Compliance note: GLP-1 microdosing is off-label and not FDA-approved as a defined regimen. Talk to a qualified prescriber before deviating from labeled dosing. This article is editorial reporting, not medical advice.
A year ago, friends compared notes on Wegovy at full 2.4 mg. Now they whisper about "microdoses" — vials from a telehealth company, a fraction of the strength on the official prescribing label, with a price tag closer to a streaming subscription than a brand-name prescription. Some swear by it. Their endocrinologists are not always swearing along.
Microdosing GLP-1 medications occupies an awkward middle ground in 2026: widely promoted, lightly evidenced, and tangled in a regulatory fight over compounded drugs that the FDA is actively prosecuting. The practice is real. The science behind it is, charitably, immature.
What microdosing actually means (and what it doesn't)
Start with what microdosing isn't: an FDA-approved regimen. The agency has never sanctioned a "microdose" GLP-1 indication — every microdose protocol is off-label by definition. The term came from psychedelics research, where it described amounts so small the compound is "not considered pharmacologically active," per Pharmaceutical Technology. Whether that translates meaningfully to a peptide that lingers in the body for six weeks is a separate question.
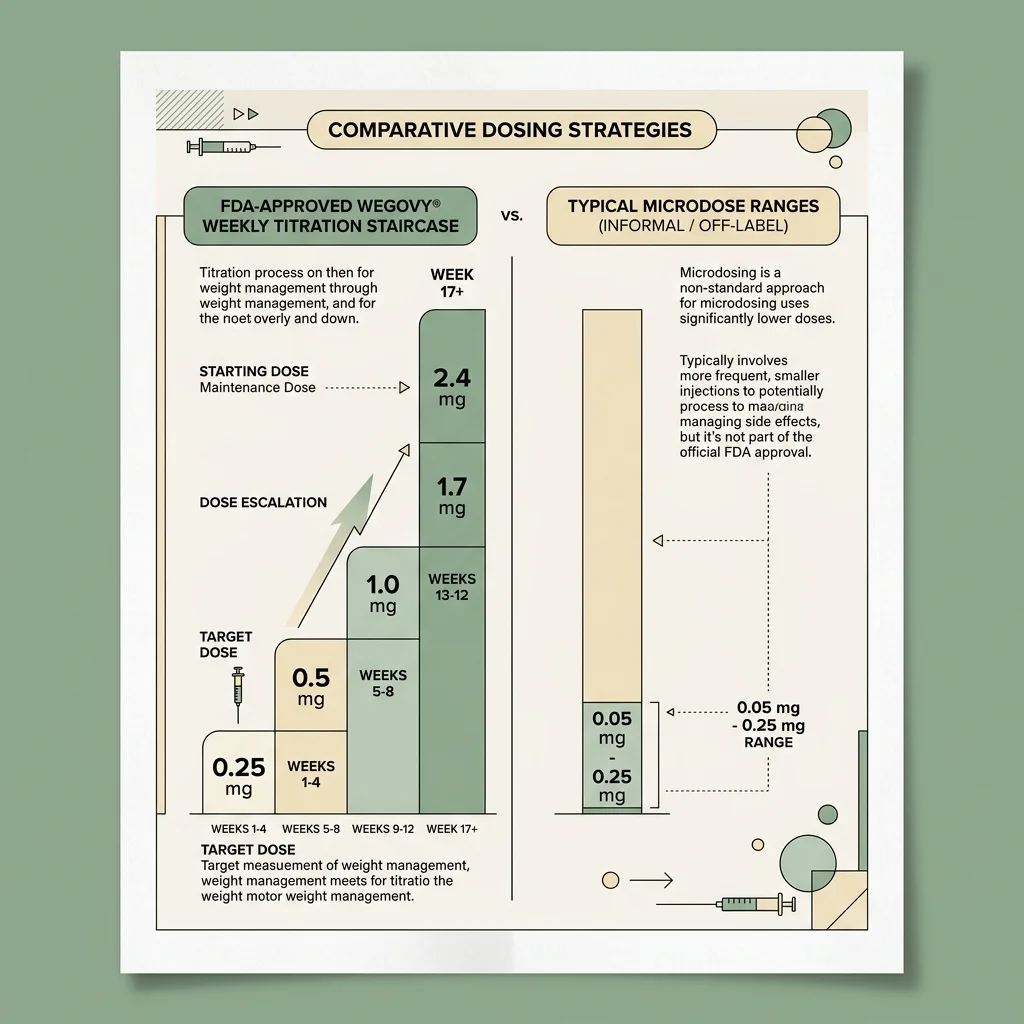
The FDA-approved Wegovy schedule is a staircase: 0.25 mg once weekly to start, escalating every four weeks to 0.5, 1.0, 1.7, and a 2.4 mg maintenance dose, per Novo Nordisk's official pen guide. The 2.4 mg figure was not a marketing decision; it was the dose at which the pivotal weight-loss trials produced their headline results.
