What does "microdosing" actually mean?
There is no official definition.
The general idea: take somewhere between 5% and 10% of a dose large enough to produce hallucinations. For psilocybin mushrooms, that typically means 0.1 to 0.3 grams of dried material, depending on the species and potency. The goal is not to trip. It is to stay below the threshold of perception while, supposedly, gaining cognitive or emotional benefits. The National Center for Complementary and Integrative Health (NCCIH) describes it as "one-tenth or one-twentieth of a typical nonclinical dose," taken because people believe it will improve mental health, productivity, or pain.
Two popular protocols have emerged, championed by researchers who have become near-cult figures in the microdosing community. James Fadiman's schedule calls for one dose day followed by two rest days. Paul Stamets advocates four days on, three days off, sometimes stacking psilocybin with lion's mane mushroom and niacin. Neither protocol has been validated in a rigorous clinical trial.
When your body processes psilocybin, it converts to psilocin, which binds primarily to serotonin 5-HT2A receptors in the brain. Think of these receptors as volume knobs on the brain's communication system. A full dose cranks them up far enough to scramble the usual patterns of neural chatter, producing hallucinations and ego dissolution. A microdose turns them a fraction of a degree.
The definitional problem matters. When the RAND Corporation surveyed Americans about microdosing, they defined it as "a subperceptual dose much lower than you would take to trip or hallucinate." Even with that guidance, 15% of lifetime psilocybin users could not say whether they had actually microdosed the last time they used.
| Factor | Microdose | Full dose |
|---|---|---|
| Typical amount (dried mushrooms) | 0.1-0.3 grams | 2-5 grams |
| Perceptual changes | None intended | Significant distortion |
| Duration of effects | 4-6 hours (subtle) | 4-8 hours (intense) |
| Frequency | Every 2-4 days, ongoing | Single sessions, months apart |
| Clinical evidence | Early, mostly null or mixed | Strong for depression (with therapy) |
This fuzziness creates a genuine problem for science. Every study measuring microdosing effects is built on participants classifying themselves, and a meaningful chunk of those participants are not sure what category they belong in.

10 million Americans microdosed last year
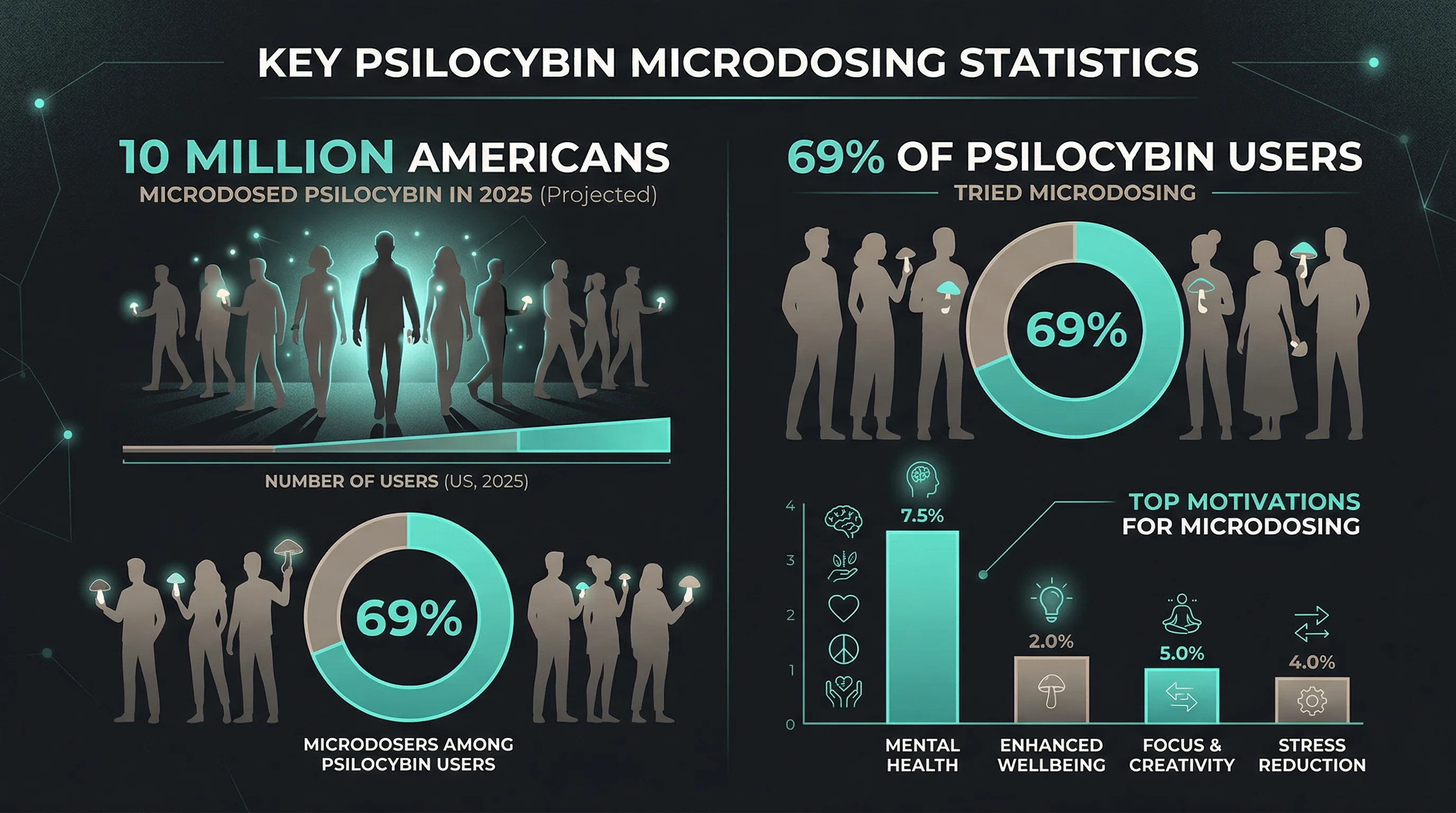
The RAND Corporation published the first nationally representative survey on psychedelic microdosing in January 2026. Based on a probability-based sample of 10,122 adults surveyed in September 2025, approximately 10 million U.S. adults microdosed psilocybin, LSD, or MDMA in the past year. Psilocybin alone accounted for an estimated 11 million past-year users, making it the most popular psychedelic in the country at 4.26% prevalence.
