Search "tesamorelin" on TikTok or Instagram and the feed fills with women in their forties showing flatter midsections, holding up vials, quoting an "18% visceral fat reduction" study. The trend is real. The drug is real. What gets lost in the captions is that this peptide was FDA-approved for one specific population — adults living with HIV who develop a particular type of central fat redistribution — and the women being marketed to right now were not part of any of its pivotal trials.
This article walks through what the research actually shows, why menopausal belly fat is biologically different, what the off-label landscape looks like in 2026, and how the side-effect profile reads with the marketing layer off.
Contents
- What tesamorelin actually is — and what the FDA approved
- Why visceral belly fat after 40 is biologically different
- The trial data: 15 to 18% — and what that doesn't mean
- The off-label landscape: longevity clinics, telehealth, compounding
- Side effects, contraindications, and the cancer question
- Cost reality and what insurance actually covers
- Who is a candidate — and who is being marketed to
- Alternatives that have evidence in women over 40
- Frequently Asked Questions
- Sources Used in This Guide
What tesamorelin actually is — and what the FDA approved
Tesamorelin is a synthetic analog of growth hormone-releasing hormone, or GHRH. It acts on the anterior pituitary to stimulate the body's own pulsatile growth hormone secretion, which then raises insulin-like growth factor-1 (IGF-1). That cascade preferentially mobilizes deep abdominal fat — visceral adipose tissue — over subcutaneous fat, as a 2019 PMC review summarizes.
The FDA first approved it as Egrifta in November 2010 (Theratechnologies). Egrifta SV replaced the original; in March 2025 the agency approved Egrifta WR, a concentrated formulation reconstituted weekly. According to Prime Therapeutics' approval summary, Egrifta WR is indicated for "reduction of excess abdominal fat in HIV-infected adult patients with lipodystrophy" — the same indication since 2010.
The labeling is unusually direct about what tesamorelin is not. MedlinePlus (NIH/NLM) states the drug is "explicitly not intended for weight loss." The Egrifta WR label itself includes "not for weight loss management" among its limitations of use, and notes that long-term cardiovascular safety has not been established. Off-label prescribing is permitted, but every claim made outside HIV medicine sits outside the FDA-reviewed evidence base.
Why visceral belly fat after 40 is biologically different
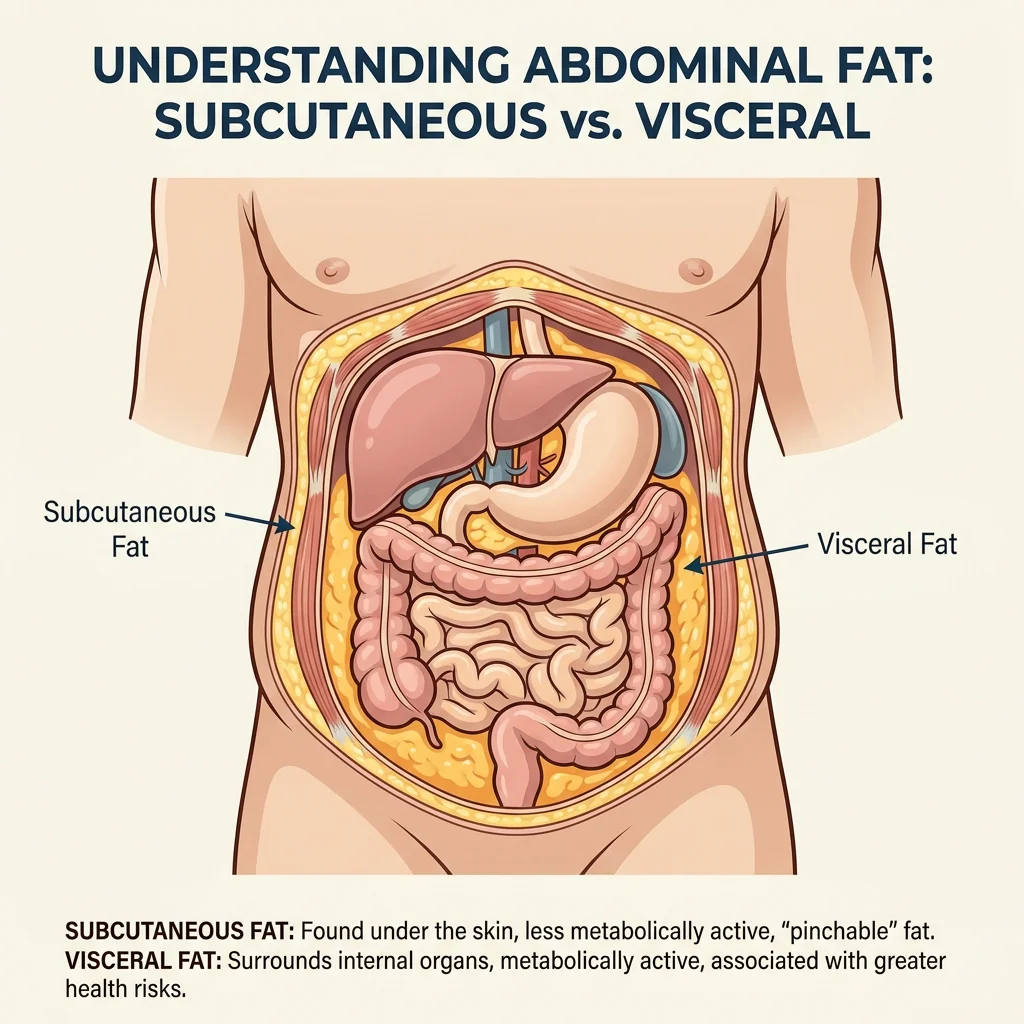
Interest in tesamorelin among women over 40 makes sense because the fat that accumulates around the midsection during the menopause transition is biologically a different tissue than the subcutaneous fat carried in their twenties. Visceral fat sits inside the abdominal cavity, wraps around organs, secretes inflammatory signals, and tracks cardiometabolic risk much more tightly than waist size alone.
