Your body already makes this anti-inflammatory compound
Palmitoylethanolamide -- PEA for short -- is a fatty acid your body produces every time tissue gets damaged. Cells in your skin, gut, brain, and immune system manufacture it on the spot, right in the cell membrane, when they detect stress or inflammation. Nobody invented PEA in a lab. You have been making it since birth.
Chemically, PEA belongs to the N-acyl-ethanolamine (NAE) family -- the same class of lipid messengers that includes anandamide, one of your body's primary endocannabinoids. Scientists first isolated PEA from soybean lecithin and egg yolk in 1957, though it took decades before anyone figured out what it actually does.
When inflammation flares -- from an injury, an infection, even psychological stress -- cells ramp up PEA production to bring things back to baseline. During chronic conditions, your body burns through PEA faster than it can make it. Endogenous levels drop. The supply runs dry.
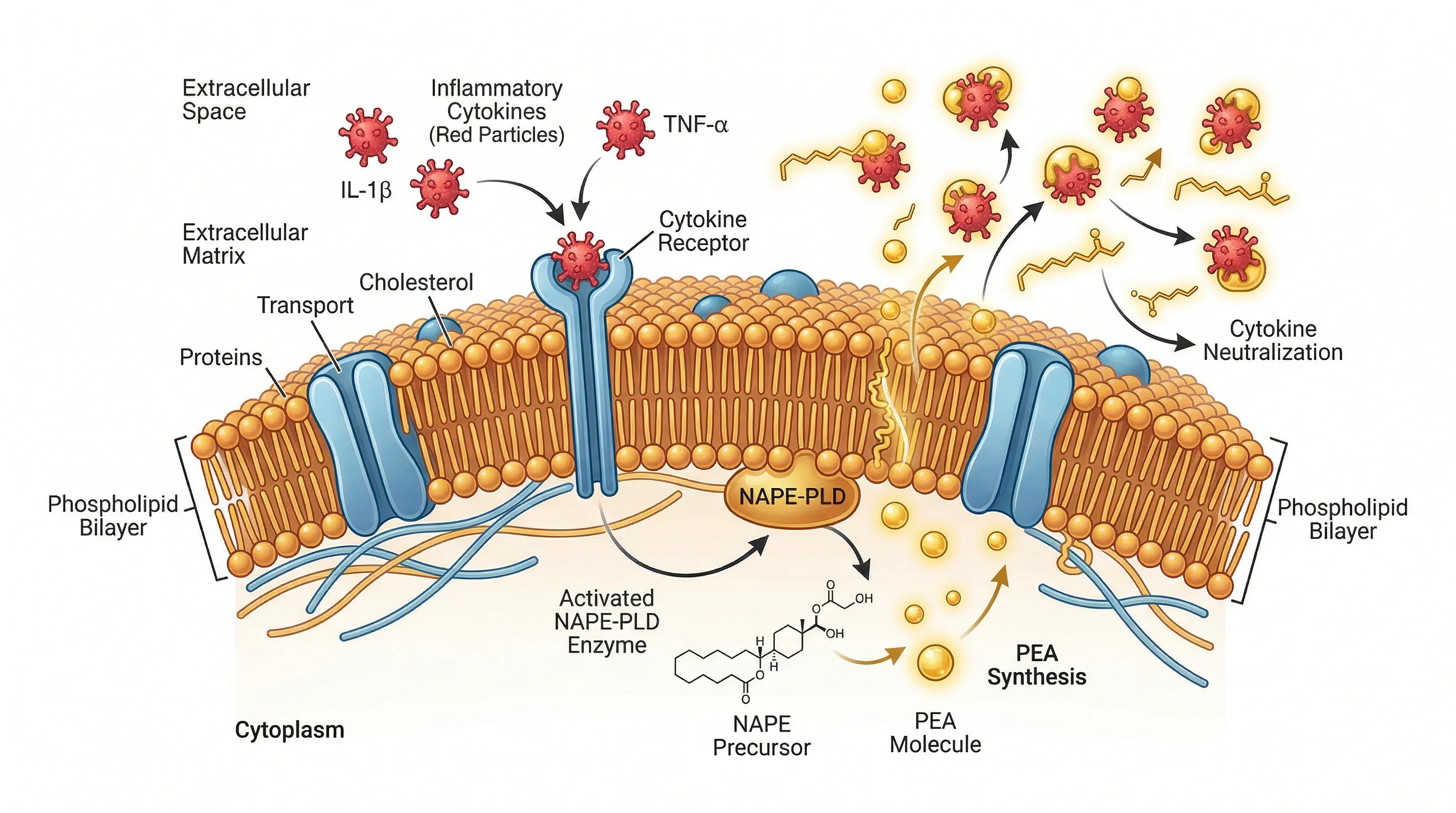
PEA's relationship with the endocannabinoid system is where it gets technical. PEA does not bind directly to cannabinoid receptors CB1 and CB2 the way THC does. It works through a nuclear receptor called PPAR-alpha, which sits inside the cell nucleus and acts as a master switch for inflammatory gene expression. When PEA activates PPAR-alpha, it shuts down NF-kB -- the transcription factor responsible for turning on production of inflammatory cytokines like TNF-alpha, IL-1beta, and IL-6.
PEA also inhibits FAAH, the enzyme that breaks down anandamide. More anandamide sticking around means more activation of CB1 and CB2 receptors. Researchers call this the "entourage effect" -- PEA amplifies what your endocannabinoid system is already doing without directly touching the receptors.
There is a third mechanism. PEA stabilizes mast cells -- the immune cells that release histamine and trigger allergic and inflammatory cascades. Nobel laureate Rita Levi-Montalcini, who discovered nerve growth factor, characterized this mast cell stabilization, naming it ALIA (Autacoid Local Injury Antagonism). PPAR-alpha activation, FAAH inhibition, mast cell stabilization: PEA hits inflammation from three angles simultaneously.
