What makes retatrutide different from other weight loss drugs
Every major weight loss medication on the market right now works by pulling one or two hormonal levers. Semaglutide (sold as Ozempic and Wegovy) targets one receptor -- GLP-1. Tirzepatide (Mounjaro and Zepbound) hits both GLP-1 and GIP receptors. Retatrutide adds a third.
Developed by Eli Lilly, retatrutide is a triple hormone receptor agonist that simultaneously activates receptors for three gut hormones: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon. No other drug in clinical development does all three at once in a single molecule.
Structurally, retatrutide is a 39-amino-acid synthetic peptide built on a GIP backbone, conjugated to a C20 fatty diacid moiety that lets it bind to albumin in the bloodstream. That albumin binding extends its half-life to roughly six days -- the peptide hitches a ride on a much larger protein, staying in circulation long enough for once-weekly dosing.
The receptor potency profile is deliberate and uneven. Retatrutide hits the GIP receptor at an EC50 of 0.0643 nM, about 8.9 times more potent than natural human GIP. At the GLP-1 receptor (EC50: 0.775 nM), it runs at roughly 40% of the natural hormone's potency. At the glucagon receptor (EC50: 5.79 nM), about 30%. Lilly's engineers calibrated the molecule so that glucagon's blood-sugar-raising effects would be held in check by the insulin-stimulating actions of GIP and GLP-1 agonism.

That third receptor -- glucagon -- is what separates retatrutide from everything else in the pipeline. Glucagon has historically been the hormone endocrinologists associated with raising blood sugar (it's the rescue injection for severe hypoglycemia). But it also ramps up energy expenditure, drives fatty acid oxidation in the liver, and reduces appetite through mechanisms distinct from GLP-1. Lilly's bet: that adding controlled glucagon agonism on top of GIP and GLP-1 would produce weight loss beyond what dual agonists achieve, without the blood sugar spikes you'd expect from a glucagon-boosting drug.
The Phase 3 data suggests that bet paid off.
Phase 3 trial results: TRIUMPH-4 data breakdown
On December 11, 2025, Eli Lilly released topline results from TRIUMPH-4, the first Phase 3 trial of retatrutide to report data.
Participants on the 12mg dose lost an average of 28.7% of their body weight over 68 weeks -- that translates to 71.2 pounds shed from an average starting weight of 248.5 pounds. The 9mg group lost 26.4%, or about 64.2 pounds. Placebo participants lost 2.1%.
For context: bariatric surgery, the current gold standard for treating severe obesity, typically produces 25-30% weight loss in the first postoperative year. Retatrutide is hitting surgical-level weight loss with a weekly injection.
BMO Capital Markets had set a bull-case prediction of 25% weight loss and a 60% reduction in osteoarthritis pain scores. Retatrutide exceeded both numbers. The firm called it "a true TRIUMPH."
TRIUMPH-4 enrolled 445 adults with obesity or overweight and knee osteoarthritis, randomized 1:1:1 across retatrutide 9mg, 12mg, and placebo. Eighty-four percent of participants had a baseline BMI of 35 or higher. The trial had two co-primary endpoints: weight loss and pain reduction. Both were met at both doses.
Pain dropped by 75.8% on the WOMAC pain subscale with 9mg and 74.3% with 12mg, compared to 40.3% with placebo. About 1 in 8 patients on retatrutide were completely free of knee pain by trial end -- 14.1% on 9mg and 12.0% on 12mg, versus 4.2% on placebo.
Cardiovascular risk markers also improved. At the highest dose, retatrutide lowered systolic blood pressure by 14.0 mmHg and reduced non-HDL cholesterol, triglycerides, and high-sensitivity C-reactive protein.
