A pill that lifts growth hormone to the level of a 25-year-old, taken once a day, no injections, no doctor — that's the pitch in every bodybuilding forum and longevity podcast about MK-677. The pitch is half right. The drug does raise GH and IGF-1. It has been studied in humans for nearly 30 years across more than half a dozen indications. It has also never been approved for a single one. The 2024 FDA Pharmacy Compounding Advisory Committee that reviewed it recommended against the compounding bulks list. A Phase IIb hip-fracture trial stopped early when 6.5% of patients on the drug developed congestive heart failure. That's not the version you read on r/SARMs. The honest answer about ibutamoren sits between the bodybuilder hype and the dismissive footnote: it does what its mechanism predicts, and almost none of that translates into outcomes humans actually want.
What MK-677 Actually Is (And Isn't)
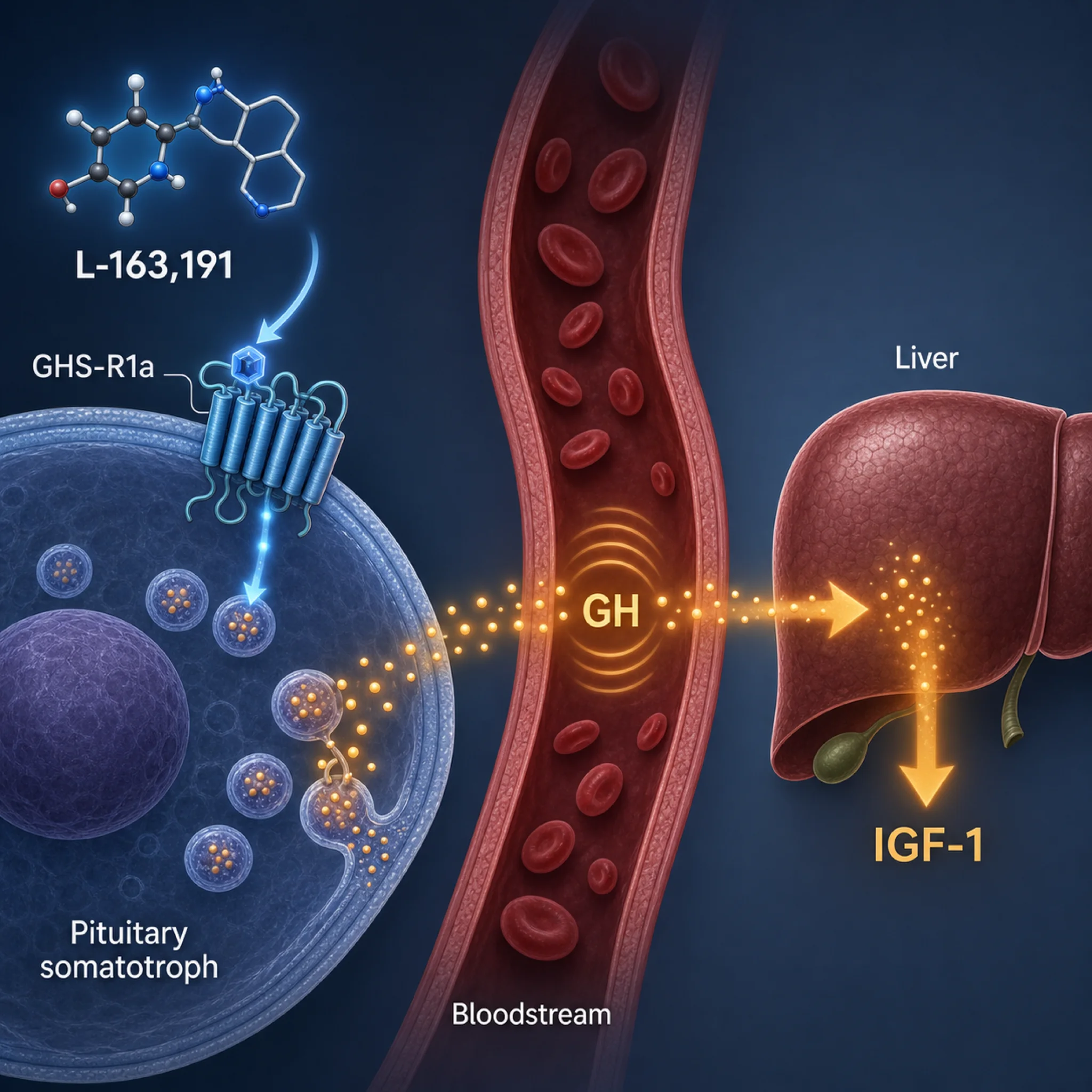
MK-677 is not a peptide. That single fact untangles most of the confusion. The compound — also written ibutamoren, ibutamoren mesylate, MK-0677, MK0677, and the more recent development name LUM-201 — is a non-peptide spiropiperidine, a small molecule synthesized in 1995 by a team led by Arthur Patchett at Merck under the internal code L-163,191. The Patchett 1995 paper in Proceedings of the National Academy of Sciences introduced it as a potent, orally active growth hormone secretagogue. It binds the ghrelin receptor (GHS-R1a) on somatotroph cells of the anterior pituitary and tells them to release more of the GH the body already makes.
Think of GH release as a thermostat. Somatotroph cells fire in pulses, mostly during slow-wave sleep, governed by GHRH (the upstream signal that turns the dial up) and somatostatin (the brake). Ghrelin — the gut peptide most know as the hunger hormone — also activates that thermostat through the GHS-R1a switch. MK-677 mimics ghrelin's action on that switch but does it orally and lasts long enough to dose once a day.
