SURMOUNT-5: The Head-to-Head Trial That Changed the Conversation
For three years the comparison was indirect. Tirzepatide had its trials, semaglutide had its trials, and clinicians stitched the numbers together with caveats — different baseline weights, different placebos, different time horizons. Then in May 2025, Aronne and colleagues published SURMOUNT-5 in the New England Journal of Medicine, and the guesswork ended.
SURMOUNT-5 was a phase 3b open-label trial that randomized 751 adults with obesity but no type 2 diabetes 1:1 to maximum tolerated doses of tirzepatide (10 or 15 mg) or semaglutide (1.7 or 2.4 mg) for 72 weeks. It is the first and only head-to-head randomized trial directly comparing the two drugs in an obesity-only population. The primary endpoint was percent change in body weight at week 72.
The result was decisive. Tirzepatide produced a mean body-weight reduction of 20.2 percent (95% CI -21.4 to -19.1) versus 13.7 percent for semaglutide (95% CI -14.9 to -12.6), with a P-value below 0.001. Translated into pounds, the tirzepatide group lost about 50 pounds on average; the semaglutide group, about 33. Waist circumference followed the same pattern — tirzepatide -18.4 cm versus semaglutide -13.0 cm.
The number most clinicians remember is the upper-tier weight loss. Thirty-two percent of tirzepatide users hit at least 25 percent body-weight loss, versus 16 percent on semaglutide. That doubling matters because 25-percent loss is the threshold where many obesity-related complications start measurably reversing: blood pressure normalizes, sleep apnea remits, joint pain eases. SURMOUNT-5 is the first trial to show that one weekly injection can put roughly a third of users in that range.
The trial wasn't perfect. It ran open-label — participants saw which auto-injector they were holding, and a "I got the newer one" placebo effect probably nudged adherence and self-reported satiety. Eli Lilly funded the study and manufactures tirzepatide; Dr. Aronne disclosed paid advisory work for both Lilly and Novo Nordisk. Industry funding doesn't invalidate good methodology, but it does earn a closer read of the protocol. The headline number still stands. It just needs replication in independent hands.