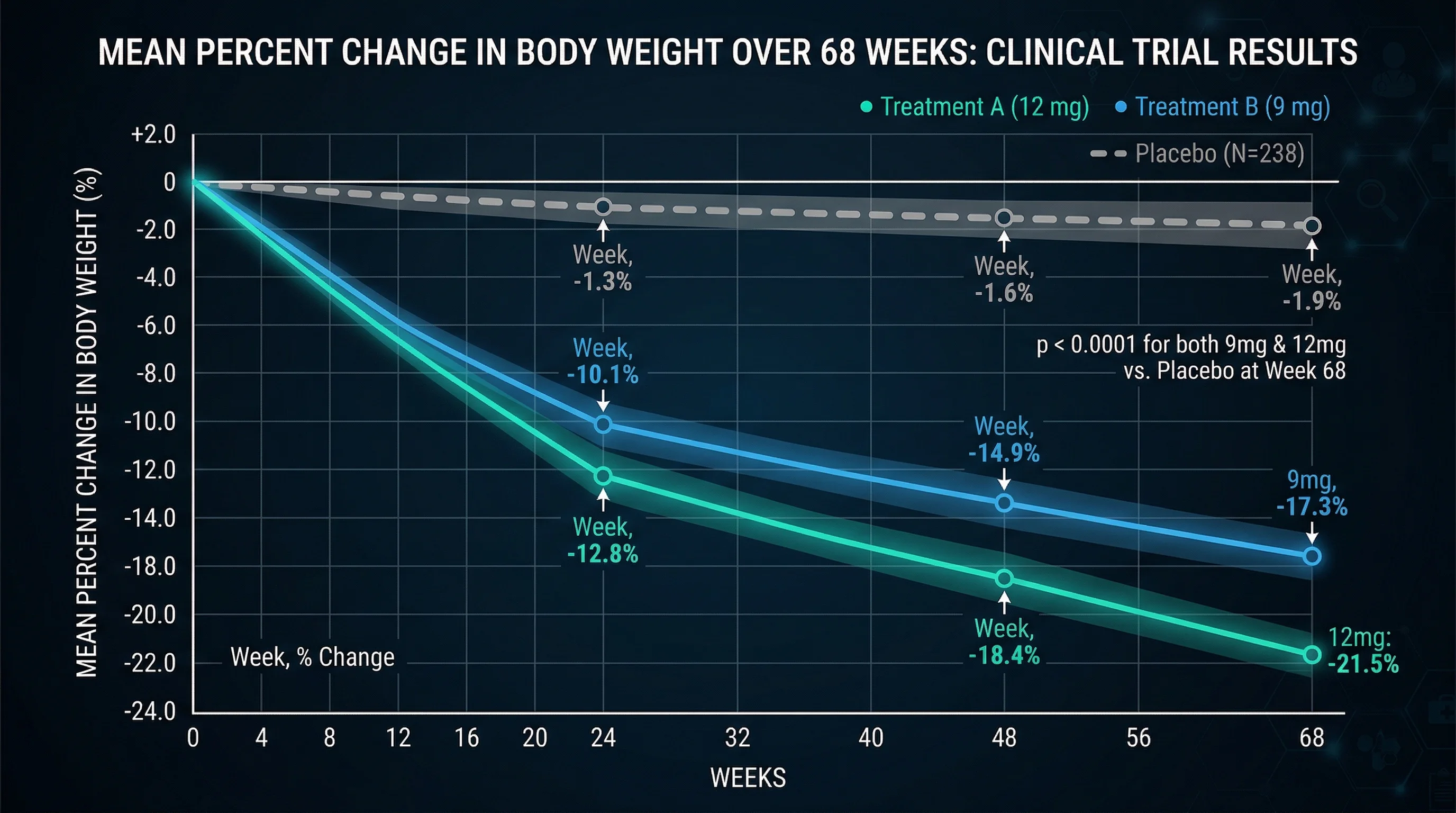
These numbers built on earlier data. The Phase 2 trial, published in the New England Journal of Medicine by Dr. Ania Jastreboff and colleagues, had shown 24.2% weight loss at 48 weeks with the 12mg dose in people with obesity but without diabetes. At that dose, 100% of participants lost at least 5% of their body weight, 93% lost at least 10%, and 83% lost at least 15%.