The Study of Women's Health Across the Nation, an NIH-funded longitudinal cohort, documents a clear inflection point. SWAN investigators reported that "about 2 years prior to the final menstrual period, the rate of fat gain doubled and lean mass started to decline." That doubling is what many women describe as "I didn't change anything and suddenly nothing fit." A 2026 cross-sectional analysis of 325 women, published in the Journal of Clinical Medicine by Szeliga and colleagues, found that visceral fat area in normal-weight women rose from 36.4 cm² premenopause to 55.7 cm² postmenopause — about a 53% increase even in women whose BMI never crossed into overweight territory.
The biology is driven primarily by ovarian hormone loss. A 2021 review of menopause and metabolic health describes how estrogen deficiency triggers transient hyperphagia and reduces spontaneous activity such as fidgeting, while follicle-stimulating hormone may independently shift fat distribution. The same review reported that metabolic syndrome rises from about 16% in premenopausal women to 42% in postmenopausal women.
None of that biology mirrors the original tesamorelin trial population. HIV-associated lipodystrophy is a fat redistribution syndrome driven by older antiretroviral regimens and chronic inflammation. The shape on the body looks similar — a hard, protruding midsection — but the upstream mechanism is different. The drug's mechanism is not specific to either condition; it works at the pituitary level. Whether it will be similarly effective and similarly safe in a perimenopausal woman is a question the published trial record does not answer.
The trial data: 15 to 18% — and what that doesn't mean
Almost every social-media post about tesamorelin cites the same headline — 15 to 18% reduction in visceral fat. Both numbers come from real peer-reviewed research, and both deserve careful reading.
The pivotal RCT, Falutz and colleagues' 2007 paper in the New England Journal of Medicine, enrolled 412 HIV patients with treatment-associated central fat accumulation. The cohort was 86% male. Participants received tesamorelin 2 mg subcutaneously daily or placebo for 26 weeks. Visceral adipose tissue decreased 15.2% with tesamorelin and increased 5% with placebo. Triglycerides fell about 50 mg/dL; IGF-1 rose 81%.
The "18%" figure that travels online comes from the open-label extension. Falutz and colleagues reported in AIDS in 2008 that continued tesamorelin from week 26 through 52 sustained roughly 18% VAT reduction — but the same paper noted: "Upon discontinuation of tesamorelin, VAT reaccumulated." A 2010 pooled analysis of 806 patients, published in JCEM, confirmed about -17.5% VAT at 52 weeks among continuing users, with loss of benefit in those who switched to placebo at week 26.
This is the most important practical fact the marketing layer obscures. The drug suppresses the depot during use; it rebuilds when injections stop. Indefinite dosing is required for ongoing effect — at full retail price, off-label, with no insurance coverage. The trials also show tesamorelin produces almost no overall weight loss; it shifts body composition rather than the scale. Subcutaneous fat is largely preserved.
The closest non-HIV trial is small but informative. Makimura and colleagues in JCEM 2012 studied 60 abdominally obese non-HIV adults with reduced growth hormone secretion. After 12 months of tesamorelin 2 mg/day, treated participants lost about 35 cm² more visceral fat than placebo (P=0.003), with a +92 μg/L IGF-1 rise and no significant change in fasting glucose or HbA1c. The population was selected for documented low GH secretion — not perimenopausal women. No published randomized trial has tested tesamorelin specifically in perimenopausal or early postmenopausal women.
The off-label landscape: longevity clinics, telehealth, compounding
If tesamorelin is FDA-approved only for HIV lipodystrophy, how is it ending up in midlife wellness clinics? Through ordinary off-label prescribing: a licensed clinician can write a prescription for any FDA-approved drug for any indication they judge medically appropriate. Decision and liability sit with the prescriber.
