A bright blue liquid that stains everything it touches, methylene blue has been quietly sitting in hospital pharmacies for over a century. It treats a rare blood disorder. Surgeons use it to mark tissue during operations. It kills parasites in fish tanks. And now biohackers are dropping it into their morning water, claiming it sharpens memory, protects the brain from aging, and supercharges mitochondria.
The gap between what methylene blue can do in a controlled clinical setting and what people are doing with it at home is wide enough to be concerning. This is a compound with genuine neuroscience behind it, studied in Alzheimer's models, traumatic brain injury, and stroke. But genuine neuroscience and a safe supplement are not the same thing. Here is what the evidence actually supports, where it falls short, and what the risks look like if you decide to experiment on yourself.
A 150-year journey from fabric dye to the WHO essential medicines list
Heinrich Caro synthesized methylene blue in 1876 for the German textile industry. It was supposed to color cloth. Within 15 years, it was coloring something else entirely: the microscope slides of Paul Ehrlich, who noticed this dye had an unusual affinity for living nerve tissue. He injected it into rats and described it as a "magic bullet" because of how selectively it accumulated in active neurons.
Ehrlich's observations led to a cascade of medical firsts. In 1891, methylene blue became the first fully synthetic compound used as a therapeutic drug in humans, treating malaria at a time when the only alternative was quinine bark. Military doctors used it extensively, though soldiers were not thrilled about the side effect of bright blue urine. That same blue urine, however, became useful in psychiatric hospitals: mixed into medications, it served as a visible indicator of whether patients were actually swallowing their pills.
Through that psychiatric application, clinicians stumbled onto something unexpected. Methylene blue appeared to have antipsychotic properties on its own. This observation became the starting point for developing phenothiazine drugs, which dominated psychiatric treatment for decades. Chlorpromazine, the drug that emptied mental asylums in the 1950s, traces its chemical lineage directly back to this textile dye.
Methylene blue holds a singular distinction in pharmacology: it is both the oldest synthetic drug still in clinical use and the chemical ancestor of modern psychiatric medication.
Today, its only FDA-approved indication is treating methemoglobinemia, a condition where hemoglobin loses its ability to carry oxygen effectively. But the World Health Organization lists methylene blue as an essential medicine, and physicians use it off-label for vasoplegic shock after cardiac surgery, ifosfamide-induced brain toxicity from chemotherapy, and cyanide poisoning.
The mitochondrial shortcut that makes researchers pay attention
Your mitochondria produce energy through a chain of protein complexes embedded in their inner membranes. Electrons pass from complex I through complexes II, III, and IV in sequence, and each handoff releases energy that drives ATP production. When any link in this chain breaks down, two bad things happen simultaneously: the cell runs low on energy, and leaked electrons generate free radicals that damage surrounding structures.
Methylene blue does something no other approved drug can do. It accepts electrons from NADH at complex I and delivers them directly to cytochrome c, essentially skipping complexes I through III entirely. This bypass has two consequences. First, complex IV (cytochrome oxidase) gets more electrons to work with, so ATP production increases. Second, fewer electrons leak out of the chain at the points where free radicals are typically generated.
| Mitochondrial effect | Mechanism | Practical implication |
|---|---|---|
| Electron rerouting | Bypasses complexes I-III, delivers electrons to cytochrome c | Maintains ATP production even when parts of the chain are damaged |
| Reduced oxidative stress | Fewer electrons leak at complexes I and III | Less free radical damage to mitochondrial components |
| Complex IV upregulation | Increases cytochrome oxidase activity and expression | Long-term improvement in respiratory capacity |
| Auto-oxidation cycling | Alternates between oxidized (blue) and reduced (colorless) forms | Acts as renewable antioxidant rather than single-use scavenger |
The auto-oxidation property deserves emphasis. Most antioxidants, like vitamin C or vitamin E, donate an electron and are spent. Methylene blue cycles between its oxidized form (blue) and its reduced form, leucomethylene blue (colorless), continuously. This makes it what some researchers call a "renewable electron cycler" rather than a consumable antioxidant. In laboratory conditions, this cycling capacity is among the most potent chain-breaking antioxidant effects ever measured.