Microdosing means something further down the staircase. Noom's program starts patients at 0.2 mg of compounded semaglutide weekly and caps out at 0.6 mg, about a quarter of the standard maximum, according to TODAY. UK clinics go lower: the Healand Clinic in Leicester starts at "a fifth of the normal dose," and the Aestha Clinic in London advertises "about a tenth of standard dosing protocols," The Guardian reported.
Professional medicine has a position. Dr. Scott Isaacs, president of the American Association of Clinical Endocrinology, told Pharmaceutical Technology: "AACE does not support the routine use of microdosing GLP-1RA medications in clinical care and recommends adherence to FDA-approved dosing guidelines." The practical implication is sharper than it first appears: the doses being marketed and the doses studied in clinical trials differ by an order of magnitude.
Why the trend exploded in 2025-2026
Three forces converged. The first was cost. Wegovy carries a list price of roughly $1,349 per month for uninsured patients; Costco offers the brand at $499. Compounded microdose programs run $119 to $199 per month at companies like Noom, with UK clinics charging about £175 per month, according to The Guardian. For someone earning a median income, that gap is the difference between filling a prescription and not.
The second was supply. When Wegovy and Mounjaro spent stretches of 2023 and 2024 on FDA's drug shortage list, compounding pharmacies could legally produce copies. Patients who rationed their vials sometimes discovered that doses spaced two or three weeks apart still suppressed their appetite — the habit stuck even after supply normalized. The shortage formally ended in early 2025: semaglutide left the list on April 24, tirzepatide on March 5, per FDA records. Compounding did not.
The third was social. In October 2024, Stanford neuroscientist Andrew Huberman interviewed performance physician Dr. Craig Koniver, who called microdosing a "gamechanger." The message moved through Andy Cohen's late-night anecdotes, Rebel Wilson's Noom spokesperson contract, and influencer channels promoting "personalized" dosing for perimenopausal women, STAT News documented. By late 2025, an Evidation survey found about 1 in 7 GLP-1 users — roughly 14.6 percent — were microdosing. Platforms vary by country (Noom, Found, Hims & Hers in the U.S.; Voy, Manual, Juniper, Numan, CheqUp in the UK), but the model rhymes: a low-friction online questionnaire and a compounded vial in the mail.
The pharmacology: what we know about smaller doses
Semaglutide is not a quick-acting drug. Its elimination half-life is approximately one week — about 165 hours; bioavailability after subcutaneous injection is 89 percent; more than 99 percent of the drug binds to plasma albumin; and steady-state concentration is reached only by Week 5 of weekly dosing. After the last shot, semaglutide remains detectable for five to seven weeks.
That matters. A weekly injection of any size accumulates. By the time someone has been on a 0.2 mg weekly microdose for a couple of months, the steady-state plasma concentration is the relevant exposure — not the size of any individual injection. The weight-loss response cannot be predicted by linearly scaling down the trial data.
The most-cited published evidence for smaller-than-label dosing comes from a 2025 paper by Dr. Calvin Wu and colleagues in the journal Obesity: a case series of two patients combined with a pharmacokinetic-pharmacodynamic model. The model suggests switching from once-weekly 2.4 mg semaglutide to a dose every 14 days cuts dose frequency in half but maintains 72 percent of the weight loss. Stretching to every 28 days keeps about half the loss with a quarter of the medication.
That research has obvious limits. Two patients are not a clinical trial. The model is a model. The paper itself acknowledges that the strategy lacks "broad clinical validation of long-term efficacy and tolerability" and explicitly calls for randomized trials of every-14-day regimens. Until those exist, the pharmacology argument for microdosing is suggestive — not settled.
The evidence problem
Here is the core tension. Semaglutide and tirzepatide were tested in trials that enrolled tens of thousands of patients, ran for over a year, and produced average placebo-adjusted weight losses of 15 percent and 19 percent of body weight respectively. The Lancet noted in February 2025 that "the practice of microdosing drugs is not new, but there is no evidence that it works for obesity."
The doses being microdosed were never inside any trial. They are, as Yale health services researcher Reshma Ramachandran told STAT News, "essentially patients [as] guinea pigs, both on the efficacy side and the safety side." Dr. Caroline Apovian, who co-directs the Center for Weight Management and Wellness at Brigham and Women's Hospital, was blunter still in a TODAY segment: "There is no evidence that these drugs are effective at that small amount. When you stop these low doses of these drugs, we don't know what's going to happen."