If you are dealing with chronic inflammation, your body's PEA reserves are probably depleted. Supplementing with exogenous PEA restocks a supply your body has been burning through. Closer to replacing a depleted nutrient than introducing a foreign pharmaceutical.
Two meta-analyses, thousands of patients, consistent results
PEA's pain-relieving effects are no longer based on animal studies and case reports. Two meta-analyses -- one from 2023, one from 2025 -- have pooled data from randomized controlled trials, and pain specialists are paying attention.
The 2023 analysis by Dr. Kordula Lang-Illievich and colleagues at the Medical University of Graz examined 11 double-blind randomized controlled trials with 774 patients combined. PEA reduced pain scores by a standardized mean difference of 1.68 on a 0-10 scale (95% CI 1.05 to 2.31, p = 0.00001). For context, a 2-point reduction on a 10-point pain scale is generally considered clinically meaningful. PEA came close to that threshold across diverse pain conditions. No major side effects were attributed to PEA in any of the included trials.
The 2025 meta-analysis went wider -- 18 randomized clinical trials with 1,196 patients drawn from PubMed, Embase, Scopus, and the Cochrane Library. Pain dropped at every measured timepoint: at 6 weeks (SMD -0.9), 8 weeks (SMD -0.98), and 24-26 weeks (SMD -1.16). The longer people took PEA, the more their pain decreased. Quality of life scores also improved (SMD -0.61, 95% CI -0.93 to -0.30).
The breakdown by pain type is where the 2025 data gets interesting. PEA worked across all three categories: nociceptive pain (SMD -0.74), neuropathic pain (SMD -0.97), and nociplastic pain (SMD -0.59). Nociceptive pain comes from tissue damage -- osteoarthritis, for example. Neuropathic pain comes from nerve dysfunction -- diabetic neuropathy, sciatica. Nociplastic pain involves central sensitization without clear tissue or nerve damage -- fibromyalgia falls here. Most pain medications work well for one category and poorly for the others. PEA showing efficacy across all three is unusual.