For the patient sitting across from a prescriber, the takeaway is that on average tirzepatide produces about 1.5 times the weight loss of semaglutide over 72 weeks. That is the new baseline. Average and best-for-you are not the same thing, and the gap between them is what the rest of this article is about.
Single Receptor or Twincretin? How They Actually Work
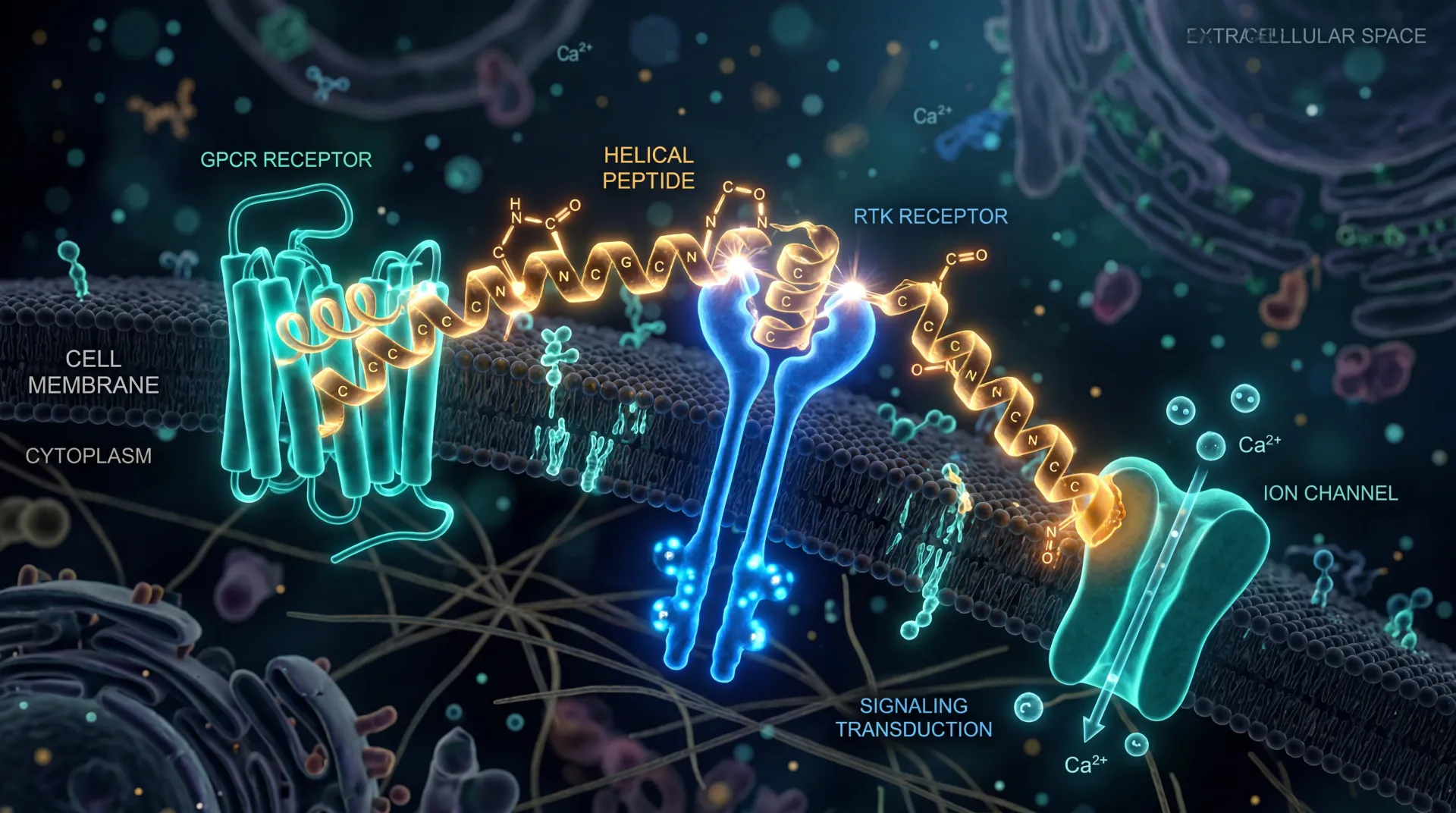
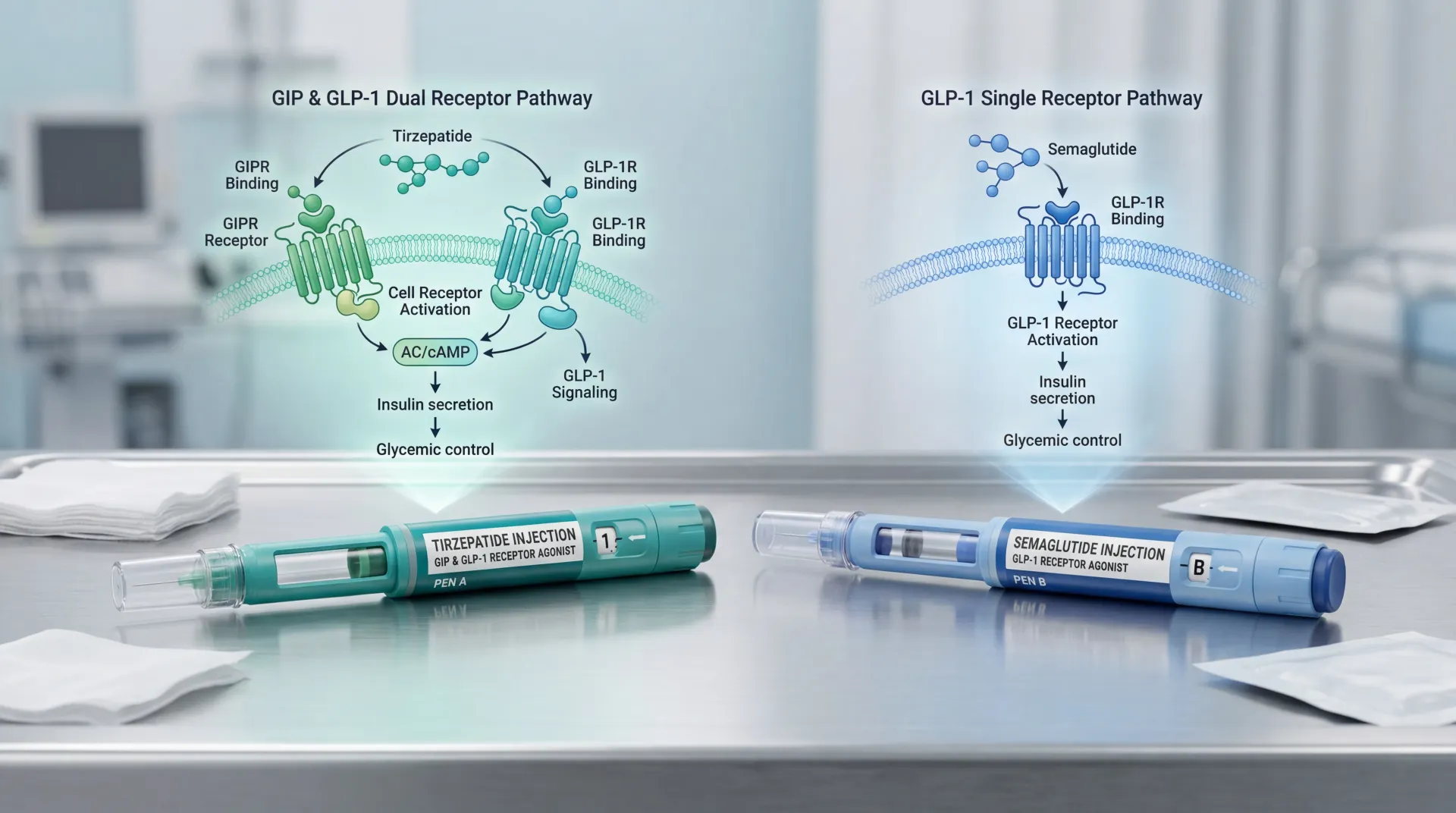
Both drugs are gut-hormone mimics. The body releases glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) after a meal. They tell the pancreas to release insulin, slow how fast the stomach empties, and signal the brain that it has had enough. Semaglutide and tirzepatide both copy these signals — but they do it differently.
Semaglutide is a selective GLP-1 receptor agonist. Think of it as a key cut to fit one lock — the GLP-1 receptor — but cut very precisely. Tirzepatide is a 39-amino-acid synthetic peptide engineered from the human GIP molecule, modified to bind both the GIP and GLP-1 receptors. Researchers nicknamed it the "twincretin." If semaglutide is one key in one lock, tirzepatide is one key cut to open two different doors at once.
The receptor affinities are not symmetric. Tirzepatide's affinity for the GIP receptor matches that of native GIP, but its affinity for the GLP-1 receptor is roughly 18 to 20 times weaker than native GLP-1. That looks like a flaw — and yet the body responds as if it isn't. The paradox points to something interesting about how the drug behaves at the cellular level.
