The drug got a nickname before it got a price tag. After Eli Lilly's investigational triple-agonist retatrutide produced a mean 24.2% body-weight reduction at 48 weeks in its Phase 2 trial published in The New England Journal of Medicine, science writers reached for monster metaphors. "Godzilla of weight loss" stuck because it captured the magnitude of effect this molecule produces relative to semaglutide and tirzepatide. The numbers were genuinely larger by a margin that the field hadn't seen.
Then came December 11, 2025, and the first Phase 3 readout. Lilly announced that participants in TRIUMPH-4 taking the 12 mg dose lost an average of 28.7% of their body weight, about 71.2 lbs, over 68 weeks. The Phase 2 result wasn't a fluke. The Phase 3 result, if anything, was larger.
That's the part that gets the headlines. The part that doesn't is everything around it. The study was in adults with obesity and knee osteoarthritis specifically. The discontinuation rate from side effects ran almost five times the placebo rate. A new skin-sensation side effect appeared. And the drug remains investigational: not FDA-approved, not for sale, and the cardiovascular outcomes data won't land until 2029. This guide unpacks what the trials actually show, what they don't, and why "Godzilla" is a useful frame for the science but a misleading one for the stakes.
What Retatrutide Actually Is
Retatrutide (formerly LY3437943) is a synthetic peptide given as a subcutaneous injection once a week. It's a single molecule that binds and activates three different hormone receptors: the GLP-1 receptor, the GIP (glucose-dependent insulinotropic polypeptide) receptor, and the glucagon receptor. That triple action is what makes it pharmacologically novel. Semaglutide hits one of those receptors. Tirzepatide hits two. Retatrutide hits all three.
Each receptor does something different in the metabolism of body weight. GLP-1 reduces appetite signals to the brain and slows the rate at which the stomach empties. GIP reinforces satiety signals through receptors in the hypothalamus. Glucagon, the third receptor and the genuinely new addition, does the unusual thing of raising energy expenditure and triggering lipolysis, the breakdown of stored fat. A 2025 review in Biomolecules described the receptor binding profile in detail: retatrutide is most potent at the GIP receptor (EC50 of 0.0643 nM), then GLP-1 (0.775 nM), then glucagon (5.79 nM). So it grabs GIP hardest, but the glucagon arm is what changes the energy equation.
Picture body weight as a savings account. Semaglutide and tirzepatide reduce deposits: they make you eat less and absorb calories more slowly. Retatrutide does that and increases withdrawals, telling your liver and fat tissue to spend more energy. Faster and larger weight loss is what you'd predict from that combination, and the trials so far suggest the prediction holds.
The drug has a half-life of about six days, which is what makes once-weekly dosing practical, and it's metabolized primarily in the liver without involving the cytochrome P450 enzymes. That last detail matters less for everyday patients than for people on multiple medications, since it lowers the risk of one drug interfering with another's metabolism.
Practically, none of this means anything yet for patients in pharmacies. Retatrutide is investigational. Lilly manufactures it for clinical trials. The molecules being sold by some online vendors and wellness clinics under the same name have not been verified by any regulator and should not be assumed equivalent.
The Phase 2 Data That Started the Hype
The trial that put retatrutide on the radar was a 48-week Phase 2 study led by Dr. Ania Jastreboff of Yale School of Medicine, published simultaneously in The New England Journal of Medicine and presented at the American Diabetes Association Scientific Sessions in June 2023. It enrolled 338 adults with obesity (BMI 30 or higher, or BMI 27-30 with at least one weight-related condition) and randomized them across placebo and four retatrutide doses: 1 mg, 4 mg, 8 mg, and 12 mg, with various starting doses to test how aggressive titration affected tolerability.
The results were the kind that change conversations in endocrinology departments. At 48 weeks, mean body-weight reduction reached 24.2% in the 12 mg group, 22.8% in the combined 8 mg group, 17.1% in the combined 4 mg group, and 8.7% in the 1 mg group, versus 2.1% on placebo. The 24.2% number drove the "Godzilla" coverage. It landed in the same neighborhood as bariatric surgery outcomes, which had previously been the only reliable way to produce that magnitude of weight loss.