Bottom line: Across two independent meta-analyses spanning 1,970 patients, PEA consistently reduced pain scores with no serious adverse events reported. Benefits appeared within 4-6 weeks and increased through 24 weeks of use.
The 2025 meta-analysis concluded that PEA is "a promising alternative to chronic opioid analgesics, potentially reducing the risk of opioid abuse and dependency." The researchers are not calling PEA a replacement for opioids. They are positioning it as a candidate to reduce how much people need them.
Both meta-analyses noted moderate to high heterogeneity across studies -- trial designs, dosing protocols, and patient populations varied. Most individual trials were small (50-150 participants), and blinding was not always robust. Several studies used PEA alongside other treatments, making it hard to isolate PEA's independent effect. Larger, more standardized trials are still needed.
The brain connection: neuroinflammation, BDNF, and cognitive function
Pain gets the headlines, but the neuroprotection data may matter more long-term. PEA crosses the blood-brain barrier -- at least in its ultramicronized form -- and influences brain function through several overlapping pathways.
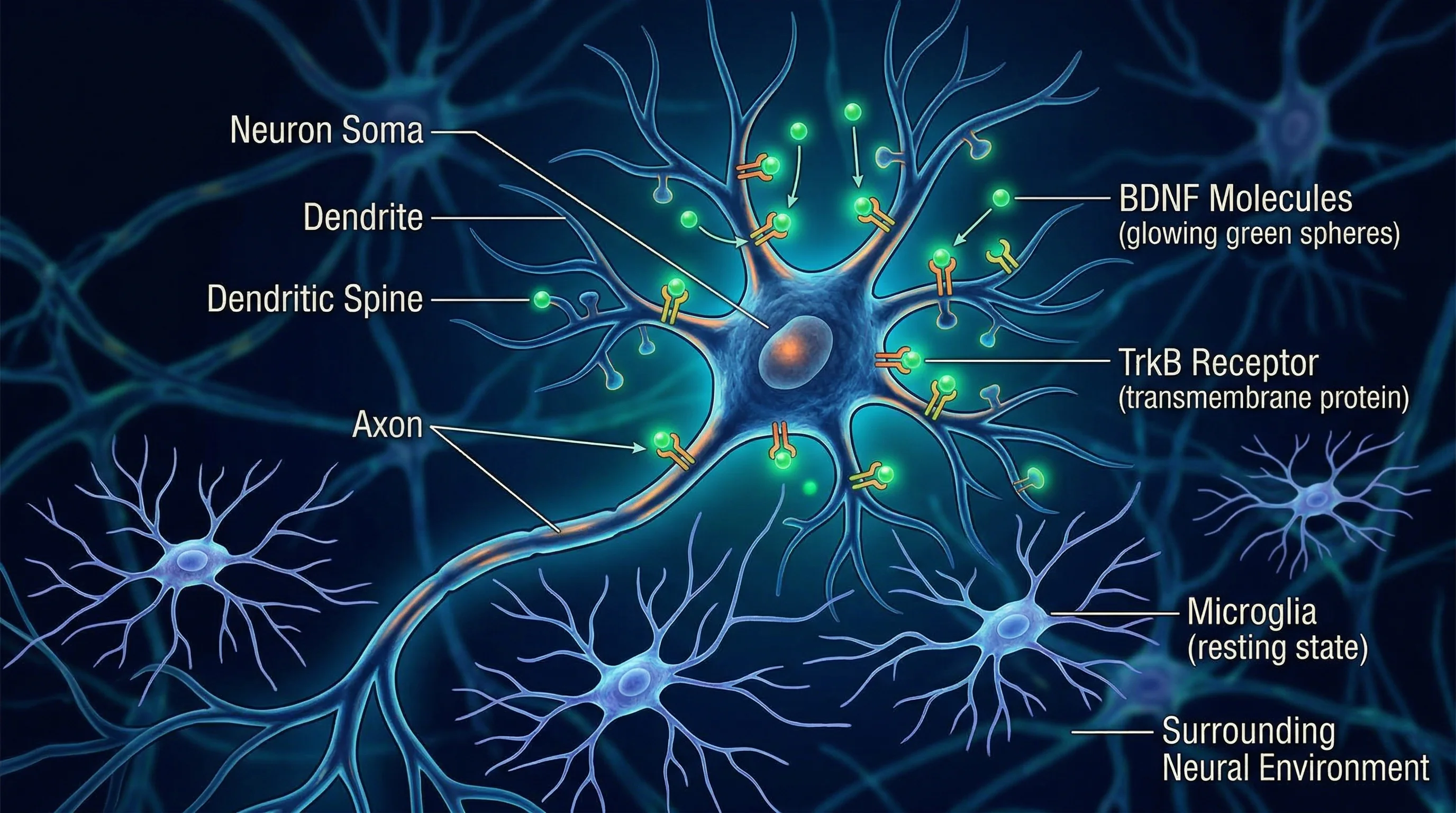
Start with microglia, the brain's resident immune cells. When they detect a threat, they switch from surveillance to attack mode and start pumping out inflammatory cytokines. That is useful during acute infections. It becomes destructive when microglia stay chronically activated, which happens in Alzheimer's, Parkinson's, traumatic brain injury, and prolonged psychological stress. PEA dampens chronic microglial activation through its PPAR-alpha pathway, reducing TNF-alpha and IL-1beta release without suppressing the immune system's ability to respond to actual threats.
