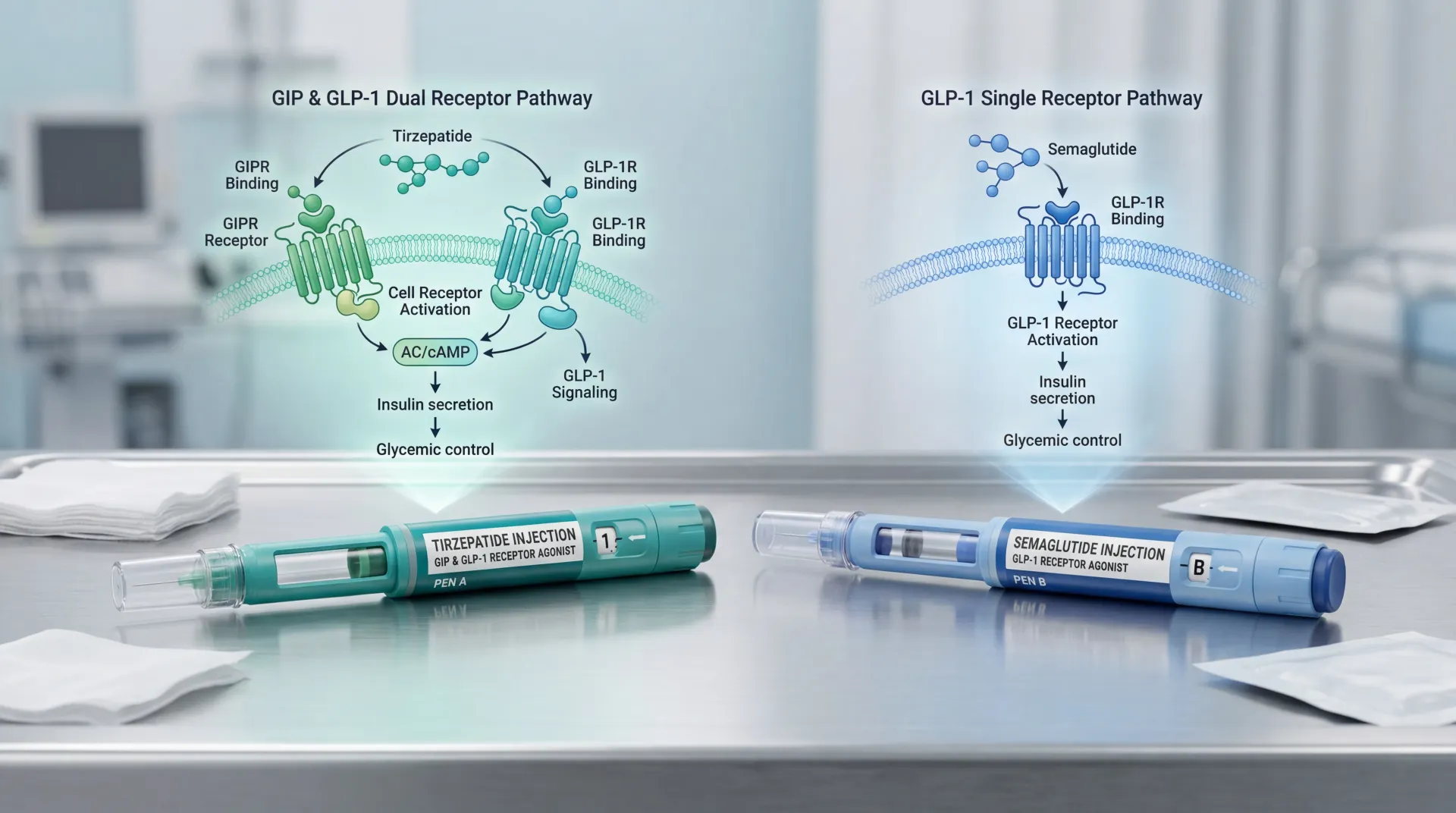
The conversation about GLP-1 medications usually starts and ends with three words: nausea, vomiting, diarrhea. Those are real, but they are also the easy part of the safety profile. The full story includes thinning hair, sulfur-smelling burps, a hollowed-out face, a measurable risk of gallstones, a recent eye-disease signal, and a memo to anesthesiologists telling them to treat your stomach like it just had a Thanksgiving meal. None of this is hidden — it is on labels, in journals, and in society guidance. It just rarely gets fifteen of your fifteen-minute appointment.