The threshold-achievement data was equally striking. By week 48, 100% of participants on the 8 mg or 12 mg dose had lost at least 5% of body weight; 93% on the 12 mg dose had lost at least 10%; and 83% had lost at least 15%, versus 27%, 9%, and 2% on placebo at those same thresholds. There was no plateau. The weight-loss curve was still descending when the trial ended, suggesting that longer treatment might produce larger effects.
The cardiometabolic numbers traveled with the weight numbers. A 2025 systematic review and meta-analysis pooling three randomized trials with 878 patients reported that retatrutide produced mean reductions of 9.88 mm Hg in systolic blood pressure, 3.88 mm Hg in diastolic, 23.51 mg/dL in fasting plasma glucose, and 0.91 percentage points in hemoglobin A1c, all statistically significant. During the Phase 2 trial, 41% of participants in the combined 8 mg group and 30% in the 12 mg group discontinued at least one antihypertensive medication because their blood pressure improved enough that they no longer needed it. That is the kind of secondary outcome that, replicated, changes how clinicians think about the drug class.
A separate Phase 2 substudy looked at liver fat, specifically metabolic dysfunction-associated fatty liver disease, the chronic accumulation of fat in liver tissue that affects an estimated two-thirds of adults with type 2 diabetes. In that substudy, 86% of participants on the 12 mg dose and 79% on 8 mg had achieved a normal liver fat content (under 5%) by week 24, climbing to 93% and 89% by week 48, versus 0% on placebo at week 24. A separate Phase 2 trial in adults with type 2 diabetes (Rosenstock et al.) reported that A1C reductions reached up to 2.16 percentage points and up to 82% of participants brought their A1C below the diagnostic threshold of 6.5%. The drug was, on these markers, behaving more like a metabolic-syndrome therapy than a weight-loss drug.
For patients trying to make sense of this, the practical takeaway is narrower than the headlines. Phase 2 produced extraordinary numbers in carefully selected, closely supervised participants on the highest dose. Real-world results, with shorter monitoring and less-controlled lifestyle support, would almost certainly look smaller. Phase 2 did its job, though: it justified the much larger Phase 3 program, and that program is now reporting out.
TRIUMPH: What Phase 3 Has Revealed (and What It Hasn't)
The Phase 3 program is called TRIUMPH, and it is enormous by retatrutide-specific standards. Lilly reports that more than 5,800 participants are enrolled across four core registrational trials, with a fifth study (TRIUMPH-OUTCOMES) eventually enrolling roughly 10,000 more for the cardiovascular safety question. The trials test five doses (2, 4, 6, 9, and 12 mg) using a step-wise titration that escalates every four weeks.
The first Phase 3 readout came on December 11, 2025. TRIUMPH-4 (NCT05931367) randomized 445 adults with obesity and knee osteoarthritis (without diabetes) to placebo, retatrutide 9 mg, or retatrutide 12 mg, for 68 weeks. The choice of population was unusual but deliberate. Knee osteoarthritis is one of the most common, painful, and disabling complications of obesity, and current treatment options short of joint replacement are limited.
The headline weight-loss numbers exceeded Phase 2. Using the efficacy estimand (the analysis that includes only participants who stayed on study drug), mean body-weight reduction was 28.7% at the 12 mg dose, 26.4% at 9 mg, and 2.1% on placebo at 68 weeks. With a baseline average BMI of 40 and starting weight of about 248 pounds, the magnitude of weight loss was described by the trial discussants as unprecedented in clinical trials. The same source confirmed that 58% of 12 mg participants achieved at least 25% weight loss, with the 9 mg dose producing 47%.
The treatment-regimen estimand, a more conservative analysis that treats trial dropouts as failures, produced smaller but still substantial numbers. In that analysis, mean weight loss was 23.7% at 12 mg, 20.0% at 9 mg, and 4.6% on placebo. That gap matters. The efficacy-estimand number is what gets quoted in press releases, but the treatment-regimen number is closer to what you'd expect in real clinical practice, where some patients won't tolerate the drug.
TRIUMPH-4 was also designed to test whether retatrutide reduced osteoarthritis pain. It did. WOMAC pain subscale scores, a validated patient-reported pain measure, dropped by 4.5 points (75.8%) at 9 mg, 4.4 points (74.3%) at 12 mg, and 2.4 points (40.3%) on placebo. About one in eight retatrutide patients reported being completely free of knee pain at the end of the 68-week trial, versus roughly 4% on placebo. Systolic blood pressure dropped by 14.0 mm Hg at the highest dose. The cardiometabolic benefits seen in Phase 2 carried over.