What exists is observational data from the telehealth platforms selling the programs and a single peer-reviewed case series. Noom claims its microdosing patients lose up to 11 pounds in 30 days and 17 pounds in 60 days; that data has not been peer-reviewed. The Wu paper is the only published work that mathematically models reduced dosing for maintenance. Everything else is anecdote, marketing, or extrapolation from full-dose trials. The discipline of separating "what trials showed at 2.4 mg" from "what someone selling 0.2 mg is claiming" is the most useful skill a patient can bring to the conversation.
Where some clinicians cautiously use lower doses
Not every clinician using sub-maximal doses is microdosing. The Wegovy prescribing information itself permits a permanent reduction from 2.4 mg back to 1.7 mg if a patient has "intolerable side effects" at the higher dose, per the official dosing guide. That is label-sanctioned dose personalization, not microdosing — and the two get conflated constantly.
Beyond the label, some endocrinologists and obesity-medicine specialists do prescribe lower-than-typical maintenance doses in specific contexts. Dr. Rekha Kumar, senior medical adviser at Found Health, has pointed to prediabetes, PCOS-associated insulin resistance, and perimenopausal metabolic changes as situations where early intervention "can prevent progression to diabetes."
The most striking real-world cases come from the Wu paper. One patient, a woman in her 50s with type 2 diabetes, dropped from a hemoglobin A1c of 11.1 percent to 5.8 percent and weaned off insulin entirely on intermittent tirzepatide, maintaining a 30 percent body weight reduction. The other, a man in his 30s, kept off 73 pounds on biweekly tirzepatide 7.5 mg. Both made significant lifestyle changes. Both are individual cases.
UK practice tells a similar story. The Healand Clinic in Leicester reports about 750 patients on roughly one-fifth of standard dosing, founder Dr. Omar Babar emphasizing perimenopausal women, The Guardian reported. In Santa Monica, ob-gyn Dr. Shamsah Amersi told Science News that 60 percent of her patients over 40 are on microdoses and report improved labs — though she acknowledges the longevity claims remain unproven. The honest framing is not "this works" but "this is what some clinicians are doing while the trials catch up."

The skeptics' case
The most consequential argument against microdosing is rebound. A 2025 systematic review and meta-analysis in Lancet eClinicalMedicine, pooling 18 randomized controlled trials and 3,771 patients, found that adults with obesity who stopped GLP-1 receptor agonists regained an average of 5.63 kilograms — and the regain was worse with semaglutide (8.21 kg) than with liraglutide (4.29 kg). Systolic blood pressure climbed back up by 7.09 mmHg in the semaglutide group. Hemoglobin A1c, fasting glucose, BMI, and waist circumference all moved in the wrong direction.
The tirzepatide picture is even sharper. A November 2025 JAMA Internal Medicine analysis of 308 patients tracked for 52 weeks after stopping tirzepatide found that 82 percent regained at least 25 percent of their initial weight loss within a year. The accompanying editorial recommended rebranding GLP-1s as "weight management" rather than "weight loss" drugs — a tacit acknowledgment that these are chronic-disease medications more like blood pressure pills than antibiotics.
If full-dose GLP-1s drive substantial regain on cessation, what happens when someone tries to maintain weight loss on a fraction of the dose? Apovian's answer in the TODAY segment was unhedged: "I believe there's a price to pay for microdosing. And that is, when you stop the microdosing, you will gain the weight back." She is not against the drugs (she has received Novo Nordisk grants), but against the proposition that smaller doses preserve outcomes the trials only documented at full doses.
Quality and population concerns layer on top. Cleveland Clinic obesity-medicine specialist Dr. W. Scott Butsch told the clinic's health newsletter: "I don't recommend using compounded versions of these medications because they're untested." Northwell Health bariatric surgeon Dr. Ann Marie Defnet was direct in The Guardian: "If you're going off-label, you're stepping into the unknown." A separate worry from obesity-medicine physician Dr. Levy in TODAY: patients with lower BMIs who qualify for telehealth microdose programs but fall outside FDA thresholds may lose more muscle than fat at sub-therapeutic doses. The skeptics' core argument is not that microdosing is uniformly dangerous; it is that the practice is not what was studied, and patients deserve to know the difference.
Side effects at low dose: smaller, not absent
The central marketing claim is fewer side effects. The pharmacology supports the direction — nausea, diarrhea, vomiting, and constipation tend to be milder at lower exposures, which is why the Wegovy schedule itself starts at 0.25 mg and steps up. But milder is not gone. Dr. McGowan, an obesity-medicine physician, told TODAY: "It's by no means a guarantee that a lower dose translates to lower side effects." Some patients get the full GI profile at any dose.