This guide pulls those side effects into one place — the documented, the emerging, and the genuinely uncertain — with the data behind each one. The point is not to talk anyone out of a class of drugs that has changed obesity care. It is that informed patients ask better questions, and a treatment that nearly one in eight American adults has now tried deserves more than the standard "you might feel a little queasy" briefing.
Contents
- The side effects everyone already talks about
- Sulfur burps: the rotten-egg problem
- Ozempic face, Ozempic butt, and the fat-distribution problem
- Lean mass loss: when a quarter of the scale drop is muscle
- Gallstones, pancreas signals, and gastroparesis
- Vision changes, mood signals, and the surgery memo
- Hair loss, kidney scares, and the thyroid asterisk
- What this all means if you take or are considering a GLP-1
- Frequently asked questions
- Sources used in this guide
The side effects everyone already talks about
Before drilling into the lesser-known stuff, the gastrointestinal load needs honest framing. Across published trials and real-world clinics, GI adverse events show up in roughly 40-70% of GLP-1-treated patients, and some series put that figure as high as 85%. A 2022 expert-consensus review in the Journal of Clinical Medicine broke it down by symptom: nausea in 15-50% of patients, vomiting in 5-20%, diarrhea in 5-25%, and constipation in 4-12% — with constipation reaching 35% in some obesity-treatment cohorts.
The nuance most patients miss is that those numbers describe how often the symptom appears at any point during titration, not how often it persists. Most cases ease as the body adapts to delayed gastric emptying — a "start low and go slow" approach, smaller meals, and avoiding high-fat foods do most of the work. What the standard pre-prescription chat tends to skip is what happens when your gut keeps slowing for months, what that does upstream and downstream, and what the second-tier safety signals look like once enough patients have been treated long enough to see them. That second tier is what the rest of this guide covers — the well-documented, the regulatory-grade, and the still-emerging.
Sulfur burps: the rotten-egg problem

One of the most-Googled, least-discussed GLP-1 side effects has a deeply unglamorous name. Sulfur burps — eructations that smell like rotten eggs — show up frequently enough on Reddit and TikTok that some pharmacies now flag them in patient handouts. The mechanism is mundane and biological. GLP-1 medications mimic a gut hormone that slows gastric emptying. As Dr. Rebecca F. Dawson at Inspira Bariatrics explained in late 2025, "When food sits in the stomach for an extended period, it can release gases containing sulfur, leading to the distinct sulfur burps some people experience." The smell itself is hydrogen sulfide gas — the same compound that gives volcanic vents and bad eggs their signature odor.
Why it deserves more than a footnote: sulfur burps often arrive together with bloating, nausea, or upper abdominal discomfort, and that cluster signals delayed emptying severe enough to matter clinically. Persistent rotten-egg eructations plus retching or fullness can be candidates for slower titration, dose reduction, or evaluation for gastroparesis. Telling someone to chew Beano misses the signal.
Ozempic face, Ozempic butt, and the fat-distribution problem
"Ozempic face" entered the English language sometime in early 2023, and Google Trends shows queries for the term peaked in April 2024 according to a 2025 systematic review in Aesthetic Surgery Journal Open Forum. The mechanism is not, strictly speaking, a side effect of the drug. It is a side effect of fast weight loss meeting an aging face that no longer remodels skin the way it did at twenty-five.
The same review traced where the volume goes: temporal hollows above the cheekbones, the cheeks themselves, tear troughs, jawline, marionette lines, and nasolabial folds. GLP-1 drugs do not preferentially target facial fat — they accelerate "the age-related gradual decrease in elastin turnover by accentuating sagging skin under a thinner bed of adipose tissue." Patients can end up looking, in the authors' clinical phrasing, "up to 5 years older than peers without such weight changes."
The same redistribution shows up below the neck. Plastic surgeons have started using "Ozempic butt" and "Ozempic arms" for loss of subcutaneous tissue and skin tone in the buttocks and upper arms. In a feature from the American Society of Plastic Surgeons, Dr. John Burns described what he was seeing in clinic: "Everybody's butt is just gone. It's not that it's smaller and tighter; it's that there is a loss of muscle tone and the prevalence of loose skin." That last clause is doing a lot of work. The volume isn't only fat — it's muscle too — which is the bridge to the next side effect, the one that matters more than appearance.