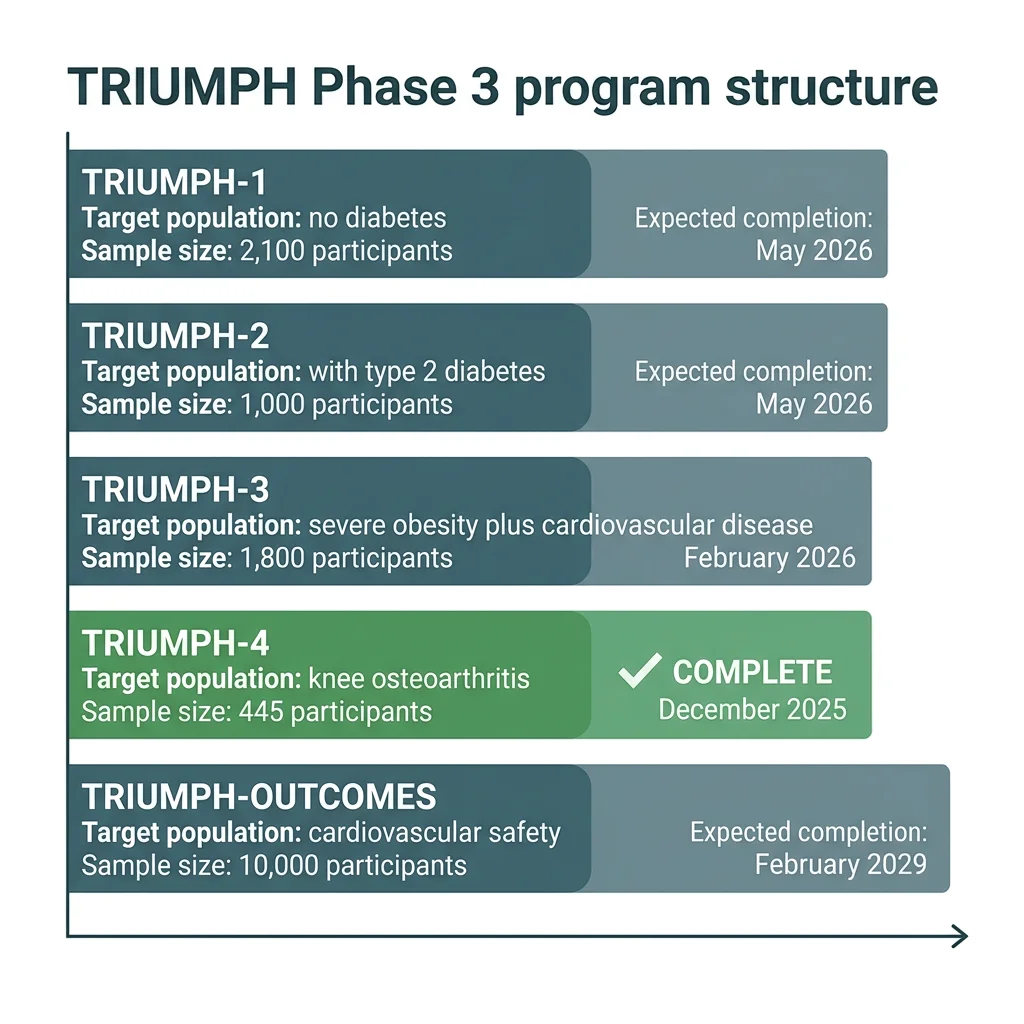
One important caveat sits behind those numbers. TRIUMPH-4 is one trial in one population: people with obesity and knee osteoarthritis, with a baseline BMI averaging 40 and 84% of participants meeting criteria for severe obesity. The seven other Phase 3 readouts expected through 2026 will test the drug in adults without obesity-related complications, with type 2 diabetes, with established cardiovascular disease, with sleep apnea, and across a wider BMI range. Until those data appear, generalizing the TRIUMPH-4 numbers to "people with obesity" overstates what the trial actually proved.
