Half the World Carries This Bacterium — Most Have No Idea
Somewhere around 4.4 billion people on Earth harbor Helicobacter pylori in their stomachs right now. That figure is staggering on its own, but what makes it stranger is that most of them will never feel a thing. This spiral-shaped bacterium has been living in the human gut for at least 60,000 years, and it has perfected the art of persistence — burrowing into the protective mucous lining, neutralizing stomach acid with its own enzymes, and settling in for what can be a lifelong residence.
For the majority of carriers, H. pylori sits quietly. But for roughly 10-20% of those infected, the relationship turns adversarial. The bacterium triggers chronic inflammation that can erode the stomach lining, produce peptic ulcers, and — in about 1% of cases — progress toward gastric cancer. The World Health Organization designated H. pylori a Class I carcinogen back in 1994, placing it in the same category as tobacco smoke and asbestos.
What has shifted in the past decade is not our understanding of the bacterium itself, but our ability to eliminate it. The standard antibiotic regimens that once cleared infections in 80-90% of patients have seen their success rates slide below 70% in many regions. This has opened space for a more nuanced conversation — one that includes natural compounds, strategic probiotic use, and the overlooked question of what happens to the gut after the bacterium is gone.
Recognizing the Warning Signs Before They Escalate
The challenge with H. pylori is that its symptoms overlap with a dozen other digestive complaints. Many people spend months — sometimes years — chalking up their discomfort to stress, diet, or aging before getting tested. Knowing what to watch for can shorten that delay considerably.