That mechanism is what supplement vendors describe. The December 2025 FDA warning letter to Musclepower Enterprise quotes one product page: "MK 677 IBUTAMOREN acts as an orally active GH secretagogue. The selective growth hormone doesn't release cortisol, the stres [sic] hormone." Half of that is correct. The cortisol claim is the part the studies disagree with.
What MK-677 is not: a SARM. The U.S. Department of Defense's Operation Supplement Safety bulletin makes that explicit: "products that contain MK-677 are often combined with SARMs (or even indicate MK-677 as a SARM). SARMs are also illegal." Selective androgen receptor modulators bind androgen receptors. MK-677 binds the ghrelin receptor. The shared regulatory status — both unapproved, both gray-market — is what created the muddle. Sport Integrity Australia summarizes the legal situation in five words: "Ibutamoren is technically considered a poison."
The Merck Years: 30 Years of Clinical Trials
The reason MK-677 has more human safety data than newer peptides like BPC-157 or epitalon is that Merck spent the late 1990s and early 2000s running real Phase II and III trials across six different indications. The FDA Pharmacy Compounding Advisory Committee briefing from October 2024 lists them: growth hormone deficiency, osteoporosis, hip fracture, sarcopenia, obesity, and Alzheimer's disease. Six conditions. Zero approvals.
The first published proof of concept came from Murphy et al. in 1998: an 8-subject, 7-day study where MK-677 reversed diet-induced nitrogen wasting in healthy males. Svensson et al. (1998) randomized 24 obese males for 8 weeks. IGF-1 rose roughly 40%, fat-free mass increased, and total body fat and visceral fat did not change. The drug grew lean tissue without burning fat tissue — a detail that matters when the bodybuilder claims arrive later.
Osteoporosis came next. Murphy et al. (2001) randomized 292 postmenopausal women to alendronate alone, MK-677 alone, the combination, or placebo for 18 months. Femoral-neck bone mineral density rose 4.2% in the combo arm versus 2.5% with alendronate alone. The win was real but limited: the same enhancement did not appear at lumbar spine, total hip, or total body, and MK-677 alone did not significantly increase BMD at any site versus placebo.
