where not a single human therapy has been proven to work. A handful of companies are dominating the space, each with different strategies and different timelines to the clinic.
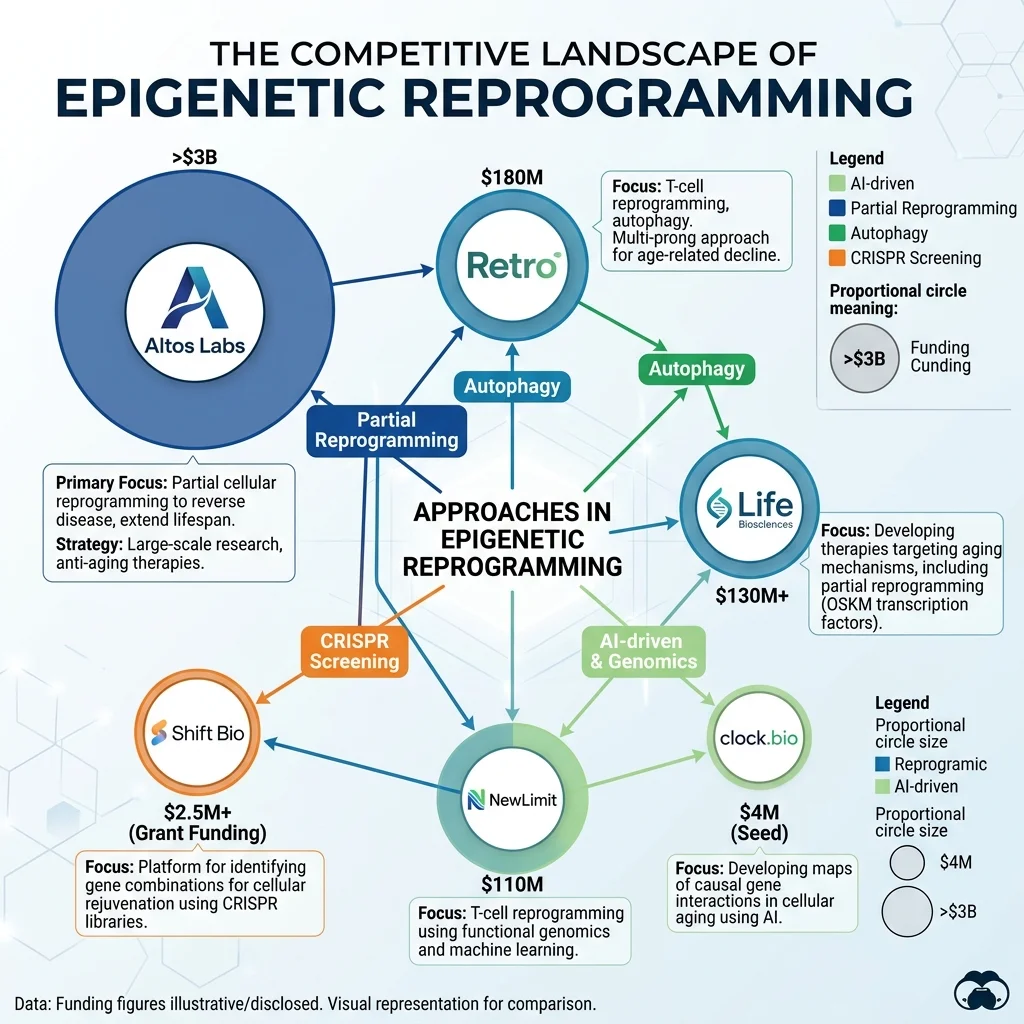
Altos Labs sits at the top of the funding pyramid. The company launched in 2022 with a reported $3 billion in capital, with Amazon founder Jeff Bezos among its key investors. Organized across research institutes in San Francisco, San Diego, and Cambridge (UK), Altos has recruited some of the world's leading aging researchers. In 2024, Altos published data showing that targeted partial reprogramming of age-associated cell states extended the lifespan of mice. More recently, the company has been testing its therapies in organs removed from the body and kept alive on perfusion machines -- an approach that could provide detailed safety data without risking living patients.
Retro Biosciences, funded with $180 million from OpenAI CEO Sam Altman, is pursuing three parallel strategies: autophagy enhancement, cellular reprogramming, and microglia therapeutics. The company dosed its first patient in a Phase 1 trial of RTR242 -- a small molecule designed to restore lysosomal function in neurons -- in late 2025. RTR242 is aimed at Alzheimer's disease, with the trial conducted in Adelaide, Australia. Retro has also partnered with OpenAI to apply AI to protein engineering, producing engineered transcription factors that improved stem cell production efficiency by up to 50-fold. Reports indicate Retro is preparing to raise as much as $1 billion for its next growth phase.