Among those 11 million psilocybin users, 69% reported microdosing at least once. For every person taking a full psychedelic dose, roughly two others are taking tiny ones. The RAND data estimates 216 million psilocybin use-days in the past year, with 47% of those days involving microdoses. That is over 100 million days of Americans taking sub-perceptual amounts of a Schedule I substance.
An earlier survey, led by Michelle Priest and published in 2026 using data from late 2023 and early 2024, estimated at least 8.4 million lifetime psilocybin microdosers. The two numbers are not contradictory (different time frames, slightly different questions) but they tell the same story: millions of Americans are doing this, and the number grew between surveys.
The motivations stand out. Priest's survey found that people who microdosed were 25 percentage points more likely to cite improved mental health as a reason for use and 12 percentage points more likely to cite physical health, compared to those who took full doses. Microdosers were 27 percentage points less likely to say they used psilocybin "for fun or pleasure."
The UK shows a parallel trend. Psilocybin use grew 37.5% in 2024 to reach 1.1% of 16-to-59-year-olds, roughly 300,000 people, nearly matching ecstasy in popularity according to Office for National Statistics data.
| Substance | Past-year US users (est.) | Past-year prevalence |
|---|---|---|
| Psilocybin | 11,059,000 | 4.26% |
| MDMA / MDA | 4,700,000 | 1.81% |
| Amanita muscaria | 3,470,000 | 1.34% |
| Ketamine | 3,270,000 | 1.26% |
| LSD | 2,980,000 | 1.15% |
| DMT | 2,190,000 | 0.84% |
Source: RAND Psychedelics Survey, September 2025 (N = 10,122)
What the data describes is a population self-medicating with an unregulated substance because conventional treatments fell short, or because the promise of a low-risk cognitive upgrade proved irresistible. Whether the science backs them up is a separate matter.
What 733 clinical trial participants revealed about psilocybin
Full-dose psilocybin therapy, where patients take enough to produce a psychedelic experience under professional supervision, has the strongest scientific backing. A 2025 meta-analysis published in JAMA Network Open pooled 12 controlled trials with 733 total participants and found a large effect size for reducing depression symptoms (Hedges g = -0.84). That qualifies as a large treatment effect by standard psychiatric benchmarks.
The meta-analysis also showed that preparatory therapy before the psychedelic session was significantly associated with better outcomes. More prep hours correlated with bigger symptom reductions. The drug matters, but so does the therapeutic scaffolding around it.