Hip fracture was where the story turned. Bach et al. (2004) enrolled 161 hip-fracture patients aged 65 and older, randomized to 25 mg MK-0677 (n=84) or placebo (n=77) for six months. IGF-1 rose 84% in the treatment arm versus 17% in placebo — a textbook biomarker response. Functional performance measures showed numerical advantages but none reached significance. The authors concluded plainly that "although MK-0677 treatment increased serum IGF-I, it is uncertain whether clinically significant effects on physical function were achieved."
The Alzheimer's program ran in parallel. Sevigny et al. (2008) in Neurology randomized 563 patients with mild-to-moderate AD to 25 mg/day MK-677 or placebo for 12 months. The mechanism was the IGF-1 hypothesis: in animals, IGF-1 helps clear beta-amyloid from the CNS. MK-677 produced exactly the IGF-1 response predicted — 60.1% above baseline at 6 weeks, 72.9% at 12 months. Cognitive endpoints showed no difference at any timepoint. The authors concluded that "the human growth hormone secretagogue MK-677 25 mg was ineffective at slowing the rate of progression of Alzheimer disease."
The most consequential trial was the last. Adunsky et al. (2011) was a multicenter Phase IIb hip-fracture trial in patients aged 60+, 62 on MK-0677 versus 61 on placebo, planned for 24 weeks. The trial did not finish. The data monitoring committee stopped enrollment early on a safety signal: 4 patients (6.5%) on MK-0677 developed congestive heart failure compared with 1 (1.7%) on placebo. The authors' published conclusion: "the AEs associated with MK-677 in a relatively small patient population makes it likely that the risk benefit of this drug for this indication is not acceptable." After 2011, Merck quietly let the program lapse. A 2011 University of Virginia trial of MK-0677 in chronic kidney disease was withdrawn before enrollment with a single explanation: "Could not obtain drug supply from manufacturer."
The compound has been tested more rigorously in humans than almost any peptide currently sold gray-market. The reason it's still gray-market — instead of FDA-approved — is the same data showing it works well biochemically and badly clinically.
GH and IGF-1 Numbers: What the Data Shows
The clearest dataset is the Nass et al. 2008 trial in Annals of Internal Medicine, run at the University of Virginia by a team led by Michael O. Thorner. Sixty-five healthy adults aged 60 to 81 were randomized 2:1 to 25 mg MK-677 or placebo daily for one year, with a crossover year following.
The biomarker results were unambiguous. Daily MK-677 raised 24-hour mean GH and IGF-1 to levels seen in healthy young adults. UVA's press release described it as restoring "20 percent of muscle mass loss associated with normal aging." Fat-free mass increased 1.1 kg in the MK-677 group (95% CI 0.7 to 1.5) and decreased 0.5 kg in placebo (P less than 0.001). Body weight rose 2.7 kg on the drug versus 0.8 kg on placebo (P=0.003). The drug did exactly what its mechanism predicted.
The catches in that same study rarely make it into bodybuilder summaries. Fasting blood glucose increased 0.3 mmol/L (about 5 mg/dL) in the MK-677 group (P=0.015), and insulin sensitivity declined. Cortisol rose 47 nmol/L (95% CI 28 to 71), a statistically significant elevation that contradicts the "selective release without cortisol" claim repeated on supplement labels. The fat-free mass gain produced no measurable improvement in strength or function. Limb fat increased 1.1 kg on MK-677 versus 0.24 kg on placebo, so part of the body weight gain was tissue you wouldn't post on Instagram.
Animal data adds one more wrinkle. Lee et al. (2018) in the Yonsei Medical Journal dosed rats with 4 mg/kg MK-677 orally for six weeks. A single dose increased peak GH 1.8-fold over baseline. Six weeks of continuous dosing abolished the GH response entirely, with a marked rise in hypothalamic somatostatin mRNA and protein. The brake on GH release tightened so hard that the secretagogue stopped being able to push past it. This is the tachyphylaxis problem, and whether it happens in humans on continuous dosing is — as the FDA briefing notes — not well characterized.
