A Swedish neuroscience startup recently branded their depression treatment "electric medicine," and the name stuck for good reason. On December 8, 2025, the U.S. Food and Drug Administration approved the first at-home brain stimulation device specifically for treating depression — a headband that delivers gentle electrical currents to your prefrontal cortex while you sit on your couch. Meanwhile, at Stanford, patients with treatment-resistant depression were walking out of the clinic after just five days with their symptoms in remission. At UCLA, researchers were proving that personalizing exactly where you zap the brain makes the difference between mediocre and meaningful results.
These aren't fringe experiments. They represent a convergence of neuroscience, engineering, and clinical evidence that is rewriting what non-invasive brain treatment looks like. With 47.8 million American adults living with major depression and 169 million people in federally designated mental health shortage areas, the appeal of treatments that don't require a weekly therapist visit or months of medication adjustment is obvious. But the field is also littered with overpromise. Understanding what the evidence actually supports — and where the gaps remain — matters more than the hype.
What Happens When You Send Electricity Through a Skull
Two technologies dominate the non-invasive brain stimulation field, and they work in fundamentally different ways. Understanding the distinction matters because it determines everything from who administers the treatment to whether you could eventually do it at home.
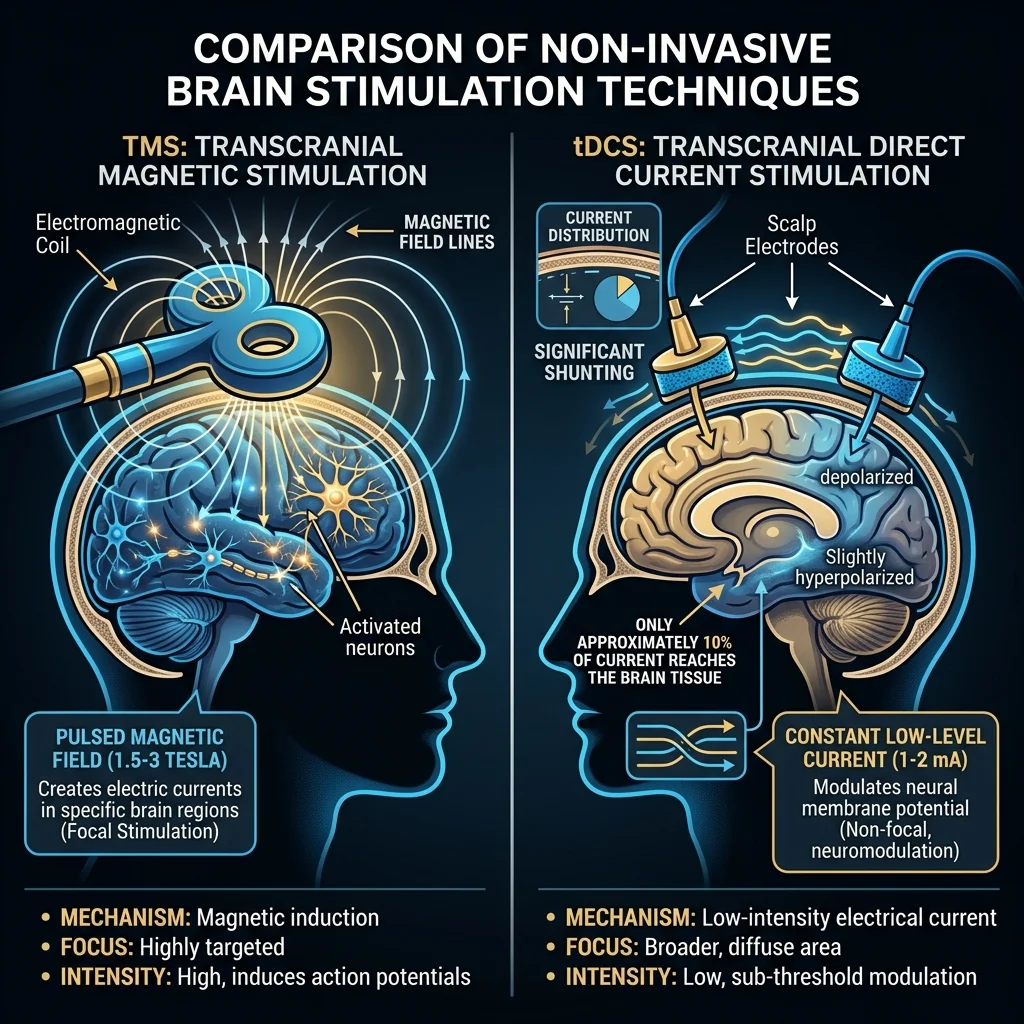
Transcranial magnetic stimulation (TMS) works like a focused electromagnetic punch. A coil placed against your scalp generates a rapidly changing magnetic field — between 1.5 and 3 Tesla, comparable to an MRI machine. Think of it as throwing a rock into a pond: the magnetic field passes through bone and tissue without resistance, then induces an electrical current in the neurons underneath. That induced current is strong enough to make neurons fire. When delivered repeatedly (repetitive TMS, or rTMS), the treatment can strengthen or weaken specific neural connections through mechanisms that resemble long-term potentiation and long-term depression — the same synaptic plasticity processes your brain uses to form memories.
The direction depends on frequency. High-frequency stimulation (10 Hz and above) and a newer variant called intermittent theta-burst stimulation (iTBS) tend to increase neural excitability. Low-frequency protocols (1 Hz and below) suppress it. A standard iTBS session delivers 1,200 pulses in under 8 minutes, compared to 37.5 minutes for conventional high-frequency rTMS — a time savings that becomes relevant when you're asking patients to sit through treatment five days a week.