The Cleveland Clinic identifies a core set of symptoms tied to active H. pylori infection: a dull or burning ache in the stomach that tends to worsen on an empty stomach, bloating that lingers after meals, unexplained nausea, early satiety (feeling full after eating very little), and unintentional weight loss. Dark or tarry stools signal bleeding in the upper digestive tract and warrant immediate medical attention.
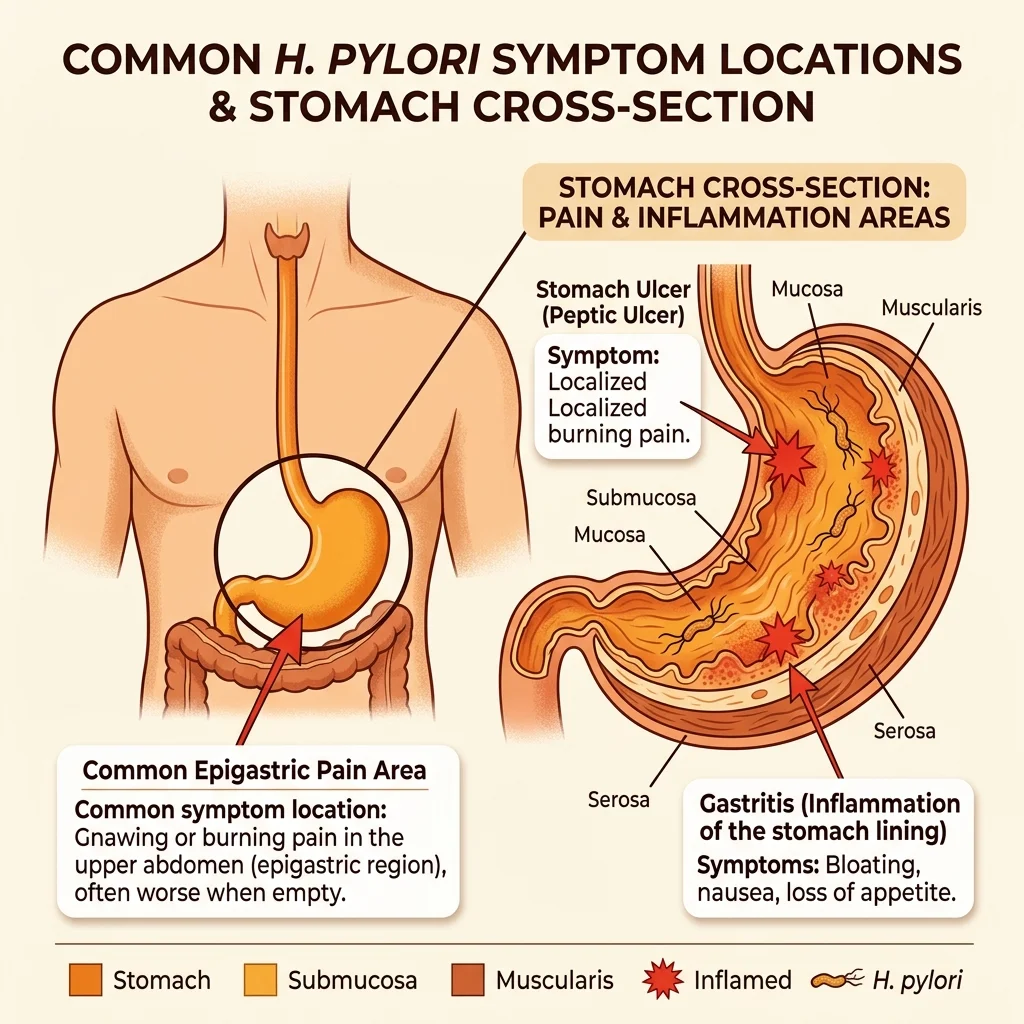
What makes diagnosis trickier is that these symptoms can come and go. A flare might last days, then disappear for weeks. This intermittent pattern frequently leads people to dismiss the problem or self-treat with antacids — which temporarily relieve symptoms but do nothing about the underlying infection.
| Symptom | What It Feels Like | When It Typically Occurs |
|---|---|---|
| Epigastric pain | Burning or gnawing ache below the ribcage | 2-3 hours after eating or during the night |
| Bloating | Persistent fullness and abdominal distension | After meals, especially large ones |
| Nausea | Queasy sensation, occasionally with vomiting | Morning or between meals |
| Early satiety | Feeling full after a few bites | During meals |
| Unexplained weight loss | Gradual drop without dietary changes | Over weeks to months |
| Dark stools | Black, tarry consistency | Indicates active bleeding — seek care immediately |
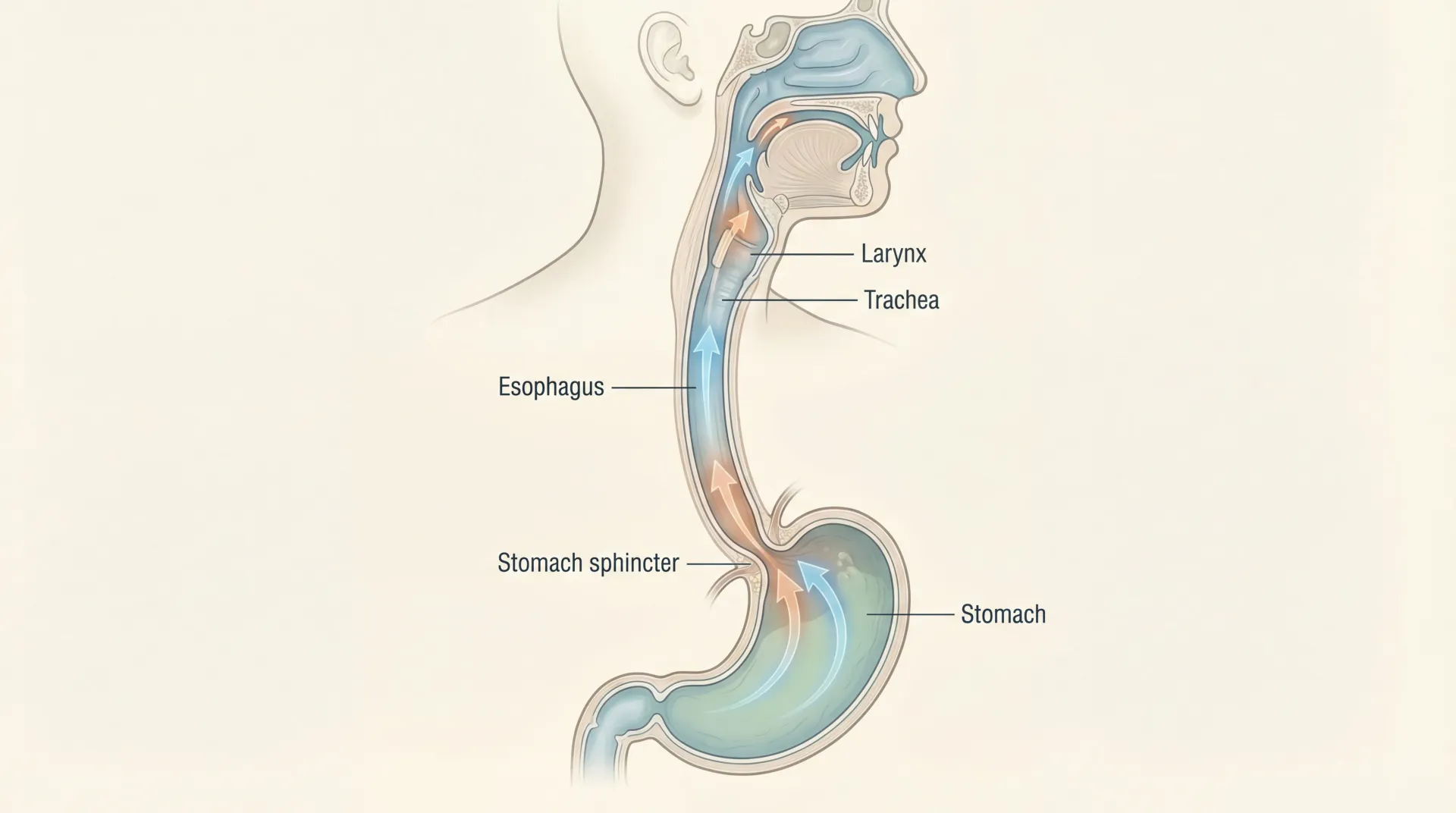
Four testing methods can confirm H. pylori: the urea breath test (you drink a solution and breathe into a bag), a stool antigen test, blood antibody testing, and upper endoscopy with biopsy. Of these, the breath test and stool antigen are preferred because they detect active infection. Blood tests can remain positive long after the bacterium is gone, which limits their usefulness for confirming eradication.