When most GLP-1 agonists bind their receptor, they trigger two downstream pathways: the cAMP pathway (which improves insulin secretion and signals satiety) and the β-arrestin pathway (which pulls the receptor inside the cell and degrades it). The β-arrestin path is essentially a self-limiter that shuts the signal off. Tirzepatide shows "biased agonism toward the cAMP pathway", meaning it activates the productive signal without pulling the off-switch as hard. The receptor stays on the cell surface longer. The signal lasts.
The GIP arm adds a second mechanism. Hindbrain GIP receptor agonism, particularly in the area postrema (the brain region that drives nausea), can attenuate the aversive signaling triggered by GLP-1 receptor activation. Practically, the GIP component may dampen the GLP-1 component's worst side effect. Both drugs cause nausea, but tirzepatide may produce less of it dose-for-dose than its raw potency would predict.
Both molecules also share a fatty-acid side chain (a C20 diacid for tirzepatide) that binds to plasma albumin. Albumin acts like a slow-release reservoir. The result is a half-life around five to seven days, which is why both drugs are dosed once a week. Tirzepatide's half-life is approximately 5 days, supporting weekly subcutaneous dosing; semaglutide's prescribing label puts its half-life close to 7 days.
The dual mechanism is not a marketing tagline. The extra weight loss in SURMOUNT-5 is plausibly explained by the GIP signal acting on adipocytes (which carry GIP receptors but not GLP-1 receptors) and by the biased GLP-1 signaling that keeps the satiety message active for longer. Whether your body sees that benefit depends on individual variation in receptor density and signaling. Even a superior average still leaves a meaningful subset of patients who respond better to semaglutide.
Numbers Side by Side: Pivotal Trials and Real-World Data
SURMOUNT-5 is the cleanest comparison, but it is not the only data. The two drugs each have their own pivotal trials, and both have been studied in large real-world cohorts that look at how they perform outside the controlled environment of a phase 3 study. Three things stand out when you line them up.