The compound is also remarkably brain-penetrant. After intravenous administration, brain tissue concentrations reach roughly 20 times plasma levels within about an hour. Oral bioavailability ranges from 53% to 97%, with plasma levels peaking in 30 to 60 minutes and a half-life of approximately 5 to 6 hours.
None of this is disputed. The mitochondrial mechanism is well-characterized and reproducible. The question is whether these cellular effects translate into meaningful cognitive benefits in living humans, and that is where the picture gets more complicated. If you are interested in other compounds that target mitochondrial function, NAD+ and NMN supplements work through a different pathway that also supports cellular energy production.
Memory, connectivity, and the mixed results in human brains
Animal studies on methylene blue and cognition are extensive and largely positive. Memory-enhancing effects in rodents were first described more than 30 years ago, and they have been replicated in spatial memory, fear extinction, habituation, and object recognition tasks. Rats given low-dose methylene blue after learning consistently show better retention when tested later.
The human data is thinner. The most rigorous human neuroimaging study to date used a randomized, double-blinded, placebo-controlled design with 28 healthy adults at the University of Texas Health Science Center. Participants received either 280 mg of pharmaceutical-grade methylene blue or a placebo, then underwent functional MRI scanning. The results were mixed in a telling way.
Methylene blue strengthened resting-state functional connectivity in brain regions linking perception and memory functions. The drug appeared to enhance communication between brain areas that collaborate during recall and learning. At the same time, it reduced cerebral blood flow during a motor task. The researchers interpreted this as evidence that MB was making neural processing more efficient, requiring less blood flow to maintain the same output.
| Study type | Finding | Strength of evidence |
|---|---|---|
| Rodent memory (30+ studies) | Enhanced memory consolidation at 0.5-4 mg/kg | Strong and consistent across paradigms |
| Human fMRI (Rodriguez et al., 2017) | Increased functional connectivity in memory networks | Single RCT, 28 participants, needs replication |
| Human CBF/metabolism (2023) | Reduced blood flow and oxygen consumption | 8 participants, dose-dependent, reproducible across species |
| Alzheimer's clinical trial (TauRx, Phase 3) | Failed to meet primary endpoints for tau aggregation inhibitor form | Large trial but tested modified form (LMTM), not standard MB |
| Traumatic brain injury (rats) | Motor recovery sustained 180 days with monthly dosing | Small sample, animal model |
A 2023 study added an important wrinkle. Researchers at King's College London gave healthy volunteers intravenous methylene blue at 0.5 and 1.0 mg/kg and measured cerebral blood flow and oxygen metabolism directly. Contrary to their own hypothesis, they found that MB reduced both global cerebral blood flow and brain oxygen consumption at both doses. The same pattern appeared in rats. The researchers suggested this might reflect the hormetic nature of the drug: doses that are therapeutic for damaged mitochondria might overshoot in brains that are already functioning normally.
For traumatic brain injury, the animal data is more encouraging. Rats given a single intravenous injection of MB (1 mg/kg) thirty minutes after brain trauma showed reduced motor impairment from day 7 through day 120. Monthly injections extended those benefits to at least 180 days, with reduced microglial inflammation and preserved white matter structure on brain imaging.
The Alzheimer's disease story is the most cautionary. TauRx Therapeutics spent years developing a modified form of methylene blue called LMTM as a tau aggregation inhibitor. Their phase 3 trials did not meet primary endpoints. While some post-hoc analyses showed possible benefits for patients not already on other dementia drugs, the overall results were disappointing enough that the drug has not been approved for Alzheimer's treatment.
Why the dose that helps is dangerously close to the dose that harms
Methylene blue follows a pharmacological pattern called hormesis, where low doses produce beneficial effects and higher doses produce the opposite. This is not a vague relationship. The beneficial range for mitochondrial enhancement in animal studies falls between 0.5 and 4 mg/kg. Above approximately 7 to 10 mg/kg, the same compound begins to inhibit mitochondrial respiration, increase oxidative stress, and paradoxically cause the very damage it prevents at lower doses.