Lean mass loss: when a quarter of the scale drop is muscle

This is the side effect that does not show up on the label but probably matters more than several that do. When weight comes off, some of it is muscle and connective tissue, regardless of method. The question is how much.
A 2025 sub-analysis of body-composition data from two landmark trials, published in SAGE Open Medical Case Reports, put hard numbers on it. In the STEP-1 sub-study, 95 patients on semaglutide lost about 6.9 kilograms of lean soft tissue alongside 10.4 kilograms of fat — roughly 40% of total weight lost was lean tissue. In SURMOUNT-1, 124 patients on tirzepatide lost 5.6 kilograms of lean tissue against 15.9 kilograms of fat, putting the lean share closer to 26%. The literature uses 25% of weight lost as fat-free mass as a rough threshold for "high or low." Both trials sit at or above that line, with semaglutide running notably higher.
The cardiology field has been pushing back on alarm framing. A 2024 review in Circulation by Linge, Birkenfeld, and Neeland argued that, viewed through MRI-based muscle volume z-scores, changes most healthy adults experience on these drugs look "adaptive" — what you would expect for the amount of weight lost — and improved insulin sensitivity and reduced muscle fat infiltration may improve quality even as quantity drops. They added a caveat: "factors such as older age and prefrailty may influence the selection of appropriate candidates for these therapies because of risk for sarcopenia."
Translation: a 38-year-old with strong baseline muscle tolerates this composition shift fine. A 72-year-old who was already losing grip strength does not. Resistance training and adequate protein during a GLP-1 course are not optional accessories — they are the reason your knees still want to climb stairs at the end of the year.
Gallstones, pancreas signals, and gastroparesis
The biliary system is where the data is cleanest. A meta-analysis of 76 randomized trials and 103,371 patients, published in JAMA Internal Medicine in March 2022, found that GLP-1 receptor agonists raised the risk of composite gallbladder or biliary disease by a relative risk of 1.37 (95% CI, 1.23-1.52). The authors translated that into roughly 27 extra gallbladder or biliary events per 10,000 patients per year. The risk wasn't uniform: when the analysis isolated trials at the higher doses prescribed for weight loss, the relative risk jumped to 2.29. Higher dose, longer duration, weight-loss indication — each modifier moved the curve up.
The signal also showed up in a 2022 FAERS case series in the same journal covering 36 postmarketing acute cholecystitis reports. Forty-seven percent of those cases came on within 90 days of starting therapy, 30 patients ultimately required cholecystectomy, and seven also developed pancreatitis (two of those fatal).
Pancreatitis is on the Ozempic label as a serious warning, but the population-level evidence is messier than the warning's prominence suggests. An August 2025 review in the Cleveland Clinic Journal of Medicine walked through it: a Cleveland Clinic retrospective of 161 patients with prior pancreatitis history found only about 10% developed recurrent pancreatitis after starting a GLP-1, and fewer than half of those recurrences were attributable to the drug. A network meta-analysis of more than 102,000 trial participants returned a relative risk of 0.96 — essentially neutral. The reviewers concluded that "denying these valuable therapeutic medications to patients with a history of pancreatitis seems unwarranted," and noted that what risk does exist correlates more with rapid weight loss (over 1.5 kg per week) than with the medication class itself. The label warning still applies; the better mental model is "rapid weight loss in some predisposed people stresses the pancreas, and these drugs cause rapid weight loss."
Gastroparesis and bowel obstruction are the most recent additions. In September 2023, the FDA updated Ozempic's label to include ileus — paralytic intestinal obstruction — after 20 cases turned up in the agency's adverse-event database, including two deaths, against more than 8,500 GI-disorder reports tied to semaglutide. The agency said it could not "reliably estimate frequency or establish a causal relationship," but the report volume was enough to warrant the label change. Severe constipation that is not responding to fluids, fiber, and occasional polyethylene glycol is a phone call, not another bowl of prunes.
Vision changes, mood signals, and the surgery memo
The vision signal is recent enough that most endocrinologists are still figuring out how to talk about it. In July 2024, Hathaway and colleagues at Massachusetts Eye and Ear published a retrospective matched-cohort analysis in JAMA Ophthalmology looking at nonarteritic anterior ischemic optic neuropathy — NAION — in patients prescribed semaglutide. NAION is a sudden interruption of blood flow to the optic nerve, with no proven treatment to reverse the vision loss. Among 710 patients with type 2 diabetes, the hazard ratio for NAION on semaglutide was 4.28 (95% CI, 1.62-11.29) — roughly four times higher than matched controls — with a 36-month cumulative incidence of 8.9% versus 1.8%. In an overweight and obese cohort of 979 patients, the hazard ratio was 7.64 (95% CI, 2.21-26.36), with cumulative incidence of 6.7% versus 0.8%.