The FDA granted Breakthrough Therapy designation to psilocybin-assisted psychotherapy for treatment-resistant depression in 2018 and for major depressive disorder in 2019. That designation falls short of approval but acknowledges enough preliminary evidence to fast-track development.
Most popular coverage blurs this distinction. Nearly all the strong evidence applies to full doses administered with professional therapy. Microdosing is a different practice entirely: a fraction of the dose, no supervision, on a repeating schedule that lasts weeks or months. Whether the same mechanisms work at sub-perceptual levels remains an open question.
Two ongoing trials aim to answer it. A Phase 3 trial is testing psilocybin microdoses of 2-3mg daily, 4 days per week for 2 weeks, for palliative care distress in 120 participants across 7 sites. A Phase II protocol published in 2025 describes a double-blind trial of 2mg psilocybin weekly for 4 weeks versus placebo for major depressive disorder in 40 participants, described by its authors as the first rigorous microdosing-specific clinical trial. Neither has reported results.
Full-dose psilocybin therapy is entering regulatory maturity. Microdosing research is still in pilot stage, running on surveys and lab studies while those clinical trials slowly enroll.
Your brain changes on a microdose. Your mood might not.
A 2025 randomized, double-blind, placebo-controlled trial gave participants either microdoses of psilocybin truffles or an identical placebo over multiple sessions, then measured cognitive performance, emotional processing, and subjective well-being. The result: microdosing did not significantly affect behavioral or subjective measures compared to placebo. Some initial effects on social cognition and mood vanished after statistical correction for multiple comparisons. Participants remained effectively blinded. They genuinely could not tell whether they had gotten the real thing.
A separate 2025 meta-analysis pooled 14 microdosing studies with 1,614 total participants across 59 effect sizes. No cognitive improvements in any domain. The analysis found a significant decrease in cognitive control, which runs counter to the sharpened-focus narrative popular among microdosing advocates. Neither substance type, dosage, nor duration moderated the effects.
That looks like a clean negative verdict. Vince Polito and Paul Liknaitzky of Macquarie University argue it is not. Their rapid review of all 19 placebo-controlled microdosing studies documented real neurobiological changes: altered default mode network activity, increased neural complexity, elevated brain-derived neurotrophic factor (BDNF) levels, and increased pain tolerance. All measured against placebo. All reaching statistical significance.
Picture a medication that measurably changes your blood chemistry and shows up on brain scans, but when you ask patients whether they feel better, the answer is no different from sugar pills. That is roughly where microdosing evidence stands.
Polito and Liknaitzky laid out eight reasons the "just placebo" conclusion is premature: the studies used small samples, tested only a handful of doses, may have used doses that were too low, recruited exclusively healthy volunteers rather than depressed patients, and assessed a narrow window of effects. The measured impact of participant expectations on outcomes was small.
Researchers on both sides agree on one thing: the evidence is not mature enough for a verdict. Brain imaging picks up something real. That something has not translated into measurable cognitive or emotional benefits in any completed study. Whether trials with clinical populations, actual depressed patients rather than healthy undergraduates, will show a different picture is what those two ongoing trials are designed to answer.
The risks that get lost in the hype
Psilocybin itself is difficult to fatally overdose on. The lethal dose is more than 500 times the therapeutic dose, and NIDA describes psilocybin as having "a low level of toxicity." That safety margin is real.
The non-fatal harms get less airtime.
The NCCIH lists microdosing-specific side effects: insomnia, increased anxiety and depression, poor mood, low energy, gastrointestinal symptoms, headache, disrupted senses, temperature dysfunction, poor focus, and impaired social skills. That is the federal government's assessment based on published research, not anecdotal forum posts.
Some people should avoid psilocybin entirely, at any dose. The NCCIH states it is "not safe for people with psychotic conditions like schizophrenia, schizoaffective disorder, or severe forms of bipolar disorder and borderline personality disorder."
The cardiac question. Psilocybin activates 5-HT2B serotonin receptors, and chronic use of 5-HT2B agonists, even at low doses, may cause cardiac fibrosis and valvulopathy, according to researchers cited by Polito and Liknaitzky. The concern draws from the pharmacology of other drugs targeting the same receptor, including fenfluramine, the weight-loss drug pulled from the market for heart valve damage. Nobody has studied whether regular psilocybin microdosing carries this risk. The concern is theoretical but pharmacologically grounded, and it applies specifically to the chronic dosing pattern microdosing requires.