Key point: If you have persistent stomach pain that worsens on an empty stomach, bloating that does not respond to dietary changes, or unexplained weight loss, ask your doctor specifically for an H. pylori stool antigen test or urea breath test. Do not rely on symptom-based diagnosis alone.
Why Standard Treatment Keeps Failing So Many People
For decades, the playbook was straightforward: prescribe two antibiotics (typically clarithromycin plus amoxicillin or metronidazole) alongside a proton pump inhibitor, run the course for 14 days, and retest. This triple therapy worked well into the early 2000s. It does not work well anymore — and understanding why matters for anyone weighing their treatment options.
The primary culprit is antibiotic resistance. A comprehensive review in Frontiers in Microbiology documented the scope of the problem across regions:
| Region | Clarithromycin Resistance | Metronidazole Resistance | Levofloxacin Resistance |
|---|---|---|---|
| Europe | 21.4% | 38.9% | 15.8% |
| United States | 17.4% | 43.9% | 57.8% |
| Asia | 17% | 44% | 18% |
| Latin America | 12% | 53% | 15% |
| Africa | 15% | 91% | 14% |
When the main antibiotics in your regimen face resistance rates above 15%, the math gets unforgiving. But resistance is not the only reason treatment fails. H. pylori has biological survival tricks that go beyond simple antibiotic resistance. The bacterium can shift from its active spiral shape into a dormant coccoid form — playing dead, in effect — and resume activity once antibiotic pressure lifts. It also forms biofilms, protective colonies embedded in a matrix that antibiotics struggle to penetrate.