In March 2026, Lilly reported TRIUMPH-2 results in participants with type 2 diabetes. According to Scientific American, participants on the highest dose lost an average of 36.6 pounds over 40 weeks with A1C reductions of 1.7-2 percentage points. Dr. Rozalina McCoy, an endocrinologist at the University of Maryland who was not involved in the trial, said the data showed "greater efficacy" over placebo for both blood sugar and weight compared with prior tirzepatide and semaglutide trials.
No other drug in controlled trials has matched these weight loss numbers. Whether that gap holds as more TRIUMPH data rolls in is the next question, but the Phase 2-to-Phase 3 trajectory has been consistent.
GIP, GLP-1, and glucagon: why three receptors beat two
To understand why retatrutide outperforms its predecessors, it helps to know what each receptor does when activated -- and why the combination produces effects none of the three hormones achieves alone.
GLP-1 receptor. This is the pathway semaglutide (Ozempic/Wegovy) made famous. When you activate GLP-1 receptors: insulin secretion increases in response to meals, gastric emptying slows, and appetite drops. The stomach emptying slowdown means food sits longer, you feel full faster, and you eat less. GLP-1 receptors in the brain -- particularly the hypothalamus and brainstem -- directly suppress appetite signals. This is the "I'm just not hungry" effect semaglutide users describe.
GIP receptor. Glucose-dependent insulinotropic polypeptide was historically considered a minor player in metabolism. That changed when tirzepatide showed that adding GIP agonism to GLP-1 produced substantially more weight loss than GLP-1 alone. GIP stimulates insulin release after meals, plays a role in lipid metabolism, and appears to enhance the appetite-suppressing effects of GLP-1 when the two are activated together. GIP agonism also seems to reduce the nausea caused by GLP-1, which matters for tolerability.
Glucagon receptor. This is where retatrutide diverges from everything else on the market. Glucagon, produced by alpha cells in the pancreas, has been primarily understood as a blood-sugar-raising hormone -- the opposite of insulin. But it does more than that. Glucagon reduces appetite through pathways distinct from GLP-1, increases energy expenditure by promoting thermogenesis, and drives fatty acid oxidation in the liver. It tells your liver to burn stored fat for fuel instead of storing it.
The liver effects are worth singling out. In a Phase 2 substudy published in Nature Medicine by Sanyal and colleagues, retatrutide at the 12mg dose reduced liver fat by 86% relative to baseline, and 93% of participants on that dose achieved normal liver fat levels (below 5%) by 48 weeks. For comparison, semaglutide reduces liver fat by approximately 50% after 72 weeks, and tirzepatide by about 47% after 52 weeks. The researchers found that near-maximal liver fat reduction occurred at around 20% body weight loss, suggesting the glucagon component drives additional fat clearance from the liver beyond what weight loss alone explains.
| Receptor | Natural hormone | Primary weight loss mechanism | Drugs that target it |
|---|---|---|---|
| GLP-1 | Glucagon-like peptide-1 | Appetite suppression, slowed gastric emptying | Semaglutide, tirzepatide, retatrutide |
| GIP | Glucose-dependent insulinotropic polypeptide | Amplifies GLP-1 effects, improves lipid metabolism | Tirzepatide, retatrutide |
| Glucagon | Glucagon | Increases energy expenditure, promotes liver fat oxidation | Retatrutide only |
The glucagon component raises an obvious concern: won't it raise blood sugar? In practice, GLP-1 and GIP agonism counterbalance glucagon's hyperglycemic effects. In the Phase 2 diabetes trial, retatrutide reduced HbA1c by up to 2.02% at 24 weeks, actually improving blood sugar control. The molecule's engineering ensures GIP receptor activation (where retatrutide is most potent) and GLP-1 receptor activation produce enough insulin stimulation to offset glucagon-driven glucose production.
All three mechanisms in one molecule: appetite suppression from GLP-1, metabolic amplification from GIP, and active liver fat-burning from glucagon. One weekly injection. The weight loss numbers reflect that layered pharmacology.