How Retatrutide Compares to Tirzepatide and Semaglutide
The honest comparison answer is that there is no published head-to-head randomized controlled trial of retatrutide against tirzepatide or semaglutide. Every comparison in the literature is either a meta-analysis pooling separate trials or a network meta-analysis stitching them together statistically. Both methods have known weaknesses (different populations, different trial durations, different placebo responses), and the field has been openly calling for direct comparators.
The indirect comparisons consistently point in the same direction, though. A 2025 network meta-analysis by Salhab and colleagues, published in the Journal of the Endocrine Society, pooled 12 trials and reported that retatrutide produced mean weight loss of 23.77% versus 16.79% for tirzepatide; in absolute terms, that worked out to 16.34 kg versus 11.82 kg. The same analysis found higher adverse-event relative risk for retatrutide (4.10) than tirzepatide (2.78), so the efficacy gap comes with a tolerability cost.
The comparison broadens further when you include the rest of the GLP-1 class. The Abouelmagd meta-analysis summarized the published context: dulaglutide 4.5 mg produces about 5% weight loss; semaglutide 2 mg around 7.2%; semaglutide 2.4 mg in the STEP 2 trial 10.6%; tirzepatide 15 mg about 12% over 40-72 weeks. Against that backdrop, retatrutide's Phase 2 24.2% number and Phase 3 28.7% number look less like Godzilla and more like the next obvious step in a class of drugs that has been improving steadily for two decades.
| Drug | Receptor targets | Approval status | Mean weight loss (best dose) |
|---|---|---|---|
| Semaglutide (Wegovy) | GLP-1 | FDA-approved | 10.6% (STEP 2, 2.4 mg) |
| Tirzepatide (Zepbound) | GLP-1 + GIP | FDA-approved | 12-16.8% (15 mg) |
| Retatrutide | GLP-1 + GIP + glucagon | Investigational | 24.2% Phase 2; 28.7% Phase 3 (12 mg) |
One often-cited number sharpens the picture. In a Phase 2 trial of adults with type 2 diabetes, up to 63% of participants treated with retatrutide lost at least 15% of body weight by week 36; in the SURPASS-2 trial, only 40% of participants on tirzepatide 15 mg achieved that same threshold by week 40. Same threshold, similar timeframe, different drug. Patient populations differed, so this doesn't prove retatrutide is universally better, but it does suggest the gap isn't an artifact of cherry-picked dose comparisons.
Body composition is what's missing from any cross-trial comparison. The Katsi review explicitly flagged this gap: weight loss isn't always good weight loss, since losing lean mass and bone density alongside fat changes the long-term picture, and current Phase 2 and 3 trials have not adequately characterized the lean-to-fat ratio of the weight that comes off. Until those analyses are published, the question of what kind of weight retatrutide removes remains open.
Side Effects, Safety Signals, and Cardiovascular Questions
The headline tolerability story is the gastrointestinal one, the same one that has shaped patient experience with semaglutide and tirzepatide, just somewhat amplified. In TRIUMPH-4, nausea affected 38.1% of participants on 9 mg and 43.2% on 12 mg, versus 10.7% on placebo. Diarrhea hit 34.7% and 33.1% (vs 13.4%); constipation 21.8% and 25.0% (vs 8.7%); vomiting 20.4% and 20.9% (vs 0.0%). These rates are higher than what tirzepatide produces in comparable trials but consistent with the dose-related pattern seen across the incretin class.
Phase 3 also produced a genuine surprise: a category Lilly called dysesthesia, meaning abnormal skin sensations, often described as tingling, burning, or pins-and-needles. Dysesthesia occurred in 8.8% of participants on 9 mg and 20.9% on 12 mg, versus 0.7% on placebo. Lilly characterized the events as generally mild and rarely a cause for stopping the drug. The signal had been hinted at in earlier reviews. The 2025 Katsi review listed "skin hyperesthesia" as a less-common adverse effect in pre-Phase-3 reporting. But the Phase 3 magnitude is larger than expected and warrants attention as more data emerges.
Discontinuation rates tell their own story. In TRIUMPH-4, treatment discontinuation due to adverse events ran 12.2% at 9 mg, 18.2% at 12 mg, and 4.0% on placebo. Some of those discontinuations were for "perceived excessive weight loss," patients leaving the trial because they thought they'd lost too much. Filtering to participants with a baseline BMI of 35 or higher, the 12 mg discontinuation rate fell to 12.1%, suggesting that the drug is more tolerable in the higher-BMI population it was designed for.
