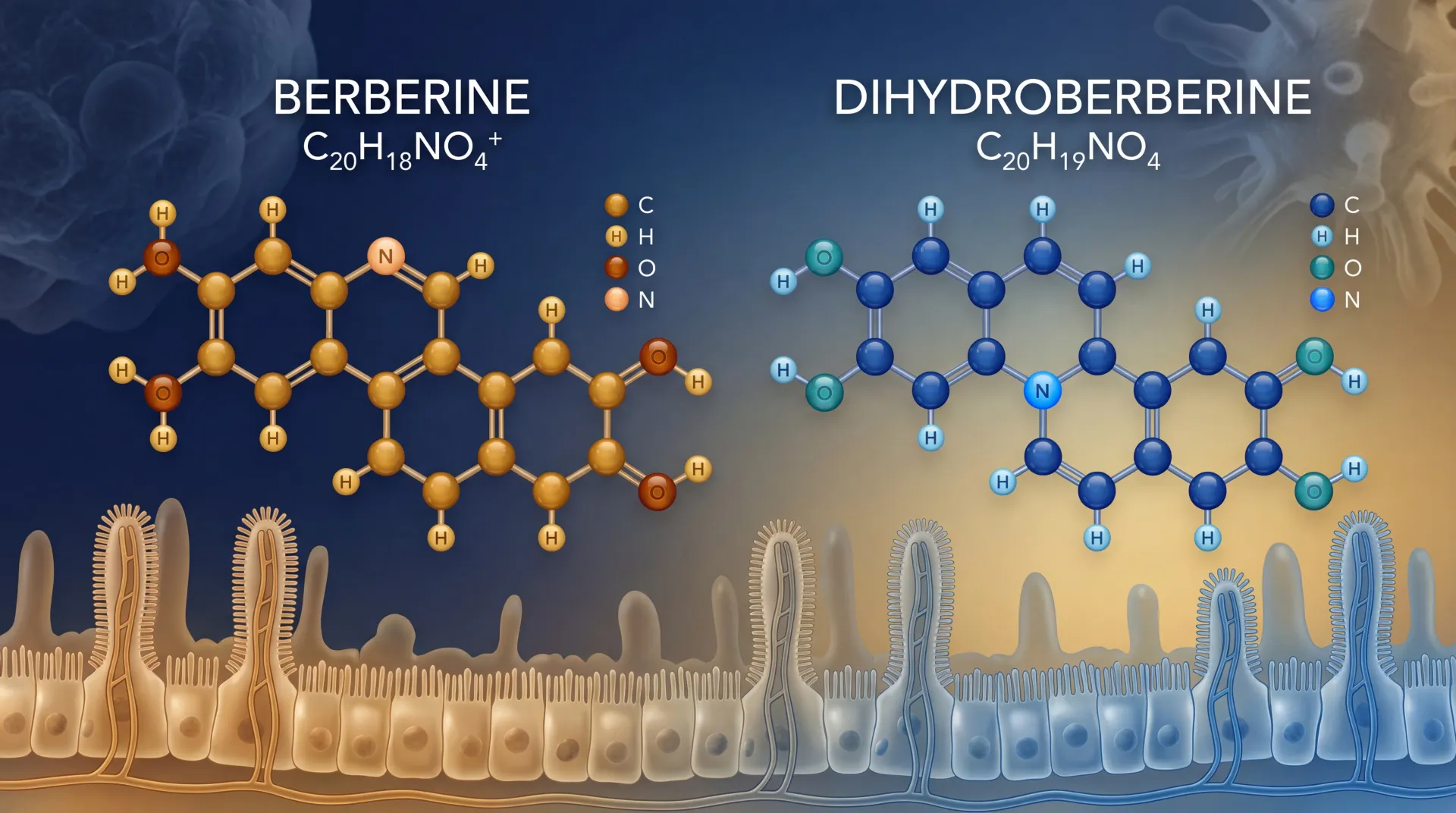
What Makes Dihydroberberine Different from Berberine
Berberine has been part of traditional Chinese medicine for over 400 years, pulled from the roots and bark of plants like goldenseal, barberry, and coptis. It belongs to a class of nitrogen-containing plant compounds called isoquinoline alkaloids, and modern research has zeroed in on its effects on blood sugar, cholesterol, and insulin resistance. The problem? Your body struggles to absorb it.
Dihydroberberine (DHB) is berberine with one small structural tweak. A single chemical bond in berberine's backbone gets reduced -- converted from a double bond to a single bond -- and this minor change dramatically shifts how the compound moves through intestinal walls. The reduced form slips past cellular barriers that block standard berberine, then converts back to berberine once inside your body. Think of it like a disguise: DHB changes shape to get past the bouncer, then reverts to berberine once inside the club.
The only commercially available form of DHB is GlucoVantage, manufactured by NNB Nutrition and produced via a patented preparation method. This is the form used in the only published human pharmacokinetic trial on dihydroberberine, which makes it both the most studied and the only option on the market.
DHB isn't a different supplement with different effects. It's the same active compound -- berberine -- delivered through a more absorbable vehicle. Whether the improved absorption translates to meaningfully better clinical outcomes is a separate question, and the answer requires more human data than currently exists.
The Bioavailability Problem with Standard Berberine
Oral berberine has a bioavailability problem that researchers have called "notorious." Less than 1% of what you swallow reaches your bloodstream, according to multiple pharmacokinetic studies. Examine.com puts the figure at approximately 5% making it into circulation in its natural state, though absorption enhancers can boost this by 6-fold or more.
Two mechanisms account for most of the loss. First, berberine dissolves poorly in water, which limits how much gets absorbed through the intestinal lining. Second, the intestine and liver aggressively break it down before it reaches systemic circulation -- a process called first-pass metabolism. The compound that survives these gatekeepers is a fraction of the original dose.