Host factors add another layer. Genetic variations in the CYP2C19 enzyme affect how quickly your body metabolizes proton pump inhibitors. Fast metabolizers burn through the PPI before it can suppress enough acid to let antibiotics work. Vitamin D deficiency, smoking, and incomplete course adherence further reduce success rates.
Between bacterial adaptation and host variability, a one-size-fits-all approach to H. pylori is becoming medically outdated. Drug sensitivity testing — where a sample is cultured and tested against specific antibiotics — now achieves success rates above 90%, but it requires lab infrastructure that remains unavailable in many clinical settings. For people dealing with recurrent infection or conventional treatment failure, this gap between what is possible and what is accessible is exactly where natural adjunct therapies enter the picture.
Evidence-Based Natural Eradication Protocols
A critical distinction up front: no natural compound tested in clinical trials eradicates H. pylori as reliably as antibiotics when used alone. The research on standalone natural treatments shows eradication rates between 16% and 56%. That is not a number that justifies replacing your doctor's prescription.
Where the science gets compelling is in combination. When specific natural agents are added alongside conventional antibiotic therapy, eradication rates climb — sometimes dramatically — while side effects drop. Think of these compounds as force multipliers rather than standalone weapons.
Sulforaphane from Broccoli Sprouts
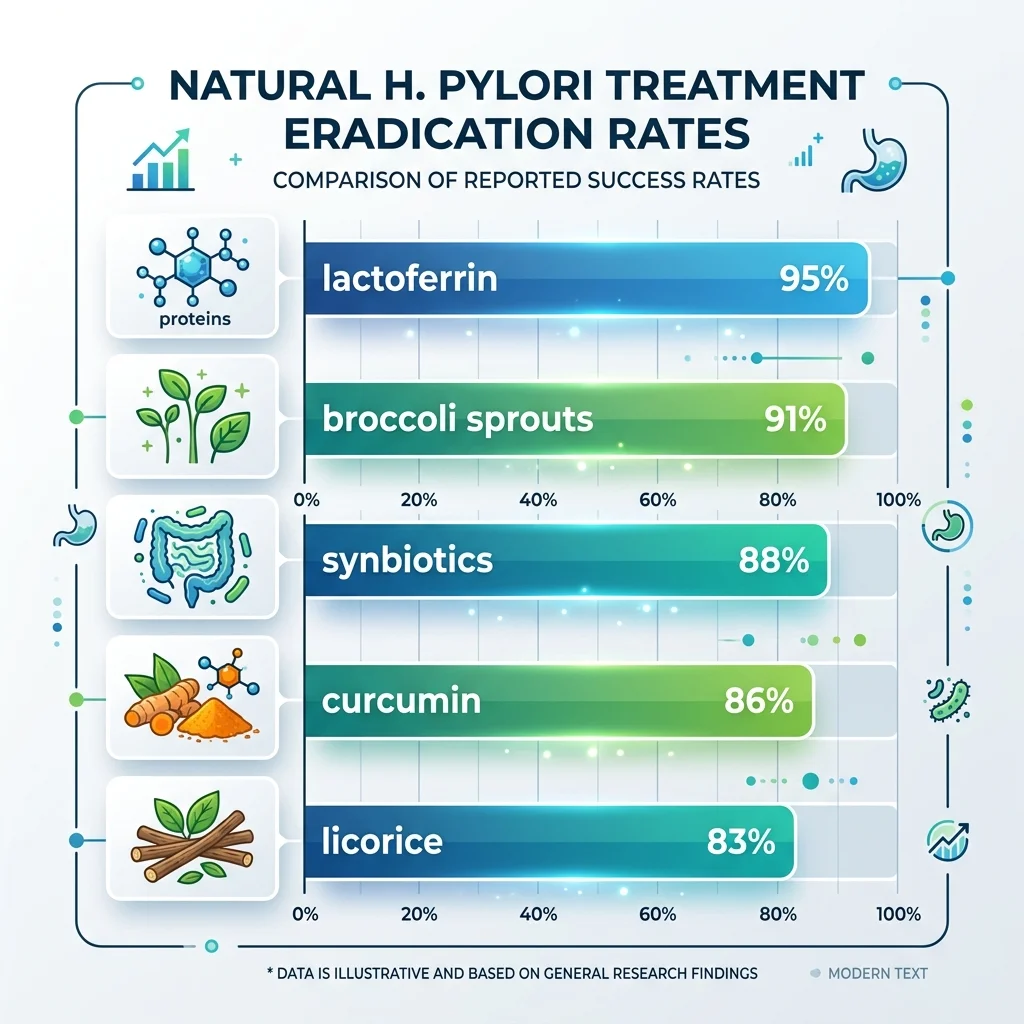
Sulforaphane, the primary bioactive compound in broccoli sprouts, may be the most clinically validated natural agent against H. pylori. It works by inhibiting the bacterium's urease enzyme — the same tool H. pylori uses to neutralize stomach acid and survive. In a controlled human trial, participants consuming 70 grams of glucoraphanin-rich broccoli sprouts daily for eight weeks showed significant reductions in bacterial colonization markers. When combined with triple therapy, a systematic review found broccoli sprouts pushed eradication from 56% to 91.7% — a substantial improvement.
One caveat worth noting: colonization reductions in the standalone studies reversed after participants stopped consuming the sprouts. This means consistent daily intake during and after a treatment course matters more than sporadic use.
Probiotics and Synbiotics