BDNF -- brain-derived neurotrophic factor -- is the other piece. BDNF promotes the growth of new neural connections, strengthens existing synapses, and supports neuron survival under stress. Chronic pain, depression, and neurodegeneration are all associated with depleted BDNF levels. Research shows that PEA upregulates BDNF expression, promoting neural repair and reversing the synaptic remodeling that chronic pain causes. Chronic pain rewires your brain to amplify pain signals. PEA appears to help undo some of that rewiring by boosting the molecule your brain needs to build healthier connections.
The cognitive data is preliminary but worth tracking. A systematic review by Colizzi and colleagues (2022) found that PEA supplementation improved Mini-Mental State Examination (MMSE) scores by 3.80 points. The confidence interval was wide (95% CI -0.16 to 7.75), so confirmation in larger trials is needed. But a nearly 4-point improvement on the MMSE is clinically relevant -- roughly the difference between "mild cognitive impairment" and "normal range" for many patients.
The mood data is stronger. A double-blind, placebo-controlled trial found that adding ultramicronized PEA to standard antidepressant treatment produced significantly greater improvements in depressive symptoms compared to placebo. PEA is not being positioned as a standalone antidepressant, but as something that may help existing treatments work better -- which tracks with its ability to reduce neuroinflammation, a recognized driver of treatment-resistant depression.
For anyone dealing with chronic pain and noticing their thinking feels foggy, these overlapping effects matter. NSAIDs do nothing for BDNF. Opioids actively impair cognition. PEA addresses the inflammation, the neural repair, and the mood component at once.
PEA vs CBD: same shelf, different science
PEA and CBD sit next to each other in supplement stores and both get marketed for pain and inflammation. They do target overlapping systems, but the differences in mechanism, evidence, and practical use are worth understanding.