Transcranial direct current stimulation (tDCS) is subtler. Instead of forcing neurons to fire, it nudges them. Electrodes on the scalp deliver a weak direct current — typically 1 to 2 milliamps — that shifts the resting membrane potential of neurons underneath. Picture turning the volume knob on a radio slightly up or down: neurons under the positive electrode (anode) become slightly more likely to fire, while those under the negative electrode (cathode) become slightly less likely. The current doesn't trigger action potentials directly. It modulates existing neural activity by adjusting the threshold at which neurons decide to fire.
There's a catch, though. Standard tDCS electrodes are large — 25 to 35 square centimeters — and the skull absorbs roughly 90% of the current before it reaches brain tissue. The stimulation is diffuse, often hitting regions beyond the intended target. This is why a newer approach called high-definition tDCS (HD-tDCS) matters: it uses an array of five smaller electrodes (2 x 2 cm each) arranged in a ring pattern, concentrating the current on a specific brain region rather than spreading it across a wide area.
| Feature | TMS | tDCS |
|---|---|---|
| How it works | Electromagnetic induction generates current in neurons | Direct current shifts neural firing thresholds |
| Strength | 1.5-3 Tesla magnetic field | 1-2 milliamps current |
| Brain penetration | Magnetic field passes through skull without resistance | ~10% of current reaches brain tissue |
| Triggers action potentials? | Yes — forces neurons to fire | No — modulates existing firing patterns |
| Session duration | 3-40 minutes (iTBS fastest) | 20-30 minutes typical |
| Setting | Clinic only (specialized equipment) | Clinic or home (portable) |
| Focal precision | High (figure-of-eight coil) | Low standard; high with HD-tDCS array |
Both technologies target the dorsolateral prefrontal cortex (DLPFC) for depression — a brain region involved in emotion regulation that tends to be underactive on the left side in people with depression. But TMS requires specialized equipment, a trained technician, and clinic visits. tDCS is portable, cheaper, and simple enough that patients can potentially use it at home with remote supervision. That practical difference shapes which patients get access to which treatment.
From Six Weeks to Five Days: The Stanford Protocol That Rewrote the Timeline
Standard TMS for depression takes six weeks. You show up at a clinic five days a week, sit through a 20-to-40-minute session, go home, and repeat. It works for many patients, but the time commitment is a barrier — especially for people who are already struggling to function.