The pivotal placebo-controlled trials produced ceiling numbers that look closer than they are. STEP 1 was the trial that put semaglutide on the obesity map: Wilding and colleagues randomized 1,961 adults to once-weekly 2.4 mg semaglutide or placebo for 68 weeks, finding mean weight loss of -14.9 percent versus -2.4 percent — a 12.4-percentage-point difference. Half of the semaglutide arm — 50.5 percent — reached at least 15 percent loss. SURMOUNT-1 did the same job for tirzepatide a year later: Jastreboff and colleagues randomized 2,539 adults across four arms, with the 15 mg group losing 20.9 percent of body weight versus 3.1 percent on placebo over 72 weeks. Fifty-seven percent of the 15 mg arm reached at least 20 percent loss; 91 percent reached at least 5 percent.
The real-world picture sharpens the gap. A propensity-score-matched cohort study published in JAMA Internal Medicine looked at 18,386 adults from electronic health records — not a controlled trial, but a real population of patients filling prescriptions. Tirzepatide users were 3.24 times as likely as semaglutide users to reach at least 15 percent body weight loss (HR 3.24; 95% CI 2.91-3.61). At 12 months, mean weight change was -15.3 percent on tirzepatide versus -8.3 percent on semaglutide. 42.3 percent of tirzepatide users hit ≥15 percent loss versus 18.1 percent on semaglutide.
The body composition data deserves its own moment. In the SURMOUNT-1 DXA substudy, tirzepatide produced a 33.9 percent reduction in fat mass versus 8.2 percent on placebo, with a 10.9 percent loss of lean mass versus 2.6 percent. The fat-to-lean loss ratio was roughly 3:1 — similar to what bariatric surgery produces, and similar to what carefully managed lifestyle weight loss can produce. Visceral fat — the metabolically dangerous type that wraps around organs — dropped 40.1 percent on tirzepatide versus 7.3 percent on placebo. That visceral effect is plausibly the pathway through which both drugs reduce cardiovascular events, normalize blood pressure, and improve fatty-liver markers.
The headline-versus-real-world gap is almost certainly about adherence. In the JAMA cohort, 55.9 percent of tirzepatide users and 52.5 percent of semaglutide users discontinued treatment — a far higher rate than the controlled trials, which used careful titration and full insurance coverage. People who stay on the drug get the trial-like results. People who can't tolerate it, can't afford it, or can't refill it get something less. We will return to that discontinuation problem in the section on stopping.

Dosing, Titration, and the Practical Weekly Routine
Both drugs follow a slow climb. The ceiling-dose magic only works if you reach the ceiling, and you only reach the ceiling if your gut tolerates each step on the way up. The titration schedules are similar enough to seem identical, but the differences shape who succeeds.
Tirzepatide for chronic weight management (Zepbound) starts at 2.5 mg subcutaneously once weekly, with the dose increased by 2.5 mg every 4 weeks as tolerated, to a maximum of 15 mg weekly. The available strengths step in 2.5 mg increments — 2.5, 5, 7.5, 10, 12.5, and 15 mg — which gives prescribers room to pause, slow, or even reverse a step if a patient struggles. If a dose is missed, the next can be given within 96 hours; otherwise the missed dose is skipped and the patient resumes the regular weekly schedule.
Semaglutide for chronic weight management (Wegovy) follows a five-step titration over 16 weeks: 0.25 mg, 0.5 mg, 1 mg, 1.7 mg, then 2.4 mg, with each step held for at least four weeks before escalating. The lower starting dose (0.25 mg versus 2.5 mg, in different milligram units that are not directly comparable) reflects semaglutide's higher per-milligram potency at the GLP-1 receptor. SURMOUNT-5 used the maximum tolerated dose of either drug — meaning some participants stayed on the second-highest dose if the top step caused unmanageable side effects. For semaglutide, that meant 1.7 or 2.4 mg; for tirzepatide, 10 or 15 mg.
The injection itself is mechanically simple. Both come in single-use, pre-filled auto-injector pens. The patient pinches a fold of skin on the abdomen, thigh, or upper arm, presses the pen against the skin, holds for the click-and-count, and discards. Most people learn the routine in one office visit. The harder part is the timing — keeping the same day of the week, leaving room to titrate up if a step felt too aggressive, and (for tirzepatide users) remembering that the 96-hour grace window does not extend forever.