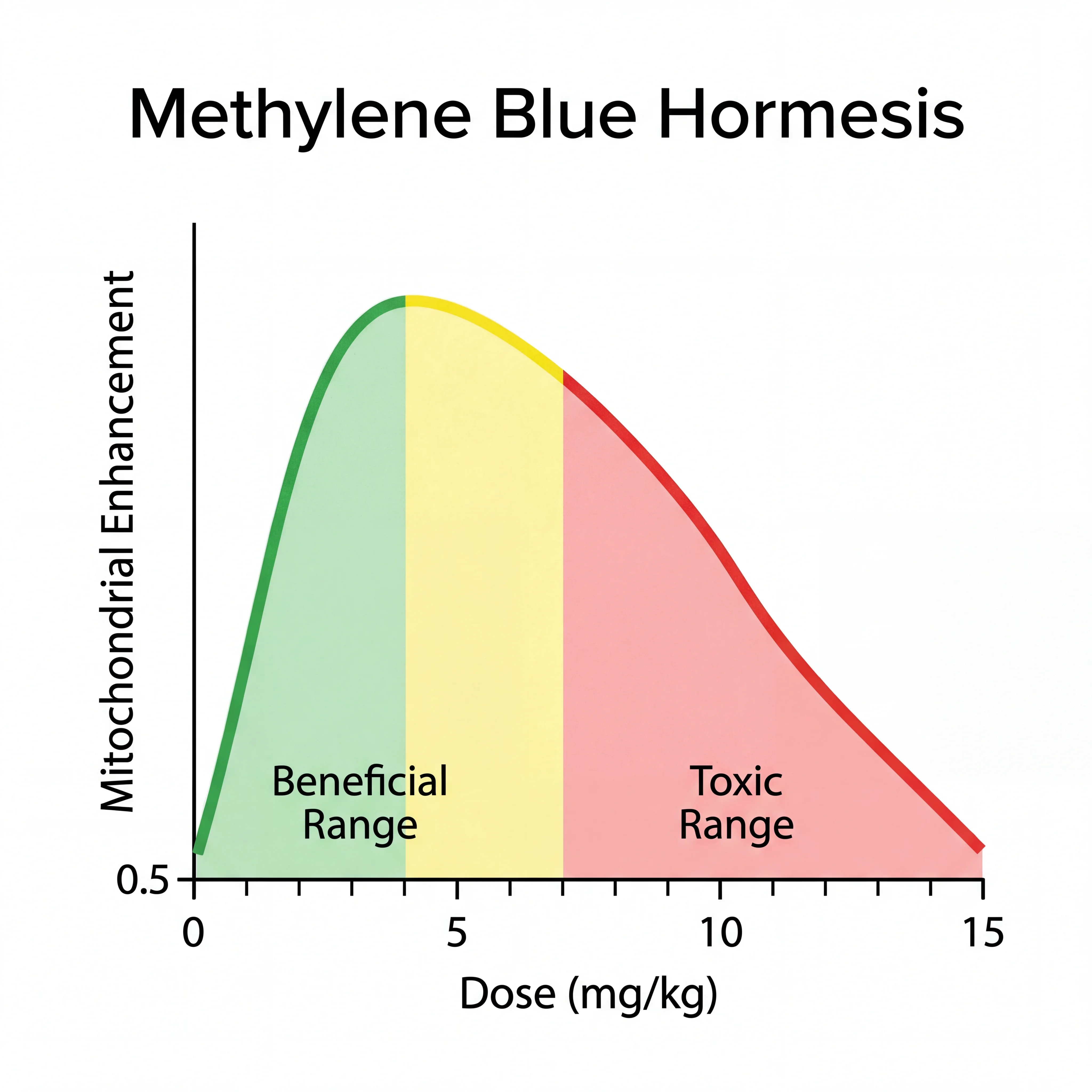
| Dose range | Effect on mitochondria | Clinical observation |
|---|---|---|
| 0.5-4 mg/kg | Enhanced electron transfer, increased complex IV activity, reduced ROS | Memory enhancement in animal models, increased neural connectivity in one human study |
| 4-7 mg/kg | Transition zone with diminishing returns | Therapeutic window for methemoglobinemia treatment (1 mg/kg IV) |
| Above 7-10 mg/kg | Inhibited respiration, increased oxidative stress | Can cause methemoglobinemia (the condition it treats at low doses), hemolysis, neurotoxicity |
This creates a real problem for self-dosing. A 70 kg person aiming for the low end of the beneficial range (0.5 mg/kg) needs 35 mg. At the high end (4 mg/kg), that is 280 mg. The harmful threshold begins around 490 mg for the same person. The gap between "maybe helpful" and "actively toxic" is narrow enough that getting the concentration wrong in an unregulated supplement could push someone past the tipping point.