A team at Stanford, led by Dr. Nolan Williams, asked a straightforward question: what if you compressed the entire treatment course into five days? The answer became the Stanford Accelerated Intelligent Neuromodulation Therapy (SAINT) protocol, and its results shook the field.
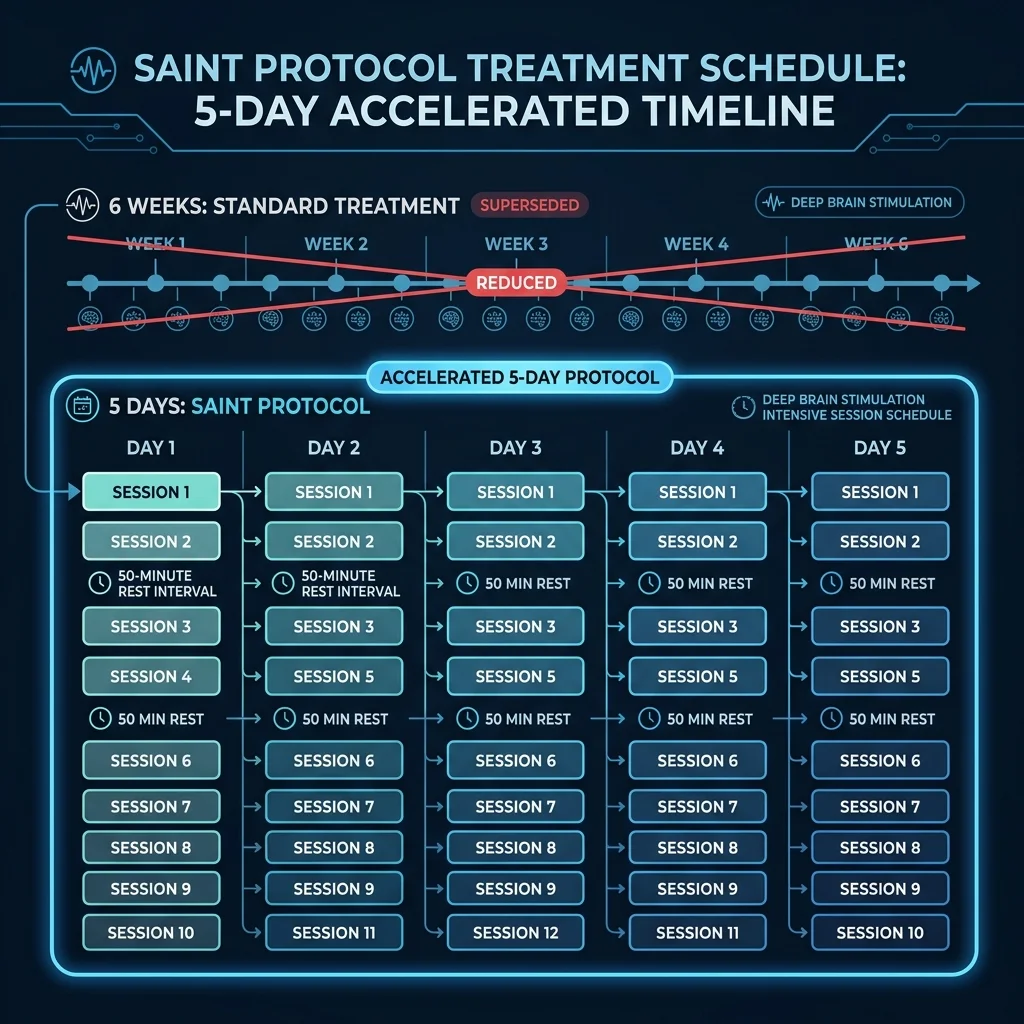
The protocol is aggressive by design. Patients receive 10 iTBS sessions per day for five consecutive days — 50 sessions total, with 1,800 pulses per session and 50-minute rest intervals between sessions. That's 90,000 total pulses delivered in a week, compared to roughly 18,000 over a standard six-week course. But the volume alone isn't what makes SAINT different.