One practical wrinkle worth flagging: tirzepatide delays gastric emptying enough that it can reduce the absorption of oral medications, including hormonal contraceptives. Patients on oral birth control are advised to use a non-oral or barrier method during initiation and for four weeks after each dose escalation. Semaglutide carries a similar but less aggressively worded warning. If you are on a thyroid medication, antibiotic, or any drug with a narrow therapeutic window, the timing of the oral dose may need to shift.
For the prescriber, the practical decision often comes down to side-effect predictability. Patients who tolerated metformin, who have eaten through a stomach bug without vomiting, who do not have severe baseline reflux — these patients can usually push through the titration in either direction. Patients with histories of gastroparesis, severe IBS, or active gallbladder disease may need to start lower, climb slower, and accept a lower ceiling.
Side Effects, Safety, and the Tolerability Trade-off
The two drugs share most of their side-effect profile. Both are dominated by gastrointestinal complaints — nausea, vomiting, diarrhea, constipation — and both can trigger gallbladder events, pancreatitis flares, and (in animal studies) thyroid C-cell tumors. The profiles diverge on raw frequency and on a handful of biliary outcomes that only became clear with the larger 2025 datasets.
The largest comparative safety analysis to date is a 2024 systematic review and meta-analysis covering 13 randomized controlled trials and 26,894 obese adults without diabetes. Tirzepatide raised the relative risk of any GI adverse event to 2.94 versus placebo; semaglutide raised it to 1.68. In absolute terms, 79.8 percent of tirzepatide-treated participants reported at least one GI event versus 25 percent of controls; for semaglutide, 30.81 percent versus 12.7 percent. The authors attributed part of the gap to higher tirzepatide doses (10-15 mg) versus semaglutide (2.4 mg) — the comparison is between maximums, not equivalent potencies.
The trial-level numbers tell a more nuanced story. In SURMOUNT-1, treatment discontinuation due to adverse events was 4.3 percent on tirzepatide 5 mg, 7.1 percent on 10 mg, 6.2 percent on 15 mg, and 2.6 percent on placebo. STEP 1 reported GI-related discontinuation of 4.5 percent on semaglutide versus 0.8 percent on placebo. Both numbers are far lower than what the meta-analysis implies because most GI events are mild and self-limiting — they show up in adverse event tallies without driving people to quit.
The biliary picture flipped in 2025. The 2024 meta-analysis had found semaglutide significantly elevated cholelithiasis risk by 159 percent (RR 2.59), while tirzepatide showed no statistically significant biliary risk increase. A retrospective cohort study presented by Rodrigues and colleagues at the 2025 American College of Gastroenterology meeting confirmed and extended the finding: tirzepatide users had relative risks of 0.58 for cholelithiasis, 0.54 for cholecystitis, 0.47 for choledocholithiasis, and 0.57 for other gallbladder complications, all relative to semaglutide users. The conclusion was unambiguous: "Tirzepatide had a better biliary safety profile compared to semaglutide."
Why the difference? Rapid weight loss itself raises gallstone risk by changing bile composition — and tirzepatide produces faster early loss. So a smaller per-pound biliary effect on tirzepatide is only visible because the dual-agonist's other mechanisms appear to be partially protective. The exact pathway is still being worked out.
Two safety items apply to both drugs equally and deserve mentioning. Both carry a boxed warning for medullary thyroid carcinoma based on rodent data, and both are contraindicated in patients with personal or family history of MTC or multiple endocrine neoplasia syndrome type 2. Both have been linked to acute pancreatitis flares in patients with prior pancreatic disease. And both can cause significant lean mass loss alongside fat loss — in the SURMOUNT-1 DXA substudy, lean mass dropped 10.9 percent on tirzepatide versus 2.6 percent on placebo. Resistance training and adequate protein intake (≥1.2 g/kg body weight) during treatment are increasingly considered standard of care to protect muscle.
Practically, this means that the drug with the higher gastrointestinal frequency may end up with the better long-term tolerability for some patients — because the biliary risk that drives surgical intervention is lower on tirzepatide, even if the day-to-day nausea is a closer match than the meta-analysis suggests.

Cost, Insurance, and the End of the Compounding Workaround
The clinical comparison ends in tirzepatide's favor more often than not. The economic comparison is messier, and for many patients it is the only comparison that actually shapes the decision.