Compounding this is the fact that methylene blue pharmacokinetics vary considerably between individuals. Oral bioavailability ranges from 53% to 97%. Someone absorbing at the high end could effectively receive nearly double the dose of someone absorbing at the low end, even from identical capsules. Genetic variation in the enzymes that metabolize MB adds another layer of unpredictability.
This is similar to what happens with other compounds that have nootropic potential but require careful dosing. The difference is that methylene blue has a narrower therapeutic window than most supplements on the market.
The drug interaction that can kill you, and other safety concerns
Methylene blue is a potent monoamine oxidase inhibitor. This single fact makes it incompatible with an entire class of common medications that millions of people take daily.
If you take any SSRI (fluoxetine, sertraline, escitalopram, paroxetine), SNRI (venlafaxine, duloxetine), tricyclic antidepressant, certain opioids (tramadol, fentanyl, meperidine), or MAO inhibitors, combining them with methylene blue can trigger serotonin syndrome. This is not a theoretical risk. Fatal cases have been documented. The Mayo Clinic warns that patients should avoid serotonergic medications for at least 72 hours after receiving methylene blue.
Serotonin syndrome symptoms include agitation, rapid heart rate, high blood pressure, dilated pupils, muscle twitching, heavy sweating, and high fever. Severe cases progress to seizures, irregular heartbeat, and organ failure. This can develop within hours of combining MB with serotonergic drugs.

| Risk category | Specific concern | Who is affected |
|---|---|---|
| Serotonin syndrome (potentially fatal) | MAOI activity combined with serotonergic drugs | Anyone on SSRIs, SNRIs, TCAs, MAOIs, tramadol, fentanyl |
| Hemolytic anemia | Overwhelms red blood cell antioxidant defenses | G6PD-deficient individuals (400 million people worldwide) |
| Paradoxical methemoglobinemia | High doses cause the condition it treats at low doses | Anyone exceeding therapeutic dose range |
| Product contamination | Industrial-grade MB contains heavy metals and impurities | Anyone purchasing non-pharmaceutical-grade product |
| Blue discoloration | Skin, urine, and sclera turn blue-green | Everyone, dose-dependent |
| Photosensitivity | Increased sensitivity to UV radiation | Everyone taking MB |
G6PD deficiency deserves special attention. This genetic condition affects roughly 400 million people worldwide, with higher prevalence in populations of African, Mediterranean, and Southeast Asian descent. People with G6PD deficiency cannot generate enough NADPH to handle the oxidative stress methylene blue creates in red blood cells. The result can be severe hemolytic anemia requiring blood transfusion. Many people carry this condition without knowing it.
Then there is the product quality issue. Harvard Health points out that industrial-grade and chemical-grade methylene blue contain impurities that are not safe for human consumption. A casual search for methylene blue online returns fish tank disinfectant alongside products marketed as supplements. Pharmaceutical-grade (USP) methylene blue exists, but it is a prescription product, not what most people buying it on the internet are getting.
Separating the hype from the evidence
The biohacking community has latched onto methylene blue for understandable reasons. The mitochondrial mechanism is real and elegant. The brain penetration is exceptional. The history is fascinating. And the research papers, if you only read the titles and abstracts from animal studies, paint an appealing picture.