The probiotic data is broad and increasingly robust. Lactobacillus and Bifidobacterium species combat H. pylori through multiple pathways: they compete for attachment sites on the stomach lining, produce bacteriocins (natural antimicrobial peptides), suppress urease activity, and reduce the inflammatory cytokine IL-8 that H. pylori exploits to establish itself. One review found that adding dual probiotics to standard therapy improved eradication by 21% compared to antibiotics alone.
Synbiotics — probiotics combined with prebiotic fiber — performed even better. In clarithromycin-based regimens, synbiotic supplementation achieved 88.4% eradication versus 68.8% in the control group. The specific strain Lactobacillus reuteri DSM17648 has drawn particular research attention because it physically co-aggregates with H. pylori cells, clumping with them and helping flush them from the stomach environment.
Probiotics also serve a defensive function during treatment. Antibiotic courses devastate the broader gut microbiome — a side effect that often manifests as diarrhea, bloating, and compromised intestinal barrier function. Supplementing with targeted strains during antibiotic treatment helps buffer that collateral damage.
Curcumin
Curcumin, the yellow pigment in turmeric, has strong laboratory credentials against H. pylori — it inhibits every strain tested in vitro. Its mechanism is distinctive: curcumin suppresses the NF-κB inflammatory pathway that H. pylori activates to maintain its foothold, and it kills the bacterium within 15-30 minutes in direct-contact laboratory tests.
In clinical settings, curcumin's value is as an adjunct. Combined with standard triple therapy, it raised eradication rates from 74.5% to 86.4% in a controlled trial. Used alone, curcumin achieved only about 5.9% eradication — an important number to remember when evaluating claims about turmeric as a standalone H. pylori cure.
Lactoferrin
Bovine lactoferrin, a protein found naturally in milk, demonstrated some of the most impressive combination data. By binding to bacterial cell wall components, it appears to increase antibiotic penetration into H. pylori cells, effectively making antibiotics more efficient. Studies reported eradication rates between 93% and 100% when lactoferrin was added to triple therapy.
Licorice Root (Deglycyrrhizinated)
Deglycyrrhizinated licorice (DGL) — the form with the blood-pressure-raising glycyrrhizin removed — works through anti-adhesion mechanisms. It prevents H. pylori from binding to the gastric mucosa, leaving the bacteria more exposed to both immune defenses and antibiotics. GutGard, a standardized licorice extract, achieved 56% negative stool antigen results as a standalone agent versus 4% for placebo. When paired with clarithromycin-based therapy, licorice increased eradication from 62.5% to 83.3%.