| Company | Approach | Lead Program | Total Funding | Stage |
|---|---|---|---|---|
| Altos Labs | Partial epigenetic reprogramming | Undisclosed (organ perfusion testing) | $3 billion | Preclinical |
| Life Biosciences | OSK partial reprogramming | ER-100 for optic neuropathies | $158 million | Phase 1 (active) |
| Retro Biosciences | Autophagy + reprogramming + microglia | RTR242 for Alzheimer's | $180 million | Phase 1 (active) |
| NewLimit | Epigenetic reprogramming + ML | Alcohol-related liver disease | ~$280 million | Preclinical (clinic in 2-3 years) |
| Shift Bioscience | AI + aging clock gene discovery | SB-000 for hearing loss | $16 million | Preclinical (animal models) |
| clock.bio | CRISPR rejuvenation atlas | Drug repurposing from 100+ gene targets | $5.3 million | Target validation |
NewLimit, with roughly $280 million in total funding (including a $130 million Series B), is taking a more methodical route. Co-founded by Coinbase CEO Brian Armstrong, the company uses single-cell genomics, epigenetic editing, and machine learning to identify safer reprogramming strategies. Its first clinical target is alcohol-related liver disease, with plans to enter the clinic within the next few years. NewLimit won't be ready for a human study for another two years, according to MIT Technology Review.
Shift Bioscience, founded by Daniel Ives, has raised $16 million and built a cell simulation platform that combines generative AI with a biological aging clock. The approach predicts which gene sets are most likely to safely rejuvenate specific cell types. Shift's first anti-aging target, SB-000, is a gene therapy aimed at age-related hearing loss. The company has also identified pro-aging targets for RNAi knockdown in liver fibrosis. Shift remains in early preclinical stages, with animal model work ongoing.
Investor Karl Pfleger captured the field's energy when he told MIT Technology Review: "Reprogramming is like the AI of the bio world. It's the thing everyone is funding." The analogy is apt in more ways than one -- both fields attract massive capital, enormous hype, and genuine uncertainty about which approaches will actually deliver.
Between the hype and the clinic: what actually happens by 2031
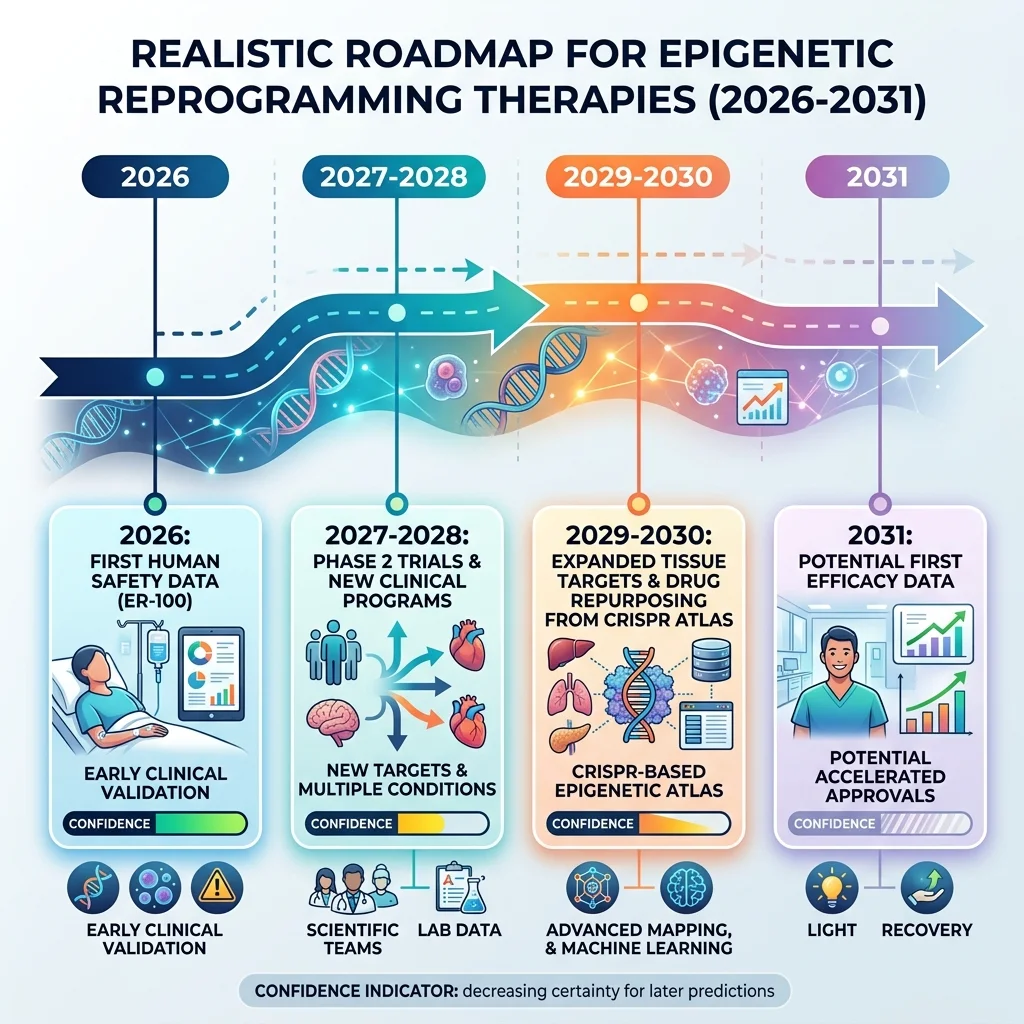
The honest timeline for epigenetic reprogramming is both more exciting and more sobering than most headlines suggest. Here is what is realistic, what is possible, and what remains out of reach for the next five years.