The caveats are substantial. The study is observational, single-center, and the absolute case count was small. Subsequent analyses in larger databases have produced mixed results. But the signal was strong enough that ophthalmology editorial boards and the manufacturer's safety information now flag visual disturbances as a reportable symptom. If you start a GLP-1 and notice sudden, painless vision loss in one eye — even a small portion of your visual field — that is an emergency-room call, not a "let's wait until your next appointment."
The mood-and-suicide story has had three acts. After postmarketing reports prompted alarm, the European Medicines Agency opened a formal review in July 2023 covering Ozempic, Wegovy, Saxenda, and related products, triggered by roughly 150 reports of self-injury or suicidal thoughts, according to the EMA's own statement. The agency stressed it was "not yet clear whether the reported cases are linked to the medicines themselves or to the patients' underlying conditions or other factors." Then, in a January 2024 Nature Medicine paper, Wang and colleagues compared semaglutide users with non-GLP-1 users in a cohort of more than 240,000 overweight or obese patients and reported a hazard ratio of 0.27 (95% CI, 0.20-0.36) for incident suicidal ideation — meaning semaglutide was associated with lower risk in their data. On January 13, 2026, the FDA formally requested removal of the suicidal-ideation warning from Saxenda, Wegovy, and Zepbound after a meta-analysis of 91 placebo-controlled trials covering 107,910 patients and a Sentinel cohort study of 2.2 million users found no increased risk of self-harm or related psychiatric events.
The honest reading: the original warning was based on a pharmacovigilance signal, the systematic data has not borne it out, and the FDA acted on that. The honest caveat: regulatory aggregate findings do not preclude individual reactions. If you or someone close to you notices a meaningful change in mood, motivation, or self-talk after starting one of these medications, that conversation belongs in a clinic, not a Reddit thread.
The anesthesia memo is older but consistently underdiscussed. In June 2023, the American Society of Anesthesiologists issued consensus guidance recommending that patients on daily-dose GLP-1s hold their dose on the day of an elective procedure, and patients on weekly dosing hold for a full week prior. The reason is the same delayed-gastric-emptying mechanism that produces sulfur burps: a stomach that has not actually emptied during the standard NPO window can lead to regurgitation and pulmonary aspiration during general anesthesia. The guidance applies regardless of indication, dose, or surgery type. This is the side effect that is not a side effect — a procedural risk that requires telling every dentist, gastroenterologist, and surgeon in your medical history.
Hair loss, kidney scares, and the thyroid asterisk
Hair shedding is documented in the trials, even if "you might lose your hair" is rarely part of the prescribing chat. A 2026 systematic review in Science Progress aggregated alopecia rates across the SURMOUNT and OASIS programs. In tirzepatide arms, alopecia appeared in 5.1-5.7% of participants depending on dose, against 0.9% on placebo. In OASIS 1, oral semaglutide at 50 mg was associated with alopecia in 7% of participants versus 3% in placebo. The dominant subtype is telogen effluvium — diffuse, temporary shedding triggered by physiological stress — which typically shows up 1.5 to 3 months after starting therapy or after a dose escalation. Telogen effluvium reverses once the trigger eases. Adequate protein, iron sufficiency, and patience tend to do the work.
Kidney function is more nuanced. The headline-level evidence is reassuring — large outcome trials show GLP-1s either neutral or beneficial for kidneys, particularly in diabetic kidney disease. The wrinkle is acute kidney injury linked to dehydration during severe GI episodes. A 2021 case series in Kidney Medicine documented two patients with established CKD whose eGFR dropped sharply within four to five months of starting semaglutide and did not recover after stopping. Volume depletion from prolonged nausea and vomiting was the leading suspect, and the authors recommended caution in patients with moderate-to-severe baseline kidney impairment. Aggressive hydration is not optional during the titration phase, and persistent vomiting is a stop-and-call signal, not a symptom to ride out.