An IGF-1 number 60-90% above baseline at week 6 doesn't tell you the drug will still be working at week 24, and the GH and IGF-1 numbers don't reliably predict the strength or body composition gains a user actually wants.
Muscle, Bone, and the Elderly: Where Promise Met Plateau
The clearest target population for MK-677 — the one Merck designed it for — was older adults losing muscle and bone with age. Sarcopenia and osteoporosis drive frailty, falls, and loss of independence. A drug that restores young-adult GH levels without injections looked, on paper, like a near-perfect intervention.
The body composition signal is genuine but narrow. The Nass 2008 trial demonstrated that 12 months prevents the age-related decline in fat-free mass that placebo subjects show. Researchers framed it as "proof of concept" because the muscle gain was real but did not translate into measurable strength or function.
Hip-fracture recovery is where the most plausible indication failed twice. Bach 2004 found no significant differences in functional performance measures despite a near-doubling of IGF-1. Adunsky 2011 was halted early on the heart-failure imbalance described above. Two well-designed trials in the population most likely to benefit, both negative, with the second negative on safety.
The sleep data is the more honest find. A small trial of 14 subjects given 25 mg/day at bedtime showed REM sleep increases of 20% in young subjects and 50% in older subjects, with roughly a 50% increase in slow-wave sleep in young subjects. The effect lines up with what a ghrelin mimetic would predict. If there is a real benefit to MK-677 for which the human data is genuinely supportive, this is probably it. The benefit is also smaller in clinical importance than the marketing copy suggests, and achievable through cheaper, safer means.
For an older adult considering MK-677 to "stay strong as I age," the practical reading is that the drug raises a biomarker without changing things that matter in life. A doctor recommending it for sarcopenia is not following the published evidence.
Side Effects: Edema, Insulin Resistance, Heart Failure
The safety profile is unusually well documented for an unapproved drug because Merck collected adverse event data systematically. The October 2024 FDA briefing compiles these into one place.
The most common effects in elderly subjects on 25 mg/day for 12-24 months: increased appetite affected 67% versus 36% on placebo (P=0.033). Mild transient lower-extremity edema affected 44% versus 27%. Transient muscle pain affected 33% versus 9%. The hunger surge is the side effect bodybuilders cite most often as the reason they cycle off — it is genuinely substantial, and a portion of the body-weight gain in clinical trials is fat tissue accumulated through caloric overshoot.
Metabolic effects matter more for long-term users. Nass 2008 documented a 5 mg/dL average rise in fasting glucose with significantly decreased insulin sensitivity over 12 months. Bach 2004 in hip-fracture patients showed increases in serum glucose, insulin, and HbA1c with more reports of edema and fluid overload (n=16) compared with placebo (n=10). The Svensson 1998 obesity trial found impaired glucose homeostasis at 2 and 8 weeks, with one subject reaching 180 mg/dL fasting glucose at 6 weeks before spontaneously resolving. The DEA's 2025 consumer bulletin specifically cites insulin resistance and the long-term risk of type 2 diabetes from chronic IGF-1 elevation.
The cardiac signal is the one the FDA flagged hardest. The PCAC briefing summarizes serious adverse events seen across the Merck program: "CHF, thrombosis, cancer, and myocardial infarction." The trial-stopper was Adunsky 2011's 6.5% versus 1.7% CHF imbalance. Healthymale, an Australian government-funded men's health body, summarizes the situation in eleven words: "Researchers stopped at least one clinical trial of MK-677 early because of concerns that it might cause heart failure." When that same site is asked "Is MK-677 worth taking?", the answer is two sentences: "No. There are no evidence of its benefits and plenty of harmful side effects."
The hunger and edema you'll notice. The 5 mg/dL rise in fasting glucose, the prolactin shift, the cardiac signal — those are not things you feel until they're not minor anymore.
Bodybuilder Claims vs. What the Studies Found
Four claims drive most MK-677 sales. Each survives only partially against the trials.