The broader peptide market around tesamorelin has shifted. In late 2023 the FDA placed several growth-hormone-related peptides into Category 2 of its bulk drug substances list — meaning they "may present significant safety risks" and are restricted from compounding. Ipamorelin, GHRP-2, GHRP-6, and ibutamoren landed on that list, while CJC-1295, BPC-157, melanotan II, and others were nominated and withdrawn. PBS NewsHour reported in March 2026 that the recategorization "creat[ed] a gray market of online retailers using research disclaimers while marketing for human consumption." UC Davis cell biologist Paul Knoepfler told PBS that "each of the ten or so most popular peptides remain somewhat of a mystery in terms of their impacts when used in people."
Tesamorelin sits in a different lane because it is FDA-approved as Egrifta — its bulk substance moves through the regulated pharmaceutical supply, and 503A compounding pharmacies can sometimes compound it for an individual patient under prescription. That status is not the same as the "research peptide" market, where vendors sell unlabeled vials of unknown purity that the FDA does not oversee. Innerbody notes that tesamorelin "isn't offered by many leading telehealth platforms" like Hims or Hers; the most reliable route is a local provider working with a specialty pharmacy.
That bottleneck is why longevity clinics and concierge medicine have become the typical access path — clinics that integrate the visit, prescription, and pharmacy relationship to make tesamorelin available to women paying cash for a non-HIV indication. Whether the prescriber is doing a careful baseline workup and ongoing monitoring varies enormously and is the question to ask before paying anything.
Side effects, contraindications, and the cancer question
The side-effect profile is consistent across the FDA label, Mayo Clinic, and Memorial Sloan Kettering. The most common issues are injection-site reactions — bruising, redness, mild pain — along with joint pain, muscle pain, and fluid retention or peripheral edema. Mayo Clinic's drug page notes that the medicine "may cause swelling (fluid retention) in some parts of your body" and warns patients to report new joint pain or numbness in the hands and wrists, which can signal carpal tunnel syndrome.
Glucose effects are a recurring concern. Stanley and colleagues, writing in JAMA in 2014, observed fasting glucose rose by about 9 mg/dL with tesamorelin at two weeks (versus 2 mg/dL placebo) before normalizing over six months. Mayo warns that "tesamorelin injection may cause changes in your blood sugar levels." For a perimenopausal woman already drifting toward insulin resistance, that is not nothing.
Contraindications are short but firm. Tesamorelin should not be used with a pituitary tumor or prior pituitary surgery, allergy to mannitol, or active or prior cancer. Pregnancy is contraindicated because the drug "may harm the fetus." MedlinePlus adds that head trauma, brain radiation, or any pituitary history should be disclosed before starting.
The cancer concern is the most discussed and least settled. Sustained IGF-1 elevation — trial data shows rises of 80 to 100% — is biologically plausible to influence tumor growth, since IGF-1 has trophic effects on many cancer cell lines. Memorial Sloan Kettering states bluntly that "the chance of cancer or tumor growth is raised with this drug." Mayo Clinic similarly tells patients that "using this medicine may increase your risk of getting cancer." The Canadian Common Drug Review noted that one trial reported malignancy at 2.9% with tesamorelin versus 1.5% placebo, while another reported 0.4% versus 3.2% — leaving the cancer signal unresolved. The trial populations are too small and too short to settle a long-term cancer question. A patient with no cancer history may sit comfortably with the data; a patient with prior breast cancer or strong family history is in a different conversation.
Cost reality and what insurance actually covers
Tesamorelin is expensive and the off-label market shoulders the full cost. CADTH's clinical drug review calculated therapy at roughly "$3,085 per box of 60 × 1 mg vials (30-day supply)" or "$37,534 per patient annually." Older U.S. distributor data cited by POZ put wholesale cost at $1,964 for a 30-day Egrifta supply. The working figure most clinics quote in 2026 is $2,000 to $3,500 per month at the cash-pay end.
Insurance covers Egrifta only for the HIV indication, with prior authorization. Off-label use is not covered by commercial insurance, Medicare, or Medicaid in any standard pathway, and manufacturer copay programs are tied to the HIV indication.
The math: a woman paying cash at $2,500 per month spends $30,000 a year on a daily injection that, based on the discontinuation data, has to continue indefinitely to maintain the effect. CADTH's pharmacoeconomic analysis concluded that even within the FDA-approved population, tesamorelin was "more expensive ($611,657 over 30 years) and equally effective" compared with standard care, "primarily because VAT reduction lacks validation as a clinical outcome predictor." VAT reduction is a surrogate marker; there is no published evidence yet that lowering VAT with tesamorelin reduces hard cardiovascular endpoints over decades.