For most of 2022 to early 2025, both drugs sat on the FDA's national drug shortage list. That listing carved out a regulatory carve-out: under sections 503A and 503B of the Federal Food, Drug, and Cosmetic Act, compounding pharmacies and outsourcing facilities were permitted to produce copies of drugs in short supply. A parallel market grew up — telemedicine clinics, men's-health platforms, and wellness pharmacies offering compounded versions for $150 to $300 a month, sometimes less. For many patients without insurance coverage, that was the only way the math worked.
The carve-out closed in 2025. The FDA declared tirzepatide off the shortage list in December 2024 and semaglutide in February 2025. Compounding pharmacies operating under Section 503A had to stop producing copies of tirzepatide by March 19, 2025, and semaglutide by April 22, 2025. Outsourcing facilities under 503B faced parallel deadlines: tirzepatide by March 19 and semaglutide by May 22, 2025. Compounders may still produce versions only when they can demonstrate a documented clinical difference — for example, an orally disintegrating tablet for a needle-phobic patient, or a formulation that excludes a specific allergen.
That left brand-name pricing as the floor for most patients. Both manufacturers responded by launching direct-to-consumer cash-pay programs at substantially reduced rates compared to the published wholesale price. List prices for monthly supplies of either branded drug have historically run in the $1,000 to $1,400 range without insurance; the cash-pay programs from Eli Lilly (LillyDirect for Zepbound) and Novo Nordisk (NovoCare for Wegovy) have brought specific dose vials down to several hundred dollars per month, with the exact figure depending on dose strength and pharmacy. Insurance coverage remains uneven — many commercial plans cover only when prescribed for type 2 diabetes (Mounjaro, Ozempic) and not for weight management (Zepbound, Wegovy). Medicare Part D historically excluded weight-loss drugs entirely, though policy is in flux.
The cost squeeze shows up directly in adherence numbers. The AARP's January 2026 reporting noted that high cost, insurance challenges, and side effects together drive most discontinuations; University of Alabama-Birmingham researchers cited approximate discontinuation rates of 50 percent within one year and 70 percent within two years across both drugs. The clinical superiority of tirzepatide does not help the patient who runs out of refills.
The cheapest version of either drug is the one you can keep affording. For some patients, semaglutide on a stable insurance formulary will produce more total weight loss over five years than tirzepatide they can only stay on for nine months.
The practical implication is that price-shopping is now a clinical decision, not a financial side conversation. Before settling on a drug, the patient and prescriber should know: what is the formulary tier on the patient's insurance, what is the manufacturer's direct-to-consumer rate for the specific dose, and what dose strength is realistic to maintain long-term. The "best" drug is the one the patient can stay on past the year-two cliff.

Who Each One Is For — and What Happens When You Stop
The honest answer to "which one is right for me?" is that the matrix has more variables than just efficacy. SURMOUNT-5 puts tirzepatide ahead on raw weight loss, but several other factors swing the recommendation.
Tirzepatide tends to be the cleaner choice for patients who: want maximal weight loss, do not have a history of severe gastrointestinal disease, can tolerate a slow titration, have insurance that covers Zepbound or can afford the LillyDirect cash-pay rate, and have no contraindications (MTC family history, MEN-2, severe gastroparesis). It carries additional FDA approvals beyond weight management — type 2 diabetes (2022) and moderate-to-severe obstructive sleep apnea in adults with obesity (2024) — which can simplify access for patients with those comorbidities.
Semaglutide remains the better choice for patients who: have established cardiovascular disease (Wegovy carries a 2024 expanded indication for reducing major adverse cardiovascular events in adults with obesity and existing CVD), have a longer track record of tolerating semaglutide for diabetes management already, have insurance coverage that favors Wegovy or Ozempic on the formulary, or who want the more extensive long-term safety data that a longer time on market provides. The cardiovascular signal in real-world data is also genuinely mixed: a Cleveland Clinic real-world cohort of nearly 31,000 patients with MASLD, obesity, and type 2 diabetes presented at ACG 2025 found tirzepatide associated with lower MACE, mortality, and heart failure rates, while a separate STEER analysis presented at ESC 2025 favored semaglutide for cardiovascular event reduction in different populations. The dust has not settled.