The protocol's second innovation is precision targeting. Rather than placing the TMS coil using skull landmarks (the standard method), SAINT uses resting-state functional connectivity MRI to find the exact spot on each patient's left DLPFC that is most strongly anticorrelated with the subgenual anterior cingulate cortex — a deep brain structure implicated in depressive rumination. The target varies from person to person, and hitting the right spot appears to matter considerably.
In the initial open-label study of 22 patients with treatment-resistant depression, 19 of 21 who completed treatment (90.5%) met remission criteria on the Montgomery-Asberg Depression Rating Scale. The intent-to-treat remission rate was 86.4%. Neuropsychological testing showed no negative cognitive side effects.
Those numbers warranted skepticism — open-label studies carry placebo effects — so Dr. Williams's team ran a double-blind, sham-controlled trial. Twenty-nine participants with treatment-resistant depression received either active or sham SNT (the protocol's updated name). Four weeks after treatment, the active group showed a 52.5% reduction in MADRS scores compared to 11.1% in the sham group. The difference was statistically significant and clinically meaningful.
A systematic review of 23 studies comparing accelerated TMS protocols to standard approaches confirmed that the key variables driving efficacy are total pulse dosage, the duration of the intersession interval (giving the brain time to consolidate changes between sessions), and whether the targeting uses MRI-guided neuronavigation versus cruder positioning methods.
The commercial translation is already happening. In September 2025, BrainsWay received FDA clearance for an accelerated deep TMS protocol that compresses the initial treatment phase from four weeks down to six days. It's not identical to SAINT — BrainsWay uses a different coil design and targeting approach — but it signals that regulators are comfortable with compressed treatment timelines when the data supports them.
For patients who have spent months cycling through antidepressants without relief, the idea of a one-week treatment is compelling. The practical question is access: SAINT requires an fMRI scan for targeting and intensive clinical supervision for the 10 daily sessions. That infrastructure exists at academic medical centers but not at the average psychiatric practice.
The UCLA Experiment: Making Brain Stimulation More Precise (and Portable)
While Stanford was compressing treatment timelines, a team at UCLA was asking a different question: does it matter exactly where you put the electrodes? The answer, published in JAMA Network Open in September 2025, appears to be yes — and the implications extend beyond academic interest.
Dr. Mayank Jog and Dr. Katherine Narr at the UCLA Brain Mapping Center conducted a randomized, double-blind, sham-controlled trial of personalized high-definition tDCS in 71 participants with moderate to severe depression. Each participant received a structural MRI, and the researchers used frameless stereotaxic neuronavigation to position the HD-tDCS electrodes precisely over a specific coordinate on the left DLPFC. Treatment consisted of 20 minutes of 2-milliamp stimulation daily for 12 consecutive working days.
The results showed a moderate effect size (Cohen d = -0.50) favoring active over sham treatment, with remission rates of 39.5% in the active group versus 13.3% in sham (P = .02). The percentage improvement in depression scores was 44.6% for active treatment versus 31.3% for sham. What caught the research community's attention was the speed: significant mood improvements appeared by day six of treatment — the midtreatment assessment.