Vitamin C and E
Antioxidant vitamins may help by reducing the oxidative stress that H. pylori uses to damage gastric tissue. A clinical trial found that adding vitamins C and E to conventional therapy lifted eradication from 48.8% to 78% — a significant adjunctive effect, and one of the simplest interventions to implement.
| Natural Agent | Standalone Eradication | Combined with Antibiotics | Primary Mechanism |
|---|---|---|---|
| Broccoli sprouts (sulforaphane) | ~30% colonization reduction | 91.7% | Urease inhibition |
| Dual probiotics | Not eradicative alone | +21% over antibiotics alone | Competitive exclusion, bacteriocins |
| Synbiotics | Not eradicative alone | 88.4% vs 68.8% control | Prebiotic + probiotic synergy |
| Curcumin | 5.9% | 86.4% vs 74.5% control | NF-κB suppression, direct bactericidal |
| Bovine lactoferrin | Not tested alone | 93-100% | Cell wall binding, antibiotic potentiation |
| DGL licorice | 56% (stool antigen) | 83.3% vs 62.5% control | Anti-adhesion |
| Vitamins C + E | Not eradicative alone | 78% vs 48.8% control | Oxidative stress reduction |
The Recovery Phase Most People Skip
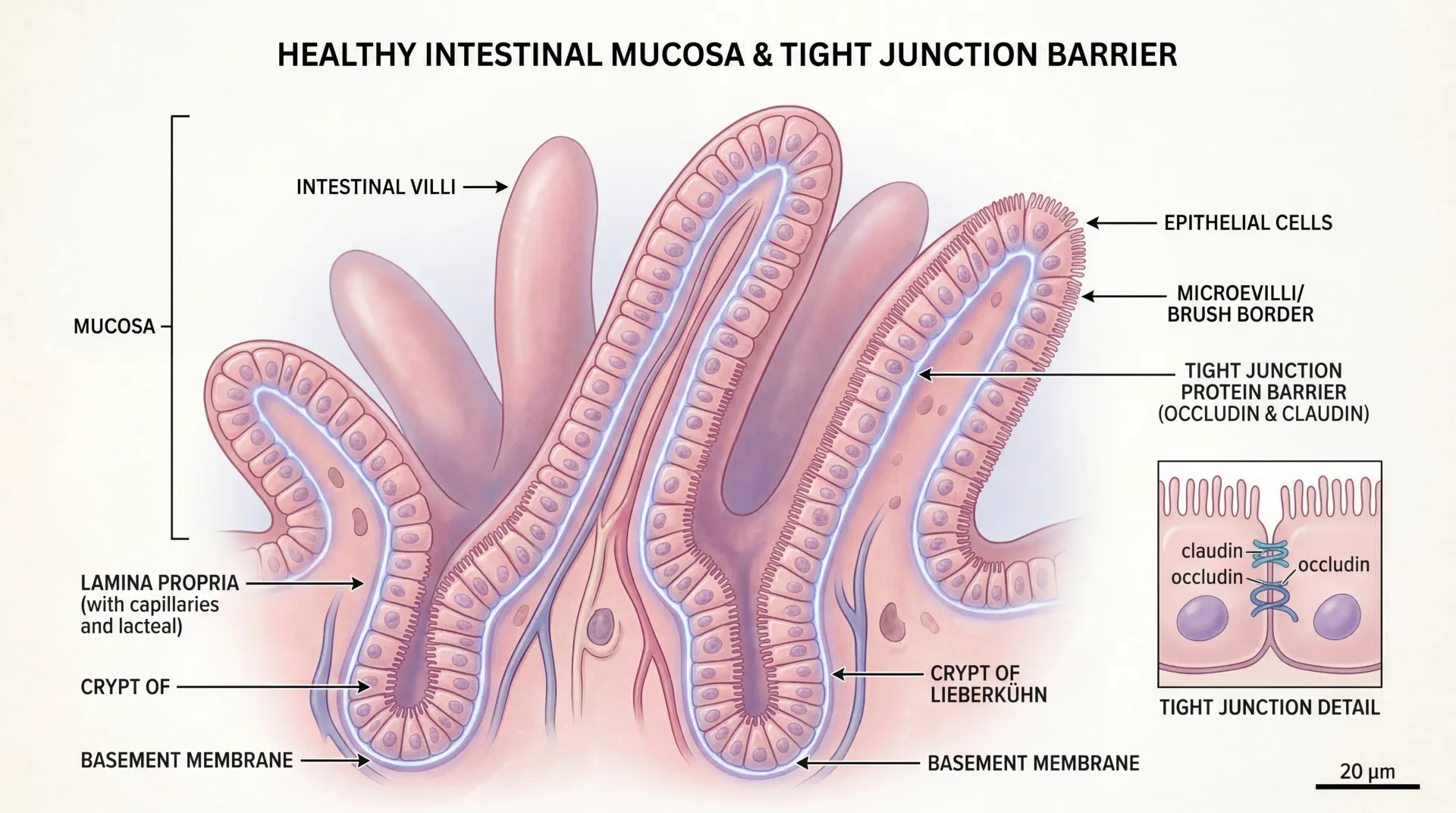
Clearing H. pylori is only half the job. What happens to your gut after eradication may matter just as much for your long-term digestive health — and it is the phase that almost nobody talks about with their doctor.
Research published in the World Journal of Gastroenterology documented that gut microbiome changes following H. pylori eradication persist for at least six months. In roughly half of studied cases, eradication led to a dominance of proinflammatory Acinetobacter species in the stomach lining, accompanied by a decrease in overall microbial diversity. Actinobacteria — a group central to gut stability — declined and stayed suppressed throughout the entire follow-up period of more than six months.

Short-term dysbiosis patterns after treatment typically include increases in Enterobacteriaceae and Enterococcus species, along with decreases in beneficial Actinobacteria. Perhaps most concerning, oral-origin bacteria — Streptococcus, Prevotella, Rothia, and others — tend to colonize the recovering stomach. Some of these species have been associated with precancerous gastric changes, which is why active recovery matters more than just waiting it out.