Retatrutide vs. Ozempic vs. Mounjaro: where each one stands
No head-to-head trial has directly compared retatrutide to semaglutide or tirzepatide. That trial may never happen -- pharmaceutical companies rarely fund studies that could show their existing products are inferior. But cross-trial comparisons, while imperfect, paint a consistent picture.
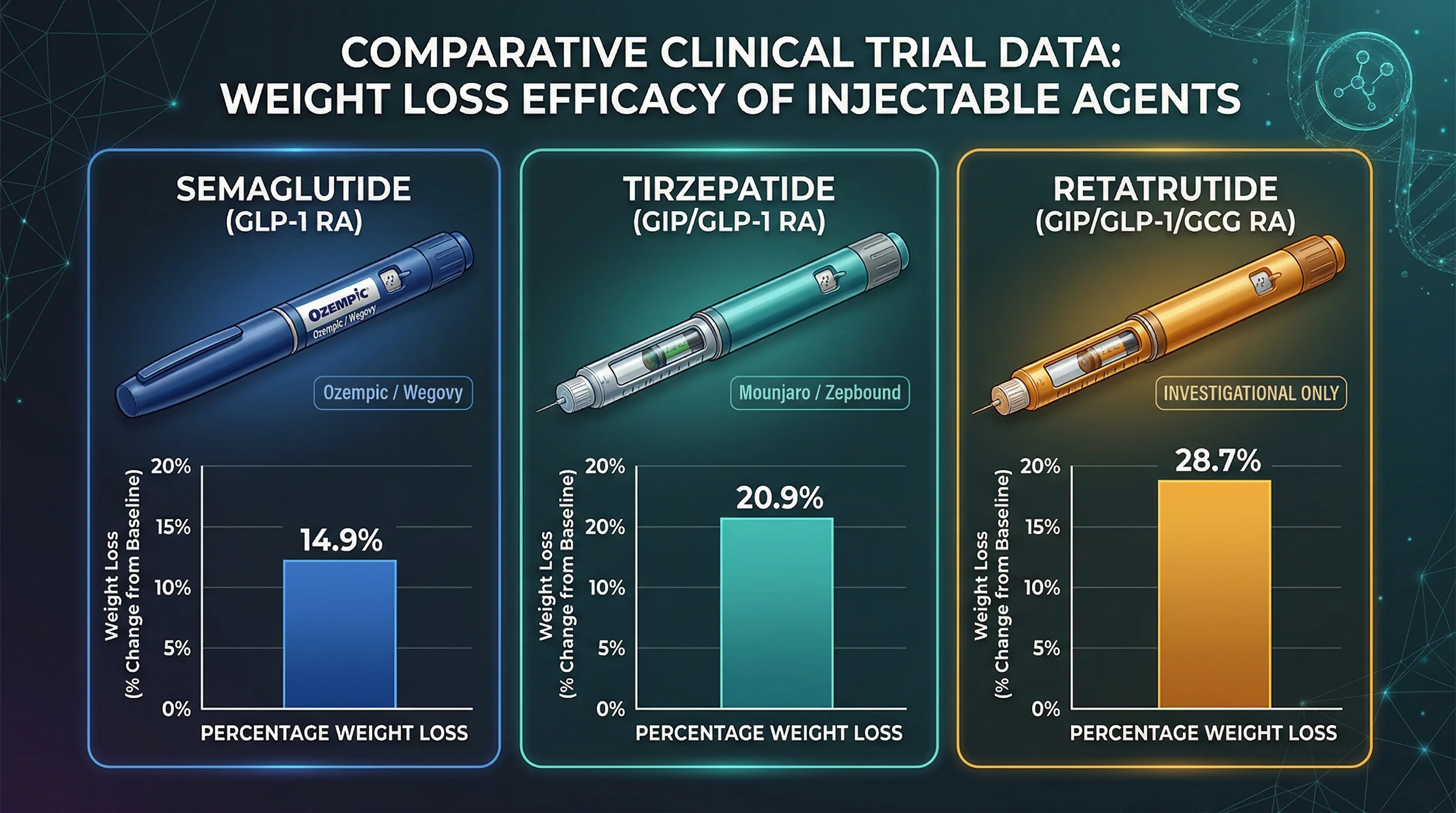
| Drug | Mechanism | Max weight loss (clinical trials) | Trial duration | FDA status |
|---|---|---|---|---|
| Semaglutide 2.4mg (Wegovy) | GLP-1 agonist | 14.9% | 68 weeks | Approved (2021) |
| Tirzepatide 15mg (Zepbound) | GIP + GLP-1 dual agonist | 20.9% | 72 weeks | Approved (2023) |
| Retatrutide 12mg | GIP + GLP-1 + glucagon triple agonist | 28.7% | 68 weeks | Phase 3 (not approved) |
A network meta-analysis published in the Journal of the Endocrine Society by Salhab and colleagues tried to quantify the gap. Pooling data from 12 clinical trials, they found retatrutide produced a mean difference of -23.77% in body weight versus placebo, compared to -16.79% for tirzepatide. Both significantly outperformed placebo, but retatrutide achieved roughly 7 percentage points more weight loss.