The near-certainty: by late 2026, we will have the first human safety data for partial epigenetic reprogramming. Life Biosciences' ER-100 trial is actively enrolling, and Rosenzweig-Lipson has indicated that enough data to support a Phase 2 decision should be available by year-end. Even if the trial shows only safety (with no vision improvement), it will have answered the most basic question: can you put reprogramming factors into a living human without causing harm? That answer alone would reshape the entire field.
The probable: additional clinical programs will enter human testing. Life Biosciences has disclosed promising preclinical data on liver reprogramming, particularly for metabolic dysfunction-associated steatohepatitis (MASH). If ER-100's eye data is clean, a liver-targeted program could follow within two years. Retro Biosciences will have Phase 1 results from RTR242. NewLimit expects to reach the clinic. Yuri Deigin, CEO of YouthBio, told Lifespan.io that regulators are "increasingly willing to evaluate therapies that aim to modify upstream epigenetic drivers of aging, rather than only treating downstream symptoms" -- a signal that the FDA's openness to Life Biosciences may extend to other companies.
The possible but uncertain: clock.bio's Atlas of 100+ rejuvenation genes could yield drug targets that bypass gene therapy entirely. If even a handful of these genes can be activated by existing small molecules, the path from discovery to clinic could be dramatically shorter than the gene therapy route. But the Atlas is unvalidated in somatic cells, unpublished in peer review, and converting a gene target into a drug candidate is measured in years, not months.
What remains out of reach: whole-body rejuvenation. The 2026 Trends in Molecular Medicine review is clear that systemic partial reprogramming "remains far from clinical readiness." The challenges of unequal tissue delivery, unpredictable organ-specific responses, and the liver/intestine vulnerability problem have no current solutions. Progress will require better delivery systems, tissue-specific gene promoters, real-time biomarkers of epigenetic age, and nonintegrating genetic tools -- technologies that are under development but not yet mature.
The field's biggest vulnerability is not scientific -- it is reputational. Sinclair has been criticized for overpromising before. If ER-100 fails or produces serious side effects, it could chill investment and regulatory willingness across the entire reprogramming space. Conversely, even modest positive signals -- a small improvement in visual function, a measurable reversal of epigenetic age markers in treated tissue -- would validate years of preclinical work and likely trigger a wave of follow-on trials.
For anyone watching this field: the next 18 months matter more than the previous decade. The mouse data was convincing. The investment is in place. The regulatory path has been cleared. What we do not yet have is a single data point from a human patient showing that partial epigenetic reprogramming works safely inside a living person. By 2027, we will.
Frequently Asked Questions
What is epigenetic reprogramming and how does it differ from genetic modification?
Epigenetic reprogramming changes how your genes are read without altering the DNA sequence itself. Your genes accumulate chemical tags (like methyl groups) over your lifetime that affect which genes are active or silent. Reprogramming strips away some of these accumulated changes, restoring a more youthful pattern of gene expression. Genetic modification, by contrast, permanently changes the DNA sequence. As the ER-100 trial registry states, the therapy "does not alter the participant's existing genes."
Is the ER-100 trial actually reversing aging, or just treating eye disease?
Officially, it is treating eye disease. The FDA approved the trial to test safety in patients with glaucoma and NAION, not as an anti-aging intervention. But the underlying mechanism -- resetting the epigenetic state of retinal cells to a younger configuration -- is fundamentally an age-reversal approach applied to a specific tissue. If it works, the same principle could be applied to other age-related conditions. Life Biosciences' CEO called the IND clearance "a major inflection point for the longevity and aging biology field," not just ophthalmology.
When will anti-aging reprogramming therapies be available to the general public?
Not for several years at minimum. The ER-100 trial is Phase 1 (safety only), and even optimistic timelines require Phase 2 and Phase 3 trials before any therapy reaches the market. For eye-specific treatments, a best-case scenario might see approval by the late 2020s or early 2030s. Whole-body rejuvenation therapies face additional barriers including delivery challenges across tissues and are described by researchers as "far from clinical readiness."
What are the biggest risks of epigenetic reprogramming?
The primary risk is that reprogrammed cells could lose their identity and become cancerous. Full reprogramming with all four Yamanaka factors has caused tumor formation in animal models. ER-100 mitigates this by using only three factors (no c-MYC), delivering locally to one eye, and using a doxycycline switch that can halt gene expression. However, the doxycycline-inducible switch itself uses genetic components from bacteria and viruses that have never been tested in humans, creating potential immune reaction risks.
How does clock.bio's approach differ from the Yamanaka factor method?
The Yamanaka factor method uses a fixed set of three or four known transcription factors to reprogram cells. Clock.bio's approach starts from the opposite direction: rather than imposing external factors, they mapped which genes human stem cells naturally use to rejuvenate themselves after damage. Their genome-wide CRISPR screen identified 100+ rejuvenation genes, potentially allowing researchers to activate the body's own rejuvenation pathways through drugs rather than gene therapy.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.