Who is a candidate — and who is being marketed to
The clearest way to think about candidacy is to compare two populations.
The trial population, as summarized in a 2019 review, was "predominantly Caucasian (83%) and male (87%)" with a mean age around 48, all on antiretroviral therapy, all with documented HIV-associated central fat accumulation. About half qualified as "responders" — at least 8% VAT reduction. These are the patients the drug was designed and tested for.
The marketing population in 2026 is largely the opposite: women roughly 38 to 55, premenopausal or perimenopausal, generally healthy, paying cash because they have noticed midsection changes that resist the diet and exercise patterns that worked in their thirties. None of these women appear in the published efficacy data. The mechanism of their belly-fat accumulation — ovarian hormone decline driving visceral redistribution — is not the same as the inflammation-and-antiretroviral-driven mechanism in the original cohort.
That does not automatically mean the drug will fail in this group. The Makimura non-HIV trial suggests the GH-IGF-1 axis can mobilize VAT in obese non-HIV adults, at least in those with documented low GH secretion. It does mean the certainty marketed online — the "18% reduction" claim repurposed for women never in those trials — is not justified by the published record. A reasonable off-label candidate, if one exists, is probably someone with elevated visceral fat documented by imaging, a careful pituitary and cancer history workup, no diabetes, no fertility plans, financial capacity to continue indefinitely, and a clinician willing to monitor IGF-1, glucose, and lipids. The marketed population skews substantially looser than that.
Alternatives that have evidence in women over 40
Several interventions for menopausal visceral fat have been studied in the actual population in question, with effects in the same magnitude as the tesamorelin trials and at far lower cost and risk.
Resistance training is the most direct match. Nilsson and colleagues in Maturitas in 2023 randomized 65 postmenopausal women with vasomotor symptoms to three days per week of supervised resistance training for 15 weeks. Compliant participants showed statistically significant reductions in visceral adipose tissue (P=0.002), abdominal subcutaneous fat (P=0.006), and the VAT-to-total ratio (P<0.001). A 2023 meta-analysis from Khalafi and colleagues in Frontiers in Endocrinology, pooling 101 randomized trials and 5,697 postmenopausal women, found a statistically significant visceral-fat reduction (SMD -0.38; 95% CI -0.62, -0.14; P=0.002), with combined aerobic and resistance training yielding the largest body-composition effects.
For women whose concern is overall weight along with visceral fat, GLP-1 receptor agonists have stronger evidence than tesamorelin. A body-composition substudy of STEP 1 by Wilding and colleagues randomized 140 adults to semaglutide 2.4 mg weekly or placebo for 68 weeks. The semaglutide group lost 15.0% of body weight versus 3.6% in placebo, with a 27.4% reduction in visceral fat mass and a 19.3% reduction in total fat mass. Semaglutide and tirzepatide are not without cost or side effects, but their efficacy data in non-HIV adults is far broader than tesamorelin's.
Sleep, alcohol, dietary protein, and stress all sit upstream of menopausal visceral fat and are better-evidenced than any peptide at the moment. Muscle mass preserved through midlife remains the strongest non-pharmacologic protection against the metabolic consequences of estrogen withdrawal.
Frequently Asked Questions
Is tesamorelin FDA-approved for weight loss in women over 40?
No. Tesamorelin (Egrifta SV, Egrifta WR) is FDA-approved only for the reduction of excess abdominal fat in HIV-infected adults with lipodystrophy. The labeling specifically states it is not for weight loss management. Any use outside that indication, including in perimenopausal women, is off-label.
How much visceral fat would I actually lose if I took it?
HIV trials show roughly 15% VAT reduction at 26 weeks and 17 to 18% at 52 weeks among continuing users. The non-HIV Makimura 2012 trial showed a meaningful but smaller absolute VAT reduction in 60 obese adults with documented low growth hormone secretion. No randomized trial has tested tesamorelin in perimenopausal women, so individual results cannot be predicted from existing data.
Do the results stick after I stop?
No. The 2008 extension data showed visceral fat reaccumulates after tesamorelin is discontinued. Maintaining the effect requires ongoing daily injections.
What about cancer risk?
Tesamorelin raises IGF-1 by 80 to 100%, and IGF-1 has biological links to tumor growth in many cell lines. Mayo Clinic and Memorial Sloan Kettering both warn that the drug may increase cancer risk, and active or prior cancer is a contraindication. The trial-level cancer signal has been inconsistent, with no definitive risk established but also no long-term safety data in non-HIV populations.
Is "research peptide" tesamorelin from an online vendor the same thing as Egrifta?
No. Egrifta SV and Egrifta WR are FDA-approved pharmaceutical products manufactured under regulated conditions. "Research peptides" sold online with disclaimers are not FDA-regulated and are not subject to potency or purity testing. The two should not be treated as interchangeable.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.