Dr. Daniel Drucker, a professor of medicine at the University of Toronto who has studied incretin biology for decades, called the retatrutide data "very solid" with "excellent" weight-loss and A1C results. He noted that the safety profile and discontinuation rates are "consistent" with established GLP-1 medicines.
Raw weight loss percentages don't tell the whole story, though. Several caveats apply to any cross-trial comparison.
Trial populations differed. TRIUMPH-4 enrolled people with obesity and knee osteoarthritis, who may be more motivated and more responsive to weight loss interventions. SURMOUNT-1 (tirzepatide) enrolled a broader obesity population. STEP (semaglutide) had its own eligibility criteria. Different baseline BMIs, comorbidity profiles, and dropout rates all affect the final numbers.
Trial durations varied. Retatrutide's Phase 3 ran 68 weeks, tirzepatide's SURMOUNT-1 ran 72 weeks, and semaglutide's STEP-1 ran 68 weeks. The weight loss curves for all these drugs were still trending downward at their endpoints, meaning longer trials might narrow or widen the gaps.
The efficacy estimand matters. The 28.7% figure for retatrutide represents participants who stayed on treatment. When including everyone randomized (the treatment-regimen estimand, which counts dropouts), the 12mg number drops to 23.7%. Still higher than tirzepatide's best numbers, but the gap is smaller than the headline figure suggests.
Retatrutide also carried higher adverse event rates and discontinuation rates than tirzepatide showed in its Phase 3 program. The superior efficacy comes with a trade-off that some patients may not accept.
What the comparison does make clear: each successive generation of incretin-based therapies has delivered meaningfully more weight loss. GLP-1 alone (semaglutide) gets roughly 15%. Adding GIP (tirzepatide) pushes past 20%. Adding glucagon on top (retatrutide) approaches 30%. The question for physicians and patients is whether the incremental benefit justifies the incremental risk.
Side effects and the dysesthesia question
Retatrutide shares the familiar gastrointestinal complaints of every incretin-based therapy -- nausea, diarrhea, vomiting, constipation -- but at somewhat higher rates, and with one new signal that caught analysts off guard.
| Side effect | Retatrutide 9mg | Retatrutide 12mg | Placebo |
|---|---|---|---|
| Nausea | 38.1% | 43.2% | 10.7% |
| Diarrhea | 34.7% | 33.1% | 13.4% |
| Constipation | 21.8% | 25.0% | 8.7% |
| Vomiting | 20.4% | 20.9% | 0.0% |
| Decreased appetite | 19.0% | 18.2% | 9.4% |
| Dysesthesia | 8.8% | 20.9% | 0.7% |
The GI side effects are broadly similar to what tirzepatide and semaglutide produce, though the nausea rate of 43% at the highest dose is higher than what tirzepatide showed in SURMOUNT trials. Lilly uses step-wise dose escalation -- starting at 2mg and increasing every four weeks -- to mitigate these effects, the same strategy used with tirzepatide and semaglutide.