PEA activates PPAR-alpha directly -- confirmed in knockout mouse models where blocking PPAR-alpha eliminates PEA's effects. CBD's mechanism is murkier. It does not bind strongly to CB1 or CB2 receptors. It may work through allosteric modulation of cannabinoid receptors, FAAH inhibition, serotonin receptor agonism, TRPV1 activation, or some combination. Researchers still cannot definitively say which pathway does what. That is not a knock on CBD -- it is an honest assessment of where the science stands.
| Factor | PEA | CBD |
|---|---|---|
| Primary mechanism | PPAR-alpha activation (confirmed in knockout models) | Multiple proposed targets; primary pathway unclear |
| ECS interaction | Indirect -- inhibits FAAH, boosts anandamide | Indirect -- may inhibit FAAH and modulate CB1 allosterically |
| Oral bioavailability | Low (improved with micronization/LipiSperse) | Low (13-19%); bell-shaped dose-response |
| Legal status | Legal worldwide as food supplement | Varies by jurisdiction; regulated differently across US states, EU countries |
| Drug testing | No impact | Risk of triggering positive THC test with full-spectrum products |
| Psychoactive effects | None | None (but legal association with THC creates confusion) |
| FDA-approved uses | None (sold as supplement) | Epidiolex approved for rare pediatric epilepsy |
| Side effects in trials | None reported at standard doses | Elevated liver enzymes, diarrhea, fatigue at high doses |
One pharmacokinetic detail gets overlooked. CBD shows a bell-shaped dose-response curve in several studies -- more is not necessarily better, and there is a narrow window where it works optimally. Go above that window and efficacy can drop. PEA does not appear to have this problem; its dose-response is more linear within the studied range.
PEA is legal everywhere and carries no drug testing risk. CBD's legal status varies between US states, between EU member countries, and between professional athletic organizations. Full-spectrum CBD products can contain enough THC to trigger a positive drug test. For anyone subject to workplace testing, competing in sports, or traveling internationally, this difference alone can be decisive.
Should they be combined? Possibly. A 2021 study by Briskey and colleagues found that PEA combined with hemp oil extract produced synergistic anti-hyperalgesic effects in mouse pain models. A 2019 randomized controlled trial by Couch et al. showed both PEA and CBD independently prevented inflammation-induced gut hyperpermeability in humans. The combination targets different pathways -- PEA through PPAR-alpha, CBD through TRPV1 and serotonin receptors -- which is theoretically synergistic. But no large human trial has directly compared PEA alone, CBD alone, and the combination for pain. That trial needs to happen.
Dosing, safety, and what to look for on the label
PEA's safety profile is unusual for a bioactive compound. Clinical trials report no serious adverse events at standard doses, and this safety record extends back to the 1950s. Earlier clinical programs using PEA under the brand name "Impulsin" treated 1,386 volunteers without major safety signals. Modern trials have continued the pattern. PEA metabolizes into palmitic acid and ethanolamine -- substances your body handles routinely.
WebMD rates PEA as "Possibly Effective" for osteoarthritis and chronic pain, and "Possibly Ineffective" for spinal cord injury pain. Worth remembering -- PEA is not a universal pain solution.