The hardest variable, and the one most under-discussed in clinic, is what happens when treatment stops. In the SURMOUNT-1 long-term extension, weight regain occurred in all tirzepatide arms during a 17-week off-treatment period. A November 2025 JAMA Internal Medicine trial followed 308 tirzepatide users through 36 weeks of treatment, then randomized them to placebo for 52 weeks; 82 percent of those who stopped regained at least 25 percent of their original weight loss within a year. STEP 1 follow-up data showed semaglutide stoppers regained roughly two-thirds of the weight they had lost within 12 months of discontinuation. A 2026 University of Oxford analysis put the average return to pre-treatment weight at about 1.7 years after stopping.
That last finding may be the most important number in this whole article. Both drugs treat obesity the way blood-pressure drugs treat hypertension — they suppress the underlying biology while present, and the underlying biology returns when they leave. The accompanying editorial to the November 2025 JAMA report argued for renaming the class from "weight loss" to "weight management" drugs, on the grounds that some patients will need to take them indefinitely. That is the framing question the patient should be answering before starting either: am I willing to take a weekly injection — and pay for it — for the foreseeable future?
If you can commit to indefinite use, tirzepatide will on average deliver more weight loss than semaglutide. If you can't, either drug will work well while you take it and lose most of its effect when you stop. The right comparison isn't six-month efficacy. It's whether the regimen you choose is one you can sustain for years.
None of this is medical advice. The choice between tirzepatide and semaglutide is a clinical decision that depends on your medical history, current medications, insurance coverage, and treatment goals. Talk to a clinician — ideally one with experience in obesity medicine — before starting, switching, or stopping either drug. The data in this article is current as of April 2026 and will continue to evolve as the SURPASS-CVOT, SURMOUNT-MMO, and ongoing real-world cohorts mature.

Frequently Asked Questions
Is tirzepatide always better than semaglutide for weight loss?
On average, yes. In the head-to-head SURMOUNT-5 trial, tirzepatide produced 20.2 percent body weight loss versus 13.7 percent for semaglutide over 72 weeks. But "average" hides individual variation — some patients respond better to semaglutide, some cannot tolerate tirzepatide's higher GI burden at top doses, and some have insurance or cardiovascular history that points back to semaglutide. The trial puts the population mean ahead for tirzepatide; it does not pick the right drug for any specific person.
Will I regain weight if I stop either drug?
Almost certainly some. Trial data shows roughly two-thirds of weight loss returns within 12 months of stopping semaglutide, and 82 percent of tirzepatide stoppers regain at least 25 percent of their original loss within a year. University of Oxford modeling puts the average return to pre-treatment weight at about 1.7 years. These drugs work as long as you take them. Some clinicians are exploring spaced-out maintenance dosing (every other week instead of weekly) to preserve a portion of the loss; preliminary data suggests this can hold roughly 75 percent of the gains, but the protocol is not yet standard practice.
Can I still get compounded tirzepatide or semaglutide cheaply?
Mostly no. After the FDA declared both drugs off the shortage list in late 2024 and early 2025, compounding pharmacies under Section 503A had to stop producing copies by March 19, 2025 (tirzepatide) and April 22, 2025 (semaglutide). Compounded versions remain legal only when they can demonstrate a documented clinical difference — for example, an orally disintegrating tablet, an allergen-free excipient, or a vitamin-enhanced formulation that addresses a specific patient need. Cash-pay programs from the brand manufacturers (LillyDirect, NovoCare) have replaced most of the compounded market for patients without insurance coverage.
How fast do these drugs work?
The titration is slow on purpose. Both drugs start at low doses for tolerability and step up every four weeks. Tirzepatide can take 16 to 20 weeks to reach the maximum 15 mg dose, and semaglutide takes 16 weeks to reach 2.4 mg. Most patients see the steepest weight loss between months three and twelve, with the curve flattening as the body finds a new set point. The 72-week pivotal trials chose that duration because it represents the time horizon over which the effect plateaus — earlier numbers underestimate, and longer numbers show modest additional loss before the curve goes flat.
What if I have a history of gallbladder disease?
Bring it up before starting. Both drugs can trigger gallstone formation, partly because rapid weight loss itself changes bile composition. A 2024 meta-analysis found semaglutide raised cholelithiasis risk 159 percent (RR 2.59) versus placebo, while tirzepatide showed no statistically significant biliary risk increase. A 2025 ACG real-world study found tirzepatide users had roughly half the relative risk of cholelithiasis, cholecystitis, and choledocholithiasis compared to semaglutide users. If you have prior gallstones or a history of biliary surgery, tirzepatide may be the safer choice — but slow titration and close monitoring matter regardless.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.