But the distance between "this compound improves memory consolidation in rats at carefully controlled doses" and "you should put drops of this in your water every morning" is enormous. Several gaps in the evidence should give anyone pause.
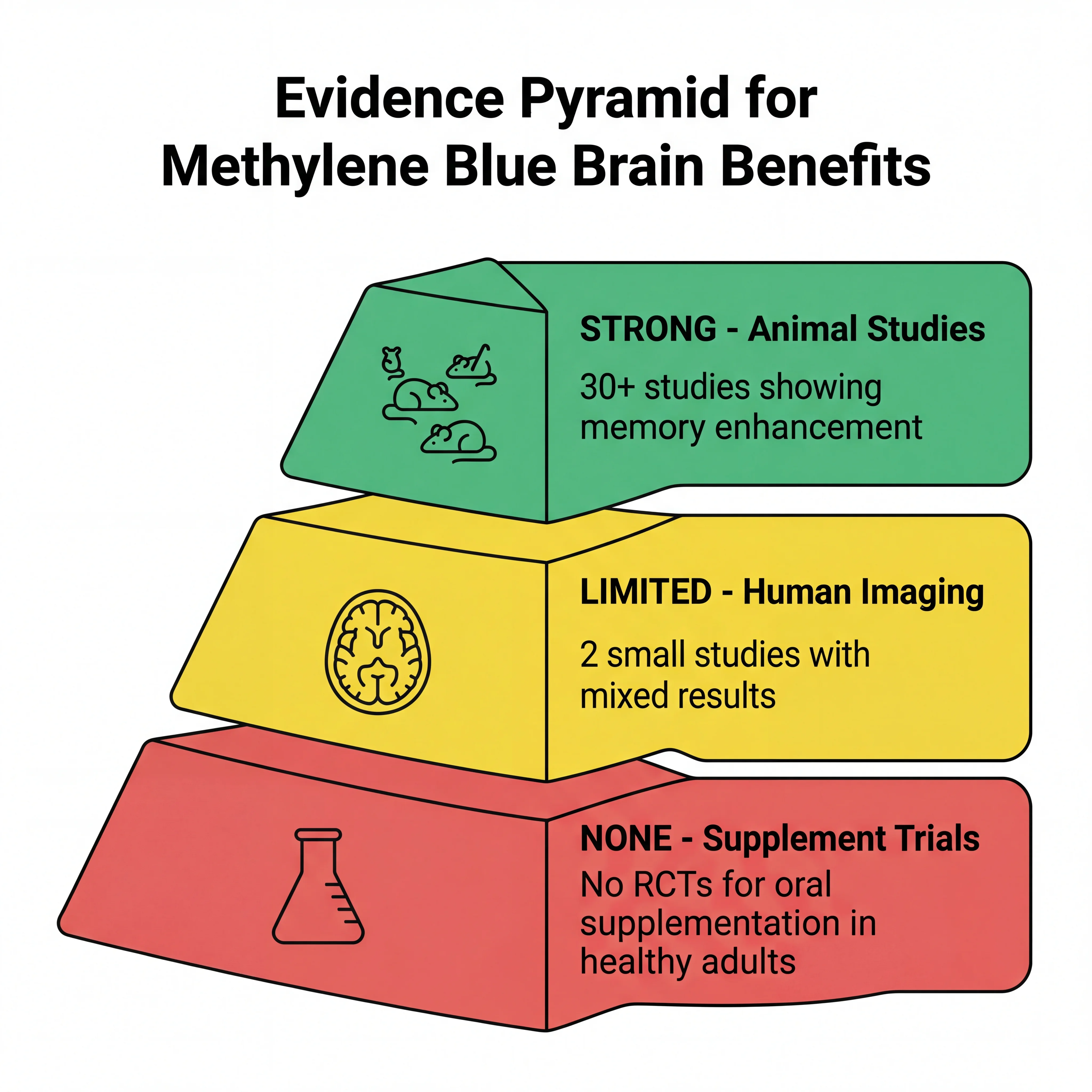
No randomized controlled trial has demonstrated cognitive enhancement from oral methylene blue supplementation in healthy humans going about their daily lives. The single positive human neuroimaging study used pharmaceutical-grade MB in a hospital setting with careful dosing, screening for contraindications, and exclusion of anyone on psychiatric medications. The 2023 King's College London study, which also used pharmaceutical-grade IV methylene blue, found the opposite of what was expected: reduced brain metabolism in healthy people.