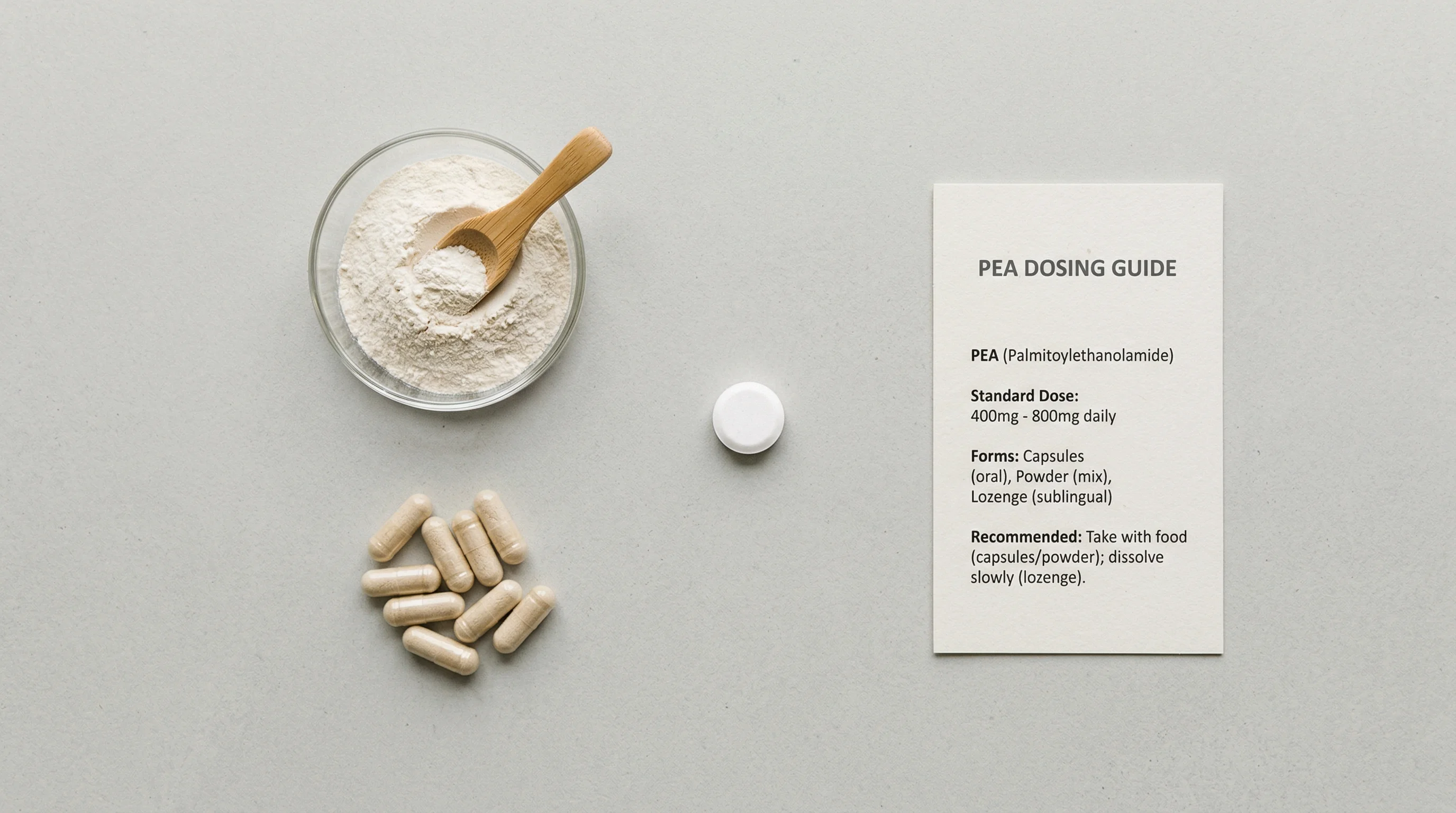
Dosing guidelines from the clinical literature: Start at 300-600mg daily for the first 2-4 weeks. Standard therapeutic dosing is 600-1200mg daily, split into two doses. Some protocols use 1200mg daily as the standard dose. Doses above 1800mg have not shown additional benefit.
Formulation matters a lot. Standard PEA powder has particles larger than 100 micrometers with poor oral bioavailability. Micronized PEA reduces particles to 2-10 micrometers. Ultramicronized PEA (umPEA) gets below 6 micrometers, improving absorption and enabling the compound to cross the blood-brain barrier. A third approach uses LipiSperse technology, which prevents lipophilic particles from clumping and improves water dispersibility.
On the label, look for "micronized PEA," "ultramicronized PEA" (often abbreviated um-PEA), or brand names like Levagen+ (which uses the LipiSperse delivery system). Avoid products that list "palmitoylethanolamide" without specifying particle size or bioavailability technology. You may be paying for PEA that passes through your gut unabsorbed.