The more concerning finding is dysesthesia -- abnormal skin sensations including tingling, burning, tenderness to touch, or a feeling that normal sensations are painful or unusual. At the 12mg dose, 20.9% of participants reported dysesthesia compared to just 0.7% on placebo. Roughly 1 in 5 patients.
This signal was not reported in the Phase 2 trial, which used the same doses. Why it appeared in Phase 3 but not Phase 2 is unclear. It could reflect the different patient population (TRIUMPH-4 enrolled people with knee osteoarthritis), the larger sample size catching a signal that Phase 2 missed, or genuine dose-duration effects. Lilly stated that these events were "generally mild and rarely led to treatment discontinuation," but BMO Capital Markets analysts noted they would be watching for this signal in upcoming TRIUMPH readouts.
Discontinuation rates tell a more nuanced story than the headline numbers suggest. Overall, 18.2% of patients on the 12mg dose and 12.2% on 9mg discontinued due to adverse events, compared to 4.0% on placebo. Those rates are higher than what tirzepatide showed in Phase 3. But Lilly made a notable disclosure: some discontinuations were attributed to "perceived excessive weight loss" rather than adverse events.
When Lilly restricted the analysis to patients with baseline BMI of 35 or higher, discontinuation rates dropped to 12.1% (12mg) and 8.8% (9mg) versus 4.8% placebo. As Fierce Biotech reported, this suggests people with lower starting weights found the pace of weight loss excessive or alarming. For patients with severe obesity -- the population most likely to be prescribed a drug this potent -- the tolerability picture is less concerning.
The meta-analysis of Phase 2 data by Abouelmagd and colleagues across three randomized controlled trials encompassing 878 patients found no statistically significant difference in overall adverse event rates between retatrutide and placebo groups (relative risk: 1.11, P = 0.24). The individual GI side effects were higher with retatrutide, but serious adverse events were rare across all trials.
The open question is whether dysesthesia represents a temporary nuisance or something that could limit the drug's real-world adoption. If upcoming TRIUMPH results show the same signal at similar rates, regulators will want to understand the mechanism. If it doesn't replicate, the Phase 3 finding may be an artifact of the specific study population.
From trials to pharmacy shelves: the road to approval
Retatrutide is not approved by the FDA or any other regulatory agency. It remains investigational. But the clinical development program is large and moving fast.
The TRIUMPH program consists of four Phase 3 trials using a basket trial design -- a relatively novel clinical trial architecture that evaluates the same drug across multiple related conditions simultaneously. The four trials have enrolled more than 5,800 participants across dozens of countries.
| Trial | Population | Key endpoints | Expected completion |
|---|---|---|---|
| TRIUMPH-1 | Obesity/overweight (with OSA subset) | Weight loss, sleep apnea | Mid-2026 |
| TRIUMPH-2 | Type 2 diabetes with obesity (with OSA subset) | Weight loss, A1C, sleep apnea | Mid-2026 |
| TRIUMPH-3 | Obesity with established cardiovascular disease | Weight loss, CV outcomes | Early 2026 |
| TRIUMPH-4 | Obesity with knee osteoarthritis | Weight loss, pain reduction | Completed (Dec 2025) |
TRIUMPH-4 was first to report. Seven additional Phase 3 readouts are expected in 2026, including TRIUMPH-1 (the broadest obesity trial) and TRIUMPH-2 (diabetes). Citi analysts have noted that TRIUMPH-1 could show weight loss exceeding 30% because it runs for 80 weeks -- 12 weeks longer than TRIUMPH-4 -- and the weight loss curves in these trials typically haven't plateaued at the 68-week mark.
Beyond TRIUMPH, Lilly is also running a separate Phase 3 program called SYNERGY evaluating retatrutide for metabolic dysfunction-associated steatotic liver disease (MASLD), building on Phase 2 data that showed 86% liver fat reduction at 48 weeks. If those results hold in Phase 3, retatrutide could become the first drug approved for MASLD -- a condition affecting an estimated 38% of the global adult population with no approved treatment in the US or Europe.