The unregulated supply chain creates its own hazards. A multistate investigation beginning in 2023 found over 180 cases across 34 states of hospitalizations linked to Diamond Shruumz brand mushroom edibles. Lab testing of similar "nootropic" gummies revealed undeclared psilocybin mixed with caffeine, ephedrine, and kratom compounds, none listed on the label. The FDA issued a warning in late 2024 that Amanita mushrooms do not meet the GRAS (Generally Recognized As Safe) standard, though products containing them remain on shelves.
Longer-term psychological effects remain poorly studied. One survey cited by The Guardian found that 8.9% of lifetime psychedelic users reported impairments lasting longer than one day, including hallucinogen persisting perception disorder (HPPD), anxiety, depersonalization, and trauma.
No published study has tracked the long-term safety of chronic microdosing in humans. For a practice that 10 million Americans did last year on a repeating schedule, that gap in the evidence deserves more weight than it usually receives.

From Schedule I to legal service centers
Psilocybin is a Schedule I controlled substance under federal law, classified as having "no currently accepted medical use and a high potential for abuse." The DEA's classification has not changed since 1970, despite the FDA's Breakthrough Therapy designations pointing in the opposite direction.
Oregon became the first state with a regulated therapeutic psilocybin program when voters approved Measure 109 in November 2020. The program, run by Oregon Psilocybin Services under the Oregon Health Authority, issued its first licenses in May 2023. Clients must be 21 or older, need no prescription, and must consume psilocybin on-site at a licensed service center under the supervision of a facilitator who has completed a state-approved training program, passed a licensing exam, and cleared a background check.
Colorado followed in November 2022 with Proposition 122, decriminalizing psilocybin and creating a Regulated Natural Medicine Access Program. Colorado tasked the Department of Regulatory Agencies with establishing the framework, and the first healing centers were expected to begin operating in 2024.
| State | Action | Year | Model |
|---|---|---|---|
| Oregon | Measure 109 | 2020 | Regulated therapeutic access (service centers + facilitators) |
| Colorado | Proposition 122 | 2022 | Decriminalization + regulated natural medicine program |
| Washington | SB 5263 | 2023 | University-based pilot program (PTSD, mood, SUD) |
| 24+ states | Various bills | 2023 | Decriminalization, research funding, therapeutic programs |
Sources: ASTHO, UC Berkeley Center for the Science of Psychedelics
Washington enacted SB 5263 in 2023, creating a psilocybin therapy pilot at the University of Washington for people 21 and older with PTSD, mood disorders, or substance use disorders. At least 24 state legislatures considered psilocybin-related bills during their 2023 sessions, ranging from decriminalization to research funding to full therapeutic programs.
Oregon's and Colorado's programs are built around supervised, full-dose sessions. Neither state's regulatory framework specifically addresses or authorizes home-based microdosing on a repeating schedule, which is what those 10 million Americans are actually doing. The legal structures and the reality of use are on parallel tracks that have not converged.
Frequently asked questions
How much psilocybin counts as a microdose?
No universal standard exists. The NIDA and most researchers describe a microdose as 5-10% of a dose that would produce psychedelic effects. For dried psilocybin mushrooms, that is roughly 0.1 to 0.3 grams, though potency varies by species, growing conditions, and batch. The RAND survey defined it as "a subperceptual dose much lower than you would take to trip or hallucinate."
Can microdosing cause a bad trip?
A true microdose, by definition, should not produce perceptual effects. But individual sensitivity varies widely, unregulated products have inconsistent potency, and what someone believes is a microdose may actually be a low-to-moderate dose. The NCCIH lists increased anxiety and depression as potential microdosing side effects. Experts interviewed by The Guardian warn that people taking psilocybin outside clinical settings can experience HPPD, anxiety, trauma, and depersonalization.
Is microdosing legal anywhere in the United States?
Psilocybin remains illegal at the federal level as a Schedule I substance. Oregon and Colorado have created supervised therapeutic programs, but these are designed for on-site, facilitator-guided sessions, not home microdosing. Some cities have deprioritized enforcement of psilocybin possession laws, but deprioritization is not legalization.
Does microdosing work for depression?
The evidence so far is inconclusive. The most rigorous double-blind trial found no significant benefit over placebo, and a meta-analysis of 14 studies with 1,614 participants found no cognitive improvements. However, neuroimaging studies show real brain changes, and two clinical trials testing microdosing specifically for depression are underway. Full-dose psilocybin therapy for depression has strong evidence from 12 controlled clinical trials, but that involves a completely different dosing approach with professional oversight.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.