Two safety signals from earlier-phase work deserve continued monitoring. The original Phase 2 trial documented a transient, dose-dependent rise in heart rate that peaked around week 24 and declined thereafter; whether that pattern persists in longer Phase 3 follow-up is one of the open questions for the obesity indication. A feature in The Pharmaceutical Journal also noted that Phase 2 reporting included one case of pancreatitis at higher doses, a single case in a small trial that doesn't establish causality but joins a long-running watch on the GLP-1 class for pancreatic safety.
Then there's what the trials haven't yet answered. Does retatrutide reduce heart attacks, strokes, and cardiovascular death? That cardiovascular outcomes question is being studied in TRIUMPH-OUTCOMES, a 10,000-participant trial in adults with established atherosclerotic cardiovascular disease. That trial isn't scheduled to complete until February 2029. Until those data land, retatrutide's regulatory case will rest on weight loss and metabolic improvements, not on the kind of hard cardiovascular outcomes that supported semaglutide's expanded indications.
Anticipated contraindications, though not yet formally established, mirror those of the existing GLP-1 class. The 2024 Deravi commentary listed personal or family history of medullary thyroid carcinoma, multiple endocrine neoplasia syndrome type 2, and prior pancreatitis as expected exclusions based on Phase 2 trial criteria and the rest of the drug class. None of that is settled. None of that is a substitute for clinician judgment.
Regulatory Timeline: When Could Retatrutide Reach Patients?
The short version: not yet, and not soon. Retatrutide is investigational. The FDA has not approved it. Neither has the European Medicines Agency, the UK MHRA, or any other regulator. The molecule does not exist as an approved prescription medication anywhere in the world.
The longer version requires reading Lilly's pipeline carefully. After the December 2025 TRIUMPH-4 readout, Lilly stated that seven additional Phase 3 trials are expected to complete in 2026. Those readouts will produce the data Lilly needs to file for the broader obesity indication. Filing typically takes months; FDA review of a major new molecular entity for obesity then takes ten to twelve months; FDA may also request an advisory committee review. Realistically, an FDA approval for retatrutide for chronic weight management could land in late 2026, though sometime in 2027 looks more plausible given the program's complexity.
The cardiovascular outcomes indication sits on a longer timeline still. TRIUMPH-OUTCOMES is set to complete in February 2029. Even if the obesity indication is approved in 2027, the kind of expanded cardiovascular labeling that broadened semaglutide's market wouldn't follow until at least 2029 or 2030.
What this means in plain terms: any retatrutide that reaches a US patient in 2026 is doing so either through a clinical trial or through an unregulated, compounded, or gray-market source. Trial enrollment is real medicine. The other routes are not.
Cost is the other practical question. Lilly has not announced a price. Independent reviewers have speculated based on current GLP-1 pricing. The 2024 Deravi commentary suggested around $1,000 for a 30-day supply, while a separate review estimated $1,200 to $1,600 per month. Either figure puts retatrutide squarely in the same affordability problem that has limited semaglutide and tirzepatide access for uninsured and underinsured patients.
The Hype Problem: Investigational Status vs. Media Narrative
"Godzilla of weight loss" is a vivid headline, and it does describe something real about the magnitude of effect this molecule produces in clinical trials. It also obscures three things that matter when patients translate headlines into decisions.
First: trial settings overestimate real-world performance. Phase 2 and Phase 3 participants get weekly check-ins, structured lifestyle counseling from registered dietitians, and the social pressure of being in a study. The Deravi commentary explicitly flagged this issue: lifestyle interventions of the intensity used in Phase 2 may not be feasible for many patients, so real-world weight loss with retatrutide will likely be smaller than the trial numbers suggest. Real-world discontinuation rates with currently approved GLP-1 drugs already run between 20% and 50% within the first year of use. Trial discipline doesn't survive into pharmacy shelves.
Second: every comparison number you've seen so far is indirect. The 28.7% Phase 3 weight-loss number from TRIUMPH-4 was generated in a population with average BMI 40 and concurrent knee osteoarthritis. The tirzepatide 12-15% number comes from different trials, different populations, different durations. There is no published trial in which the same patients were randomized to retatrutide versus tirzepatide. Until there is, treat any "retatrutide is X% more effective than tirzepatide" claim as informed speculation, not established fact.