The regulatory timeline depends on when Lilly compiles and submits data. Based on public statements and analyst estimates: NDA filing in late 2026 or early 2027, with an FDA decision possible in 2027 or 2028 depending on the review pathway. GlobalData forecasts a 2027 approval and projects retatrutide reaching $15.6 billion in annual sales by 2031.
For context, Lilly's existing weight loss franchise is already enormous. Zepbound (tirzepatide for obesity) generated $3.6 billion in Q3 2025 alone. Truist Securities estimates that Lilly's weight loss trio -- Mounjaro, Zepbound, and orforglipron -- could hit $101 billion in peak sales. Retatrutide would sit on top of that portfolio as the highest-efficacy option for patients who need or want the most aggressive pharmacological weight loss available.
That positioning matters. Lilly isn't replacing tirzepatide with retatrutide -- it's building a tiered portfolio. Patients who respond well to tirzepatide with manageable side effects may stay on it. Retatrutide would likely be reserved for patients with severe obesity, those who haven't achieved sufficient weight loss on existing therapies, or those with specific comorbidities (like MASLD or osteoarthritis) where the triple agonist's broader effects offer additional benefit.
One caveat: Phase 3 programs can still produce surprises. Long-term cardiovascular outcome data (from TRIUMPH-3 and a separate outcomes trial) will be watched closely. And the dysesthesia signal needs further characterization. If the remaining readouts are consistent with TRIUMPH-4, Lilly will file what could be the most effective weight loss drug ever submitted to the FDA.
Frequently Asked Questions
Is retatrutide available to prescribe or buy right now?
No. Retatrutide is an investigational drug not approved by the FDA or any other regulatory agency. It is only available through clinical trial enrollment. Based on the current timeline, the earliest it could reach pharmacies is late 2027 or 2028, assuming positive Phase 3 results and a successful regulatory review. Any product claiming to sell "retatrutide" outside of a clinical trial is not legitimate.
How does retatrutide compare to surgery for weight loss?
The weight loss numbers are strikingly similar. Bariatric surgery typically produces 25-30% weight loss in the first year after surgery, while retatrutide's Phase 3 data showed 28.7% at 68 weeks. The key differences: surgery is a one-time procedure with permanent anatomical changes, while retatrutide requires ongoing weekly injections. What happens to weight after stopping retatrutide is not yet well characterized in Phase 3 trials, though Phase 2 data showed some weight regain after discontinuation.
What is dysesthesia and should patients be worried about it?
Dysesthesia refers to abnormal skin sensations -- tingling, burning, or tenderness to touch. It affected about 20.9% of patients on the 12mg dose in TRIUMPH-4. Lilly reported these events were generally mild and rarely caused patients to stop treatment. The mechanism is not yet understood, and the signal did not appear in Phase 2 trials. Researchers will be watching upcoming trial results to determine whether it's a consistent finding or specific to the TRIUMPH-4 population.
Will retatrutide replace Ozempic or Mounjaro?
Likely not. Lilly is positioning retatrutide as a higher-efficacy option within a tiered portfolio, not as a replacement for tirzepatide (Mounjaro/Zepbound). Semaglutide (Ozempic/Wegovy) is made by Novo Nordisk, a different company. Each drug occupies a different point on the efficacy-tolerability spectrum. Patients with moderate weight loss goals and good GLP-1 tolerance may be well served by existing options. Retatrutide would target those needing or wanting the most aggressive pharmacological approach.
What about long-term safety -- is 68 weeks enough to know if retatrutide is safe?
It's enough for initial regulatory approval, but long-term questions remain. The TRIUMPH program includes a cardiovascular outcomes trial that will provide longer-duration safety data. The systematic review by Abouelmagd and colleagues across Phase 2 data found no significant difference in overall adverse event rates between retatrutide and placebo, but the total number of patient-years of exposure is still limited. Post-marketing surveillance will be important once the drug is approved.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.