PEA reaches peak plasma concentration within about 2 hours of ingestion, then returns to baseline relatively quickly. Twice-daily dosing is standard for that reason -- it maintains more consistent levels. The trials that showed the strongest pain reduction used daily protocols for a minimum of 4-8 weeks, so do not expect overnight results.
Who gets the most out of PEA (and who should hold off)
Based on the current evidence, PEA has the strongest case for people with chronic inflammatory or neuropathic pain who want to reduce their reliance on NSAIDs or opioids. The meta-analysis data is most robust for osteoarthritis, low back pain, diabetic neuropathy, and fibromyalgia. If you are managing one of these conditions and conventional medications are either inadequate or causing side effects, PEA is a reasonable conversation to have with your doctor.

The neuroprotection angle makes PEA potentially interesting for a different group: people concerned about cognitive decline. The BDNF upregulation and microglial modulation data, while mostly preclinical, suggest PEA could support brain health alongside its pain-relieving properties. This needs more human data before making strong recommendations, but the mechanism is plausible with early supporting evidence.
Who should hold off: anyone expecting PEA to replace strong acute pain medication. The effect sizes in the meta-analyses represent moderate pain reduction over weeks to months. PEA builds slowly; it is not an on-demand painkiller. It is also not established for spinal cord injury pain. And while PEA's safety profile is excellent, pregnant or breastfeeding women should note that safety data in these populations, while described as favorable, comes from limited studies.
Active research continues. A US-based randomized controlled trial (NCT06273462) is currently evaluating PEA for chronic inflammatory pain conditions including rheumatoid arthritis, fibromyalgia, and neuropathic pain. This is one of the first major US-based PEA trials -- most prior research has come from European institutions. Results from this trial could shift how American clinicians view PEA.
PEA's evidence base is ahead of its public awareness. Two meta-analyses with nearly 2,000 combined patients, no reported serious side effects, efficacy across three pain categories, and emerging neuroprotective data. The trials need to be larger and the research is not finished. But the evidence points in one direction, and most supplements never accumulate anything close to this level of clinical support.
Frequently asked questions
How quickly does PEA start working for pain?
Most clinical trials show significant pain reduction at the 4-6 week mark, with continued improvement through 24 weeks. Some people notice changes within the first two weeks, but PEA is not designed for immediate relief. It works by gradually reducing underlying inflammation and neuroinflammation rather than masking pain signals the way opioids or NSAIDs do.
Can I take PEA with other medications?
PEA has no reported drug-drug interactions in the clinical literature, and its metabolic byproducts (palmitic acid and ethanolamine) are substances your body routinely processes. Several clinical trials specifically studied PEA as an add-on to existing pain medications and antidepressants, finding enhanced benefits without additional side effects. Inform your healthcare provider about any supplement you are taking, particularly if you are on multiple medications.
Is PEA the same as CBD?
No. PEA and CBD are structurally different molecules that work through distinct primary mechanisms. PEA activates PPAR-alpha receptors, while CBD's mechanism involves multiple targets including TRPV1 channels and serotonin receptors. PEA is endogenous (your body makes it), legal everywhere, and does not carry drug testing risks. CBD is plant-derived, faces varying regulation, and can trigger positive drug tests in full-spectrum formulations.
What formulation of PEA should I look for?
Look for micronized or ultramicronized PEA (um-PEA), or products using LipiSperse technology (branded as Levagen+). Standard non-micronized PEA has poor absorption due to its lipophilic nature and large particle size. Ultramicronized formulations reduce particles below 6 micrometers, improving bioavailability and allowing the compound to reach the brain.
Does PEA help with anxiety or depression?
There is early evidence. A double-blind placebo-controlled trial found that ultramicronized PEA added to standard antidepressant treatment significantly improved depressive symptoms compared to placebo. PEA's effects on neuroinflammation, BDNF, and the endocannabinoid system are all relevant to mood regulation. PEA is not established as a standalone treatment for anxiety or depression and should not replace prescribed psychiatric medications without medical guidance.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.