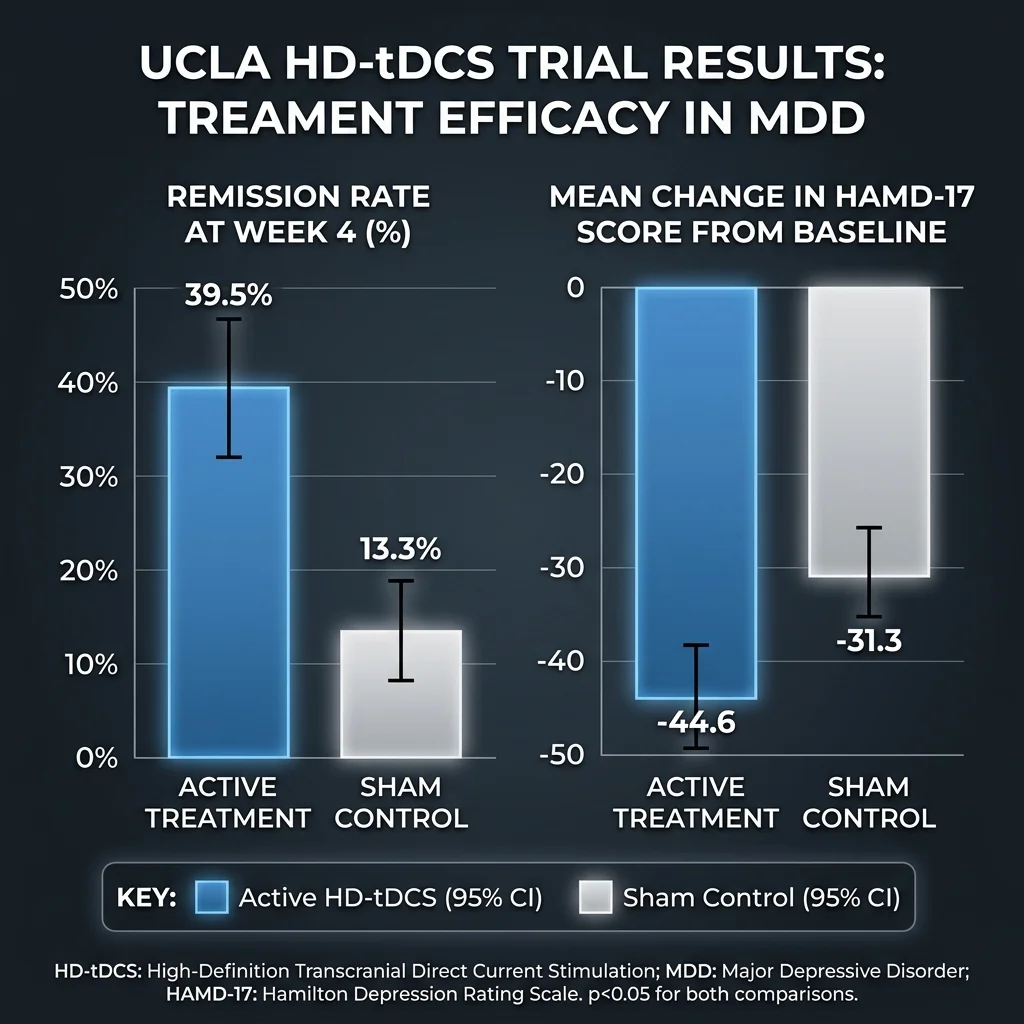
That matters because previous large conventional tDCS trials produced either no effects or smaller effect sizes that took substantially longer to appear. The ELECT-TDCS trial showed a Cohen d below 0.43 at week eight. The EMPOWER trial showed below 0.37 at week four. Both of those trials used higher current intensities, longer sessions, and more total sessions than the UCLA protocol — but with conventional large-pad electrodes rather than the focused HD array.
"By honing in on the precise brain regions affected by depression, we've shown that stimulation can significantly improve mood and daily functioning," Dr. Jog said in a UCLA Health news release. Dr. Narr added that coupled with tDCS's potential for at-home use, the approach "may offer a new treatment option for those seeking alternatives to medication or talk therapy."
Exploratory analysis revealed something else: the active treatment group showed significant improvement in anxiety symptoms as well (Cohen d = -0.48), with both psychic and somatic anxiety components improving. That's preliminary — the study wasn't designed to test anxiety specifically — but it opens a new research direction.
Meanwhile, the case for home-based tDCS keeps building. A 2026 study of home-based tDCS for bipolar depression gave 44 participants 21 remotely supervised sessions over six weeks using the standard F3/F4 electrode montage. Quality of life improved significantly (p < 0.001) and remained elevated at five-month follow-up. The study was open-label, so the results need replication with sham controls, but it demonstrates that remote supervision of home tDCS is feasible and that patients adhere to the protocol.