"It builds muscle." True at the level of fat-free mass — the Nass 2008 trial added 1.1 kg over 12 months in older adults. False at the level of strength: the same trial measured isokinetic strength as an end point and found no significant change. Bach 2004 in hip-fracture patients found no significant difference in any functional performance measure despite an 84% IGF-1 increase. The fat-free mass added by MK-677 includes intracellular water and limb fat alongside contractile tissue. None of the published clinical trials demonstrate that MK-677 increases strength a lifter would care about.
"It burns fat." The Svensson 1998 obesity trial in 24 men is the most direct test: 8 weeks of 25 mg/day in obese subjects. IGF-1 rose 40%, fat-free mass increased, and total body fat and visceral fat did not change. Nass 2008 in elderly subjects showed no change in abdominal visceral fat or total fat mass with limb fat actually increasing 1.1 kg versus 0.24 kg in placebo. The drug is not a lipolytic agent, and the hunger increase makes weight gain — including fat gain — more likely than fat loss.
"It doesn't raise cortisol." The studies directly contradict this. Nass 2008 documented cortisol increases of 47 nmol/L. The FDA's December 2025 warning letter to Monster King / GE Labs cites the Copinschi 1996 study (PMID 8768828) showing the drug's effects on adrenocortical function. The "doesn't raise cortisol" claim originates from comparisons against older injectable GHRP peptides where cortisol release is dramatic. MK-677's elevation is smaller, but smaller-than-the-worst-comparator is not the same as no-effect.
"It's safer than HGH because it uses your own pituitary." The pulsatile-versus-continuous distinction matters for receptor desensitization but does not exempt MK-677 from the metabolic and proliferative consequences of sustained IGF-1 elevation. The FDA briefing notes that adverse events known to be associated with recombinant human GH — hyperglycemia, fluid retention, glucose intolerance, increased risk of neoplasm — were also reported with ibutamoren in clinical studies.
The sleep claim is the one that holds up best. The 14-subject sleep architecture data is consistent with ghrelin signaling biology. If a user reports better sleep, it's not placebo. It's also achievable through cheaper, safer means than an unapproved gray-market drug with the side effect profile above.
Regulatory Status and Off-Label Sourcing
The legal status of MK-677 in 2026 is as clear as the clinical status. The Department of Defense's Operation Supplement Safety bulletin states: "It is not approved for human use, which makes it an unapproved drug. It is also not legal for use as an ingredient in dietary supplements or any other consumer or commercial products. It can only be used for research purposes." MK-677 is on the DoD Prohibited Dietary Supplement Ingredients List and the WADA Prohibited List. The DEA's Get Smart About Drugs program summarizes: "any use of MK-677 is currently illegal and banned in all legitimate forms of competition."
The vendor labeling problem is what makes the gray market work. The FDA's December 2025 warning letter to SARMS AMERICA illustrates the playbook. The company sold "MK-677 Ibutamoren" alongside other compounds with "For Research Use Only" labels. The FDA disagreed: "Despite statements on your product labeling marketing your products, '[F]or research use only,' and '[F]or research purposes only,' evidence obtained from your product labeling, including your website establishes that your products are intended to be drugs for human use."
The legal mechanism for excluding MK-677 from dietary supplements is laid out in the parallel December 2025 letter to Monster King / GE Labs: ibutamoren is excluded from the dietary supplement definition under section 201(ff)(3)(B)(ii) of the FD&C Act because it was authorized for investigation as a new drug (the original Merck IND), substantial clinical investigations were instituted, those investigations were made public, and the substance was not marketed as a dietary supplement before that authorization. The "supplement" framing is closed off by statute.
The October 2024 PCAC evaluation sealed the compounding pathway too. A nominator had asked FDA to add ibutamoren mesylate to the 503A Bulks List. The PCAC reviewer recommended against inclusion: "the physicochemical characterization, limited information on historical use, lack of evidence of effectiveness, and the specific safety concerns identified for ibutamoren mesylate weigh against inclusion of this substance on the 503A Bulks List."