A structured recovery protocol should address three simultaneous goals: repopulating beneficial bacteria, feeding them, and reducing residual inflammation.
Rebuilding the Microbiome
Start probiotic supplementation during the antibiotic course (take probiotics 2-3 hours apart from antibiotics to avoid killing them on arrival) and continue for at least 8-12 weeks post-treatment. Multi-strain formulations containing Lactobacillus and Bifidobacterium species have the best evidence base. Probiotic supplements with documented colony-forming unit (CFU) counts in the billions — not millions — are the research-backed choice.
Feeding Recovered Bacteria
Prebiotics — specifically inulin and butyrate-producing fibers — were recommended in the microbiome recovery literature as key post-eradication interventions. Butyrate is the preferred fuel source for colonocytes (the cells lining your colon) and plays a direct role in maintaining intestinal barrier integrity. Good dietary sources include bone broth, cooked and cooled potatoes (for resistant starch), onions, garlic, leeks, artichokes, and asparagus.
Calming Residual Inflammation
Even after the bacterium is gone, the inflammatory cascade it triggered does not shut off instantly. An anti-inflammatory eating pattern built around omega-3 fatty acids, polyphenol-rich fruits and vegetables, and fermented foods supports the transition from active inflammation to tissue repair. Continuing curcumin supplementation during recovery has mechanistic justification — its NF-κB suppression properties remain beneficial regardless of whether live bacteria are still present.
Recovery timeline: Expect 6-12 months for the gut microbiome to substantially stabilize after eradication therapy. Short-term digestive symptoms (bloating, altered bowel habits) during the first 4-8 weeks are normal collateral effects of the antibiotic course and typically resolve as the microbiome rebuilds.