The under-discussed risk is the workflow itself. Most microdose programs ship compounded vials and require patients to draw small volumes with a syringe — not the metered click of an injector pen. Cleveland Clinic warned that drawing partial doses with syringes can introduce contamination and overdosing risk. Eli Lilly was more pointed in its statement to TODAY: Mounjaro and Zepbound pens "don't contain preservatives and are approved for single-use only," meaning "microdosing of Zepbound and Mounjaro may increase the risk of contamination and therefore pose patient safety risks." The side-effect calculus shifts from one dimension (GI tolerability) to several (sterility, dose accuracy, individual response). Asking how a medication is sourced, prepared, and measured is hygiene, not paranoia.
The compounding connection
None of this would be possible at scale without a regulatory loophole. When Wegovy and Mounjaro were on FDA's drug shortage list, federal law permitted compounding pharmacies to produce essentially identical copies. After both drugs left the shortage list in early 2025, that authority should have ended. It mostly did — with one exception. Section 503A of the Federal Food, Drug, and Cosmetic Act allows compounding for an "identified individual patient" if the prescriber documents the compounded version is meaningfully different from the commercial product. FDA's April 2026 guidance clarified the exception: adding vitamin B12 within 10 percent of commercial dosing won't qualify, but a genuinely different formulation might. Telehealth platforms lean hard on this hook with "personalized" doses, microdose strengths, and combination injections.
The agency has not been silent. On March 3, 2026, FDA issued 30 warning letters to telehealth companies for false or misleading claims about compounded GLP-1 products. Commissioner Marty Makary said in a statement: "It's a new era. We are paying close attention to misleading claims being made by telehealth and pharma companies — and taking swift action." Makary noted the agency had sent thousands of such letters in the prior six months, more than in the entire preceding decade.
Manufacturers have escalated too. Novo Nordisk's official position warns that "compounded samples may contain lower-than-indicated strengths or no semaglutide at all" and often lack Good Manufacturing Practice standards. The company told TODAY: "For Wegovy, only the marked doses on the single-use, fixed-dose pens are approved for use and represent an authentic FDA-approved medicine." Litigation between Novo Nordisk and major telehealth platforms is ongoing.

Questions to ask if a provider suggests microdosing
The value of any consultation is in the specifics. A few questions reliably surface what is and isn't known:
- Why this dose specifically? Not a brochure answer — what is the clinical reasoning for sub-therapeutic dosing in your case versus FDA-approved titration?
- Where is the medication sourced? Is it a 503A pharmacy compounding for an individual prescription, a 503B outsourcing facility, or a commercial pen drawn down? Each carries different oversight.
- What evidence supports the dose I'd be on? Be willing to hear "case series and a model" or "no published evidence." That is often the honest answer.
- What's the maintenance plan if I stop? The Lancet meta-analysis shows rebound is real even from full doses; data on microdose rebound is not yet published.
- Are you affiliated with the company prescribing the medication? Telehealth physicians paid by the platform filling the prescription have a structural conflict; not disqualifying, but worth knowing.
This is the same set of questions any patient should ask about any off-label prescription. Cleveland Clinic's guidance is that dose adjustments — including downward — should happen through a physician familiar with the patient's history, not via a syringe and an internet vial.
Frequently Asked Questions
Is GLP-1 microdosing FDA-approved?
No. There is no FDA-approved microdose indication for any GLP-1 receptor agonist. The approved schedule starts at 0.25 mg weekly for Wegovy and titrates to a 2.4 mg maintenance dose; sub-titration doses and "personalized" microdose protocols are off-label by definition.
How does microdosing differ from dose reduction for tolerability?
The Wegovy label permits a permanent reduction from 2.4 mg back to 1.7 mg if a patient has intolerable side effects. That is FDA-sanctioned dose flexibility within an approved range. Microdosing typically refers to doses below 0.25 mg weekly — far outside any range studied in clinical trials.
Will I keep weight off on a microdose?
No randomized trial has tested it. The 2025 Wu case series in Obesity suggests reduced-frequency dosing can preserve a substantial portion of weight loss in some patients (N=2 plus a model). The 2025 Lancet eClinicalMedicine meta-analysis showed stopping full-dose GLP-1s leads to clinically significant regain — and rebound from microdose protocols has not been systematically studied.
Why do telehealth companies market microdoses?
The FDA's 503A compounding rule allows pharmacies to compound a "personalized" version of a commercial drug for an individual patient, which microdose strengths and combination formulations can satisfy. The cost gap between $1,349/month brand Wegovy and $119-199/month compounded microdose programs creates a substantial market — especially for patients outside FDA-approved BMI thresholds.
What are the main risks specific to microdosing?
Contamination from drawing partial doses with syringes, dose inaccuracy, sterility concerns with multi-use vials of single-use products, regain on stopping, and — for lower-BMI patients — disproportionate muscle loss versus fat loss. Both Eli Lilly and Novo Nordisk have said microdosing their products is not contemplated by the FDA label.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.