The Phase 3 Alzheimer's trials failed. The traumatic brain injury data, while promising, comes exclusively from rat models. And the biohacking community's typical source is unregulated, non-pharmaceutical-grade product of unknown purity, taken without screening for G6PD deficiency or drug interactions, at doses determined by internet forums rather than clinical pharmacology.
None of this means methylene blue is useless for the brain. The mitochondrial mechanism is compelling, and there are plausible reasons to continue studying it for neurodegenerative conditions. Compounds like lion's mane mushroom and creatine have both gone through similar transitions from intriguing animal data to human research, with mixed timelines. Methylene blue may follow a similar path. But the current evidence does not support taking it as a daily cognitive supplement, particularly not without medical supervision.
For those interested in supporting brain health through better-established approaches, peptide therapies represent another area where preliminary research is generating interest, though with similar cautions about the gap between lab findings and self-experimentation.
Frequently Asked Questions
Is methylene blue FDA-approved as a brain supplement?
No. The only FDA-approved indication for methylene blue is the treatment of acquired methemoglobinemia. All brain-related uses, including neuroprotection and cognitive enhancement, remain investigational. Products sold as supplements are not evaluated by the FDA for safety or efficacy, and pharmaceutical-grade methylene blue requires a prescription.
Can I take methylene blue if I am on antidepressants?
Absolutely not without medical supervision. Methylene blue is a potent monoamine oxidase inhibitor, and combining it with SSRIs, SNRIs, tricyclic antidepressants, or certain pain medications can cause serotonin syndrome, which can be fatal. The Mayo Clinic advises waiting at least 72 hours after methylene blue exposure before resuming serotonergic medications. Anyone considering MB should disclose all medications to their physician first.
What is the difference between pharmaceutical-grade and industrial-grade methylene blue?
Pharmaceutical-grade (USP) methylene blue is manufactured under strict quality controls with verified purity, typically above 99%. Industrial and chemical-grade formulations, including those sold as aquarium treatments or laboratory stains, may contain heavy metals, organic impurities, and other contaminants. Harvard Health has warned that purchasing the wrong grade online could make someone seriously ill.
Does methylene blue turn your skin blue?
At the doses used in clinical settings, methylene blue commonly causes blue-green discoloration of urine and can tint the skin and whites of the eyes. This effect is dose-dependent and temporary, typically clearing within a day or two after the last dose. The discoloration is cosmetically noticeable but considered medically benign.
What dose of methylene blue is considered safe?
In clinical settings, methemoglobinemia is treated with 1 mg/kg administered intravenously. The range showing mitochondrial benefits in animal studies is 0.5 to 4 mg/kg. Doses above 7 to 10 mg/kg can produce toxic effects. However, there is no established safe dose for unsupervised oral supplementation, and individual absorption and metabolism vary widely. Self-dosing without medical guidance carries meaningful risk.
Related Articles
- A beginner's guide to nootropics and cognitive enhancement - An overview of compounds that target brain performance, from well-studied options to emerging research.
- NAD+ and NMN supplements for aging and cellular repair - Another mitochondrial support strategy with its own body of evidence on energy metabolism and longevity.
- Lion's mane mushroom for brain health and nerve regeneration - A natural compound with nerve growth factor stimulation properties and cognitive research.
- Creatine benefits beyond muscle: brain, bone, and longevity - How the most studied sports supplement is gaining attention for cognitive and cellular energy support.
- Peptides for health: BPC-157, thymosin beta-4, and what is legal - A look at bioactive peptides, what the research says, and the legal status of common compounds.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.