The current development pipeline is narrow. Lumos Pharma has been advancing the molecule as LUM-201 for pediatric growth hormone deficiency; the Phase 2 OraGrowtH210 and OraGrowtH212 trials reported meeting their primary and secondary endpoints. That is the one indication where a tightly controlled use case — children with diagnosed GHD, supervised by pediatric endocrinologists — could plausibly clear the regulatory bar. None of that work supports the way the molecule is currently sold to adults online.
For a buyer thinking about ordering "research-grade MK-677" from a peptide vendor, the practical situation is unambiguous. There is no manufacturing oversight, no quality control on dose or purity, no insurance coverage if something goes wrong, no medical supervision, and explicit federal and international prohibitions on use. Healthline's coverage of FDA enforcement notes that the agency conducts physical raids and confiscations on bulk vendors. The vendor you ordered from last cycle may not exist next cycle.
Frequently Asked Questions
Is MK-677 a peptide?
No. Despite the marketing, MK-677 is a non-peptide spiropiperidine — a small molecule designed by Merck in 1995 (originally L-163,191). It mimics ghrelin's action at the GHS-R1a receptor, but the molecule itself is structurally unrelated to ghrelin or any peptide hormone. The defining practical difference is route: peptides like CJC-1295 and ipamorelin require subcutaneous injection because gut enzymes destroy peptide bonds. MK-677 is orally active and lasts long enough to dose once a day.
How much does MK-677 actually raise IGF-1?
Across the trials, 25 mg/day produces increases in serum IGF-1 ranging from roughly 40% (Svensson 1998 obese males, 8 weeks) to 60.1% at 6 weeks and 72.9% at 12 months (Sevigny 2008 Alzheimer's), to 84% in hip-fracture patients (Bach 2004). The Nass 2008 elderly cohort reached IGF-1 levels comparable to those of healthy young adults. The biomarker response is one of the most replicable findings in the entire MK-677 literature — and one of the least useful in isolation, because it does not predict the strength, function, or clinical-outcome improvements users want.
Why was the Adunsky 2011 hip-fracture trial stopped early?
The independent data monitoring committee halted enrollment because of a congestive heart failure signal: 4 of 62 patients (6.5%) on MK-0677 developed CHF compared with 1 of 61 (1.7%) on placebo. The trial authors called the risk-benefit profile "not acceptable" for hip fracture and described MK-0677 as having "an unfavorable safety profile" in that population. This is the single largest negative safety event in the MK-677 trial record and is one of the reasons the FDA's 2024 PCAC review specifically cited "serious safety concerns" including CHF.
Is MK-677 legal to buy in the United States?
No. MK-677 is an unapproved investigational drug. It cannot legally be sold as a dietary supplement (excluded by section 201(ff)(3)(B)(ii) of the FD&C Act because it was authorized for clinical investigation as a new drug), it cannot be compounded under 503A (the FDA's October 2024 PCAC recommended against bulk-list inclusion), and it has never been approved by FDA for any indication. Vendors selling it with "For Research Use Only" labeling have been targeted by FDA warning letters, including December 2025 actions against SARMS AMERICA, Monster King / GE Labs, and Prime Sports Nutrition.
If MK-677 raises GH, why doesn't it work for sarcopenia or hip fracture?
This is the central puzzle of the MK-677 program. The drug reliably increases GH and IGF-1, and reliably increases fat-free mass in healthy older adults, but the clinical endpoints — muscle strength, functional performance, ability to live independently, hip-fracture recovery — failed to follow in trial after trial. The fat-free mass added includes intracellular water and limb fat alongside contractile tissue, the cortisol and metabolic costs may offset some anabolic gain, and the rat data from Lee et al. 2018 raises the possibility of receptor desensitization through hypothalamic somatostatin upregulation over weeks of continuous use.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.