Conventional tDCS with its large, imprecise electrodes is giving way to personalized approaches guided by neuroimaging. Whether HD-tDCS can replicate UCLA's results at scale, and whether insurance will cover MRI-guided electrode placement, are the questions that will determine if this stays an academic curiosity or becomes standard care.
The Regulatory Landscape: Who's Cleared, for What, and What It Costs
The FDA has cleared brain stimulation devices for more conditions than most people realize, and the pace of approvals accelerated significantly in 2024-2025. Here's where things stand.
| Technology | Device Examples | FDA-Cleared Conditions | Setting | Approximate Cost |
|---|---|---|---|---|
| Repetitive TMS | NeuroStar, BrainsWay, MagVenture, Apollo, Horizon 3.0 | Major depression, OCD, migraines, smoking cessation | Clinic-based | $6,000-$12,000 per treatment course |
| Home tDCS | Flow Neuroscience FL-100 | Moderate to severe major depression (not treatment-refractory) | Home with prescription | $500-$800 target price |
| Cranial electrotherapy (CES) | Alpha-Stim | Anxiety, insomnia, pain | Home | $600-$1,200 |
The first TMS device received FDA clearance for major depression in 2008. A decade later, in 2018, the FDA approved deep TMS for OCD — the first brain stimulation approval for a condition other than depression. Since then, additional clearances have come for migraines and smoking cessation.
The adolescent expansion is recent. NeuroStar earned clearance for adolescent major depression in 2024, and in November 2025, BrainsWay received clearance to extend deep TMS to patients aged 15-21. BrainsWay submitted data from over 1,000 adolescents treated at U.S. TMS centers, showing an average 12-point improvement on the PHQ-9 depression scale after 36 sessions, with meaningful reductions in co-occurring anxiety.
The Flow Neuroscience approval in December 2025 was the bigger regulatory shift. It was the first premarket approval (PMA) for a home-use non-invasive brain stimulation device indicated for depression. The pivotal trial supporting the approval showed 58% remission after 10 weeks compared to 29% with a placebo device. Among Flow's 55,000 European users, 77% reported symptom improvement within three weeks. The company plans a U.S. launch in the second quarter of 2026.
On the horizon, the XCSITE platform developed by Dr. Flavio Frohlich at UNC received FDA Breakthrough Device designation. Unlike standard tDCS, XCSITE reads the patient's brain waves via EEG and delivers personalized electrical stimulation calibrated to their individual neural activity patterns. In a 2024 clinical trial, 80% of participants were declared free of clinical depression symptoms two weeks after treatment.
The cost gap between clinic-based TMS and home tDCS is enormous. A full course of TMS runs $6,000-$12,000, and electroconvulsive therapy (ECT) can reach $25,000 for a full treatment course at $300-$2,500 per session. Flow's target price of $500-$800 for the home device makes brain stimulation accessible to a different economic bracket entirely — but only if insurance covers it, which currently no plan does for home tDCS.
Which Conditions Actually Respond: Ranking the Evidence
Not all brain stimulation evidence is equal. The field has a credibility gradient, and knowing where different conditions fall on it helps separate warranted optimism from premature enthusiasm.