Thyroid C-cell tumors carry the most prominent label warning of the entire class — a black-box prohibition for patients with personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2, per Ozempic's official safety information. The warning is rooted in rodent studies in which rats developed thyroid C-cell tumors. Whether that translates to humans has been the subject of a decade of debate. A systematic review aggregating 10 trials and 14,550 participants found four thyroid cancer cases among 7,830 semaglutide-treated patients — under 1%. A French observational study cited within that review (Bezin et al.) reported an adjusted hazard ratio of 1.58 (95% CI, 1.27-1.95) for any thyroid cancer with one to three years of GLP-1 exposure, and 1.78 specifically for medullary cancer — but the European Medicines Agency, after reviewing comparable data in October 2023, stated that "there is no evidence to suggest a causal relationship between GLP-1 receptor agonists...and thyroid cancer."
The honest summary: the population-level human signal is weak, the rodent signal is real, and the contraindication for personal or family history of MTC or MEN-2 is reasonable as a precaution rather than a verdict. If you have an unexamined thyroid nodule and a family history of endocrine cancer, that conversation belongs with an endocrinologist before the prescription is written.
What this all means if you take or are considering a GLP-1
The summary version is not "these drugs are dangerous." It is that the safety profile has more dimensions than a 15-minute consult tends to cover, and the dimensions that matter most depend on who you are. A 35-year-old healthy adult with a family history of gallstones has a different risk calculus than a 70-year-old with sarcopenia concerns and chronic kidney disease, and both differ from a 50-year-old with a family history of medullary thyroid cancer.
The questions worth asking before or during therapy are tedious to type out but useful to actually ask. Is your baseline lean mass adequate, and do you have a resistance training plan? What is your gallbladder history? Have you had any episodes that could plausibly have been pancreatitis? Any first-degree relative with thyroid cancer? What is your kidney function now, and how often will it be checked? Do you have any planned procedures requiring sedation in the next year, and have all those clinicians been told? If you notice sudden vision changes, acute abdominal pain, intractable vomiting, or a meaningful shift in mood, where will you call?
None of those questions are paranoid. They are what a thoughtful endocrinologist asks if there is time on the calendar — and what, in practice, often does not get asked. The hidden side effect of GLP-1 medications might not be on this list at all. It might be the gap between what we now know about these drugs and what fits into the appointment slot you actually got.
Frequently asked questions
Is "Ozempic face" reversible?
Partially. Some volume returns naturally if weight stabilizes or modestly rebounds, particularly in younger patients with better skin elasticity. Beyond that, the systematic review in Aesthetic Surgery Journal Open Forum notes dermatologists and plastic surgeons treat persistent facial hollowing with hyaluronic-acid fillers, fat grafting, or — in larger volume changes — surgical lifts. The underlying skin elasticity loss does not reverse on its own.
Should I stop my GLP-1 before surgery?
For elective procedures, current ASA guidance from June 2023 says yes: hold daily doses on the day of surgery, weekly doses for a full week prior. The reason is delayed gastric emptying and aspiration risk during anesthesia. Tell every clinician who will be sedating you — including your dentist, gastroenterologist, and any procedural specialist — that you are on a GLP-1, regardless of indication or dose.
Are sulfur burps dangerous?
By themselves, no — they are a consequence of the drug slowing your stomach and food fermenting longer than usual. They become a signal worth flagging when they show up alongside persistent nausea, vomiting, severe bloating, or upper abdominal pain. That cluster can indicate gastroparesis severe enough to warrant dose adjustment.
How much muscle will I actually lose?
Trial data suggests roughly 25-40% of total weight lost on a GLP-1 will be fat-free mass, with semaglutide trials showing higher proportions than tirzepatide trials. The amount that translates into measurable strength or functional decline depends heavily on age, baseline muscle mass, protein intake, and resistance training. For most healthy adults under 60 who train and eat enough protein, the functional impact is small. For older adults or those with prefrailty, it is the most important conversation in the room.
Did the FDA really remove the suicidal ideation warning?
Yes. On January 13, 2026, the FDA requested removal of the suicidal behavior and ideation warning from the labels of Saxenda, Wegovy, and Zepbound after a meta-analysis of 91 placebo-controlled trials covering more than 107,000 patients, plus a Sentinel cohort study of 2.2 million users, found no increased risk of self-harm or related psychiatric events compared with controls. Individual reactions vary; meaningful mood changes after starting any medication still warrant a clinical conversation.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.