Third: even within the trials, the data set is narrow. The Katsi review specifically called out that retatrutide has not been studied in children or adolescents and is not indicated for use in patients under 18. Long-term safety beyond 68 weeks is unknown for everyone. Cardiovascular outcomes are unknown for everyone. The population in TRIUMPH-4 was selected for one specific obesity-plus-osteoarthritis indication, not for the full range of patients who might want the drug.
None of this is a reason to dismiss what the trials show. The signal is real. Three peer-reviewed reviews independently characterize retatrutide as one of the most striking weight-loss results published in any drug class: "a game changer in obesity pharmacotherapy"; "a promising therapeutic option" with "superior weight loss outcomes"; a molecule whose efficacy "support[s] moving retatrutide into phase 3 trials". The evidence base supports cautious optimism. It does not support patients sourcing unregulated compounded versions in 2026 because they read a headline.
The most useful thing a patient interested in retatrutide can do today is exactly what Lilly's enrollment pages suggest: ask a physician about clinical trial enrollment in their region, monitor the seven Phase 3 readouts due across 2026, and work with a qualified clinician on the FDA-approved options that exist now. If the Godzilla arrives, it will arrive on prescription pads. Anything else is a different drug.
Frequently Asked Questions
Is retatrutide FDA-approved?
No. As of the most recent reporting available, retatrutide is investigational only. It has not been approved by the FDA, the European Medicines Agency, the UK MHRA, or any other regulator. Lilly has produced positive Phase 2 and one positive Phase 3 readout (TRIUMPH-4, December 2025), with seven additional Phase 3 readouts expected to land across 2026. A regulatory submission for the obesity indication would follow those readouts; an actual approval is realistically expected in 2026-2027 at the earliest.
How does retatrutide compare to Ozempic and Mounjaro?
In indirect comparisons across separate trials, retatrutide has produced larger mean weight loss than either semaglutide (the active ingredient in Ozempic and Wegovy) or tirzepatide (the active ingredient in Mounjaro and Zepbound). In TRIUMPH-4, the 12 mg retatrutide dose produced 28.7% mean weight loss at 68 weeks, compared to roughly 10.6% for semaglutide 2.4 mg in STEP 2 and 12-16.8% for tirzepatide depending on the trial. There is no direct head-to-head trial yet, so these comparisons should be treated as suggestive rather than definitive. Retatrutide also produced higher rates of nausea, diarrhea, and treatment discontinuation than tirzepatide does in comparable trials.
Can I get retatrutide from a compounding pharmacy?
Some online vendors and wellness clinics market compounded peptides labeled as retatrutide, but these have not been verified by the FDA or any other regulator. Compounded retatrutide is not the same as the molecule being studied in Lilly's TRIUMPH program. The manufacturing standards, purity, and dosing accuracy are not guaranteed. The FDA has issued warnings about compounded versions of other GLP-1 drugs for similar reasons. Patients interested in retatrutide should ask their physician about clinical trial enrollment rather than purchasing from unregulated sources.
What are the most common side effects?
Gastrointestinal symptoms (nausea, diarrhea, vomiting, constipation, and decreased appetite) are the most common, occurring in 19% to 43% of participants depending on dose in the Phase 3 TRIUMPH-4 trial. Phase 3 also revealed an unexpected signal of dysesthesia, an abnormal skin sensation that occurred in 8.8% to 20.9% of retatrutide patients versus 0.7% on placebo. A transient rise in heart rate was documented in Phase 2. Treatment discontinuation due to side effects ran 12% at the 9 mg dose and 18% at the 12 mg dose in TRIUMPH-4.
Is retatrutide safer or more effective than bariatric surgery?
That's not a head-to-head question current research can answer. Bariatric surgery remains the most effective sustained weight-loss intervention for severe obesity, but it carries surgical risk and is not appropriate for everyone. Retatrutide's Phase 3 weight-loss numbers (around 28.7% at 68 weeks) overlap with the lower end of bariatric outcomes, which is why the comparison gets made. It is a comparison across very different interventions, in different populations, with different long-term evidence bases. The cardiovascular safety question for retatrutide will not be answered until TRIUMPH-OUTCOMES completes in 2029. Anyone weighing the two options should make that decision with a clinician who knows their full medical picture, not based on a percentage in a press release.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.