| Condition | Modality | Evidence Level | Effect Size | FDA Status |
|---|---|---|---|---|
| Major Depression (treatment-resistant) | rTMS/iTBS | Strong — multiple large RCTs | Large (remission 52-90% in accelerated protocols) | FDA cleared since 2008 |
| Major Depression (moderate-severe) | tDCS | Moderate — growing RCT evidence | Moderate (Cohen d ~0.50 for HD-tDCS) | FDA approved for home use Dec 2025 |
| OCD | Deep TMS / tDCS | Moderate — 16 RCTs for tES, SMD 0.61 | Moderate | Deep TMS FDA cleared 2018 |
| PTSD | rTMS | Moderate — 11 RCTs, Hedges' g = -0.975 | Large | Not FDA cleared |
| Anxiety disorders | tDCS / CES | Preliminary — exploratory findings | Cohen d ~0.48 (HD-tDCS exploratory) | Alpha-Stim CES cleared for anxiety |
| Smoking cessation | Deep TMS | Sufficient for clearance | Not quantified here | FDA cleared |
| Migraines | TMS | Sufficient for clearance | Not quantified here | FDA cleared |
Depression has the strongest foundation. For treatment-resistant depression, rTMS is considered as effective as antidepressant medications with no cognitive impairment — a meaningful advantage over electroconvulsive therapy, which works but carries memory-related side effects. The accelerated SAINT protocol produced 52.5% MADRS reduction in its double-blind trial, and multiple systematic reviews support the approach. For non-treatment-resistant depression, tDCS evidence is building: the UCLA HD-tDCS trial showed moderate but clinically relevant effects, and the Flow device's 58% remission rate earned FDA approval.
PTSD shows surprisingly strong numbers, though the evidence base is thinner. A meta-analysis of 11 RCTs found a large effect size (Hedges' g = -0.975) for rTMS targeting the right DLPFC in PTSD patients. Both excitatory and inhibitory protocols reduced symptoms, and the effects held up two to four weeks after treatment ended. Given that 7-9% of people develop PTSD in their lifetime and over half of PTSD patients also have depression, there's a strong case for larger trials — but no FDA clearance exists yet for this indication.
OCD evidence is moderate and growing. The 2026 Nature Mental Health meta-analysis found a moderate effect size (SMD 0.61) for transcranial electrical stimulation across 16 RCTs. Twice-daily treatment sessions produced larger effects than once-daily. Less than 40% of OCD patients respond fully to standard first-line treatments (CBT plus serotonin reuptake inhibitors), so an alternative with moderate efficacy fills a genuine gap.
Anxiety is the most preliminary. The UCLA HD-tDCS trial found exploratory anxiety improvements (Cohen d = -0.48), and the BrainsWay adolescent data showed anxiety reductions alongside depression improvements. Alpha-Stim has FDA clearance for anxiety via cranial electrotherapy stimulation. But dedicated anxiety trials using TMS or tDCS as the primary intervention are still sparse. The UCLA researchers were transparent about this: their study wasn't designed to test anxiety, and the findings need dedicated follow-up.
The conditions with FDA clearance (depression, OCD, migraines, smoking cessation) have the most rigorous evidence behind them, which is how they earned clearance. Conditions with large effect sizes but no clearance (PTSD) indicate scientific promise that hasn't yet crossed the regulatory bar — usually because the number of trials or participants is still too small for regulatory standards.
The Other Side: What Can Go Wrong and What We Don't Know
Brain stimulation advocates sometimes understate the limitations, and skeptics sometimes overstate the risks. The actual picture requires holding both the promising data and the caveats in mind at the same time.
Side effects are genuinely mild for most patients. TMS can cause scalp discomfort, headaches, and in rare cases seizures (estimated at less than 1 in 10,000 sessions). The UCLA HD-tDCS trial reported mild skin redness and occasional burning sensation at electrode sites, with no serious adverse events. The SAINT protocol found no negative cognitive effects on neuropsychological testing. Compared to antidepressants — which commonly cause sexual dysfunction, weight gain, and sleep disturbance — or ECT, which causes cognitive impairment that can be persistent, brain stimulation's safety profile is a genuine strength.
Durability is the biggest unknown. The SNT double-blind trial measured outcomes at four weeks post-treatment. The UCLA trial showed that effects began fading at two and four weeks without maintenance sessions. The authors noted this directly: the study protocol did not include maintenance treatment sessions, which may have contributed to nonsignificant follow-up results. Nobody has yet proven that a course of brain stimulation produces lasting remission without some form of ongoing treatment — a gap that matters enormously for real-world patients.