Myths vs. Facts: What the Science Actually Shows
| Myth | Fact |
|---|---|
| Mastic gum eradicates H. pylori | Despite widespread recommendations online, multiple clinical trials showed mastic gum monotherapy failed to eliminate H. pylori. Lab results looked promising, but patients who took mastic capsules for 14 days remained infected throughout treatment. |
| Raw garlic kills H. pylori | Garlic shows strong activity in lab dishes but translates poorly to the human stomach. A large trial found 87% negative cases with garlic versus 73% for placebo — not statistically significant. Critically, cooking or processing garlic destroys the allicin responsible for its antibacterial effect. |
| You can cure H. pylori with diet alone | Natural compounds achieve 16-56% eradication as standalone treatments. Standard triple therapy achieves around 82%, and sequential therapy reaches 92%. Natural agents shine when combined with antibiotics — not as replacements for them. |
| Everyone with H. pylori needs treatment | Most carriers are asymptomatic. Current clinical thinking increasingly favors personalized benefit-risk assessment rather than universal "test-and-treat" — especially given the microbiome damage eradication therapy itself causes. |
| Once treated, H. pylori never comes back | Reinfection rates depend on environment and hygiene conditions. In developed countries, recurrence within three years is under 10%. In areas with high transmission rates, reinfection occurs more frequently. |
| Probiotics alone can replace antibiotics for H. pylori | Probiotics improve outcomes when added to antibiotic therapy (boosting eradication by up to 21%) but cannot eliminate the infection on their own. Their role is adjunctive and protective — reducing side effects and supporting the gut microbiome during treatment. |
Frequently Asked Questions
How do I know if I have H. pylori without an endoscopy?
The urea breath test and stool antigen test are both non-invasive and highly accurate for detecting active H. pylori infection. The breath test involves swallowing a urea solution and breathing into a collection bag — the lab measures carbon dioxide isotopes produced by H. pylori's urease enzyme. The stool test detects H. pylori antigens directly. Both are preferred over blood tests, which can stay positive long after the infection has cleared.
Can I take natural supplements instead of antibiotics for H. pylori?
The clinical data does not support this approach for active, symptomatic infection. Natural compounds achieve eradication rates of 16-56% when used alone — far below the threshold for reliable treatment. Where natural agents excel is alongside antibiotics: broccoli sprout extract, probiotics, curcumin, and lactoferrin all significantly boost eradication when combined with standard therapy while reducing side effects.
How long does it take to recover from H. pylori treatment?
Symptom relief typically begins within the first two weeks after starting treatment. Full microbiome recovery, however, takes considerably longer — research indicates at least six months for the gut bacterial community to substantially stabilize. Continuing probiotic and prebiotic supplementation during this period supports more complete recovery.
Is H. pylori contagious?
H. pylori spreads person-to-person through saliva, fecal-oral contact, and contaminated water or food. Transmission is most common during childhood, particularly in crowded living conditions. Within families, if one member is diagnosed, testing other family members is often recommended to prevent reinfection cycling.
Should I avoid any foods during H. pylori treatment?
Spicy foods, citrus, coffee, alcohol, and highly processed foods can irritate an already inflamed stomach lining and worsen symptoms during treatment. Focus on easily digestible foods — cooked vegetables, lean proteins, fermented foods like yogurt, and foods that support digestive healing. Broccoli sprouts, turmeric in food form, and ginger tea are reasonable additions based on the evidence reviewed above.
Related Articles
- Leaky Gut Syndrome: Symptoms, Causes, and Evidence-Based Healing — How intestinal permeability connects to chronic inflammation and digestive recovery.
- Probiotics Benefits and Side Effects — A deeper look at probiotic strains, dosing, and what the research actually supports.
- The Gut-Brain Axis: How Your Microbiome Affects Mood and Cognition — Why gut health after H. pylori treatment matters beyond digestion.
- SIBO: Symptoms, Testing, and Treatment — Another bacterial overgrowth condition with overlapping symptoms and shared management strategies.
- Anti-Inflammatory Eating Patterns — Dietary frameworks that support post-infection gut recovery and long-term digestive health.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.