Sample sizes remain small. The SAINT double-blind trial had 29 participants. UCLA's HD-tDCS trial enrolled 71 but fell short of the planned 100 due to COVID-19 enrollment challenges. These numbers are sufficient for initial proof-of-concept but not for the kind of large-scale replication that would eliminate doubt. The systematic review of accelerated TMS flagged this explicitly: small sample sizes, few independent replications, and variability in how treatment resistance is defined all "hamper definitive conclusions."
Cost and access create a two-tier system. A full TMS treatment course costs $6,000-$12,000. Medicare and most commercial insurance now cover standard TMS for treatment-resistant depression, but accelerated protocols and home tDCS are too new for established reimbursement pathways. The Flow device at $500-$800 is cheaper than TMS, but without insurance coverage, it's still out of reach for many of the 169 million Americans in mental health shortage areas who could benefit most. Brain stimulation risks becoming another treatment that works well for people who can already access good care.
The personalization problem. Both SAINT and UCLA's HD-tDCS rely on MRI-guided targeting to achieve their results. Standard TMS uses cruder targeting based on skull measurements, and the results are correspondingly less impressive. The UCLA team was transparent that their study "was not designed to empirically validate personalization" — they used computational models that support the approach, but head-to-head trials comparing personalized versus standard targeting haven't been done yet. If the benefit of brain stimulation turns out to depend heavily on precision targeting, scaling it will require MRI access that many clinics lack.
Home-use devices introduce new concerns. The Flow device is prescription-only and includes remote monitoring, but it represents a category of technology where unsupervised use is possible. Consumer-grade tDCS devices (not FDA-cleared) have been available online for years, and the DIY brain stimulation community has a track record of experimenting with parameters that haven't been tested in clinical settings. More FDA-cleared home devices will help standardize what people use, but they also normalize a technology that requires medical guidance to use safely.
Frequently Asked Questions
Is brain stimulation a replacement for antidepressants or therapy?
For most patients, not yet. Current evidence supports brain stimulation as an additional option, not a wholesale replacement. The Flow tDCS device is approved as both monotherapy and adjunctive treatment, meaning it can be used alone or alongside medication. TMS is typically used after antidepressants have failed. The strongest research results come from patients who continued their existing medications during brain stimulation treatment, so the evidence base for standalone use is thinner than for combined approaches.
How do I know if I'm a candidate for TMS or tDCS?
For TMS, the standard indication is major depression that hasn't responded to at least one adequate antidepressant trial. FDA-cleared TMS is also available for OCD, migraines, and smoking cessation. The Flow tDCS device is indicated for moderate to severe major depression in patients who are not considered treatment-refractory to medication — a different population than TMS targets. People with implanted metallic devices, epilepsy, or certain neurological conditions are typically excluded from brain stimulation. A psychiatrist familiar with neuromodulation can assess candidacy.
Does insurance cover brain stimulation?
Medicare and most commercial insurers cover standard TMS for treatment-resistant depression, though prior authorization is usually required. Coverage for accelerated TMS protocols is still being established. Home tDCS devices like Flow have no established insurance coverage yet — the device is too new. Patients should expect to pay $6,000-$12,000 out-of-pocket for TMS if insurance denies the claim, or $500-$800 for the Flow device.
Are the effects permanent?
Current evidence does not support permanent effects from any brain stimulation protocol. The SAINT accelerated TMS protocol showed significant benefits at four weeks post-treatment, but long-term follow-up data is limited. Most clinicians expect that maintenance sessions — periodic booster treatments — will be needed to sustain benefits, similar to how some patients need ongoing medication. Research into optimal maintenance schedules is underway but not yet conclusive.
Can I buy a tDCS device and use it at home right now?
Consumer tDCS devices are available for purchase online, but these are not FDA-cleared for treating any medical condition. The Flow FL-100 is the only FDA-approved home tDCS device for depression, and it requires a prescription. Using non-cleared devices to self-treat depression carries risks including incorrect electrode placement, inappropriate stimulation parameters, and no medical oversight. The clinical trials that produced positive results used specific protocols with trained supervision — replicating those conditions with consumer hardware is not straightforward.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.