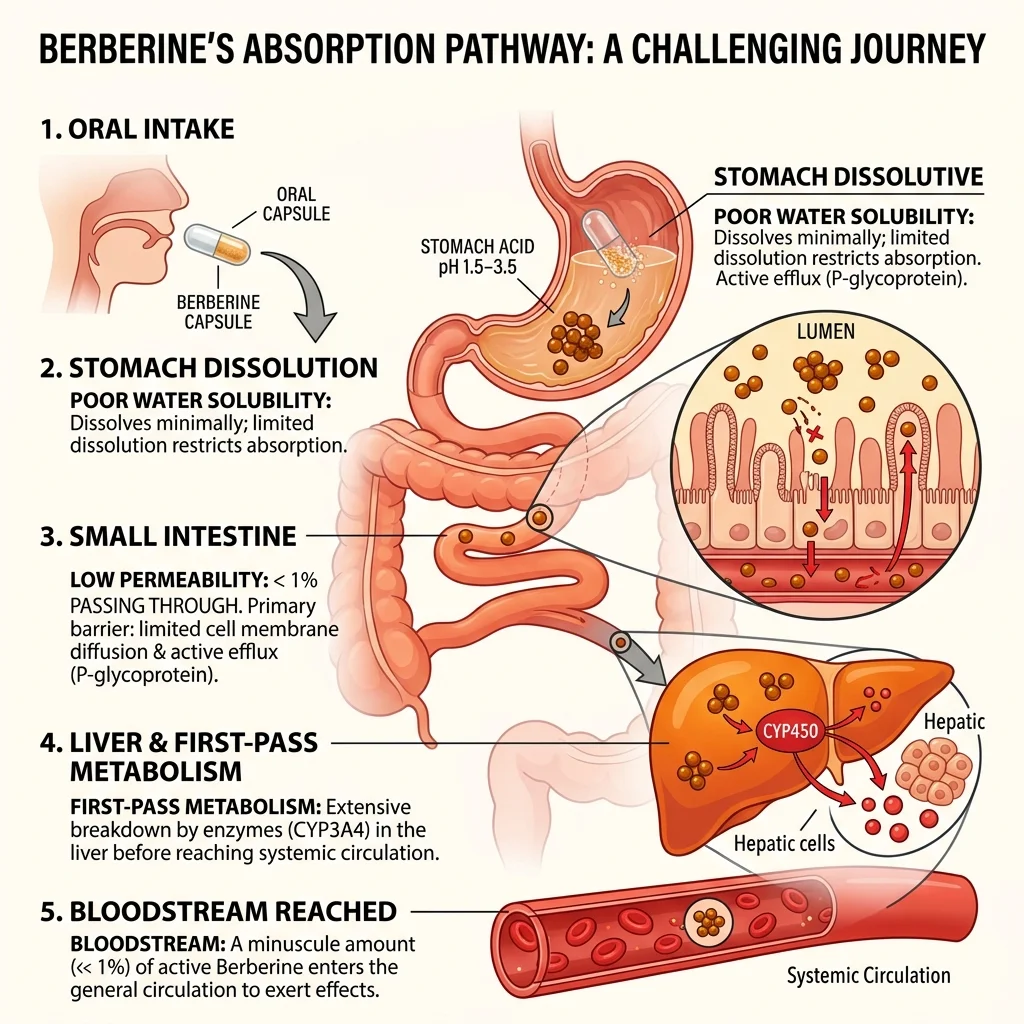
The practical consequence? High doses. The standard berberine regimen calls for 900 to 2,000 mg per day, split into three or four doses. The Cleveland Clinic notes that berberine's short half-life of several hours means you need to dose multiple times daily to maintain stable blood levels. Miss a dose and your levels drop off.
Those high doses come with a cost. In one 13-week study, gastrointestinal adverse events hit 34.5% of participants, with all incidents occurring during the first four weeks. The National Center for Complementary and Integrative Health lists nausea, abdominal pain, bloating, constipation, and diarrhea as reported side effects. Cramping, upset stomach, and GI distress appear consistently across clinical literature.
This creates an awkward tradeoff: the amount of berberine most people take is far more than their bodies can absorb, yet reducing the dose to avoid stomach problems means less of the compound reaches the blood. Researchers have tried encapsulation, spray drying, and pairing berberine with absorption enhancers to fix this. DHB represents a different strategy -- changing the molecule itself rather than the delivery method.
How Dihydroberberine Works in Your Body
Your gut bacteria are already doing what DHB supplements do -- just slowly and inefficiently. A 2015 study published in Scientific Reports by Jia-Wen Shou and Jian-Dong Jiang's team demonstrated that intestinal bacteria naturally convert berberine into dihydroberberine using nitroreductase enzymes. This was the "hidden organ" doing the work: 14 different bacterial strains could perform the conversion, with Enterobacter cloacae and Enterococcus faecium being the most efficient.
The researchers proved the gut bacteria were essential by wiping them out. Rats treated with antibiotics to create a pseudo-germ-free gut showed dramatically reduced berberine efficacy: glucose-lowering dropped by 42%, triglyceride reduction fell 53%, and cholesterol reduction decreased 44%. Fewer bacteria meant less conversion to dhBBR, which meant less absorption, which meant less berberine in the blood.
Once DHB crosses the intestinal wall, something counterintuitive happens: it converts back to berberine. The Shou and Jiang team found that duodenum and jejunum tissue reversed nearly 100% of dhBBR back to berberine within 10 minutes through a non-enzymatic oxidation reaction. Boiling the tissue to destroy enzymes didn't slow the conversion, but adding vitamin C (an antioxidant) almost completely stopped it. The reversion happens automatically -- no enzymes required.
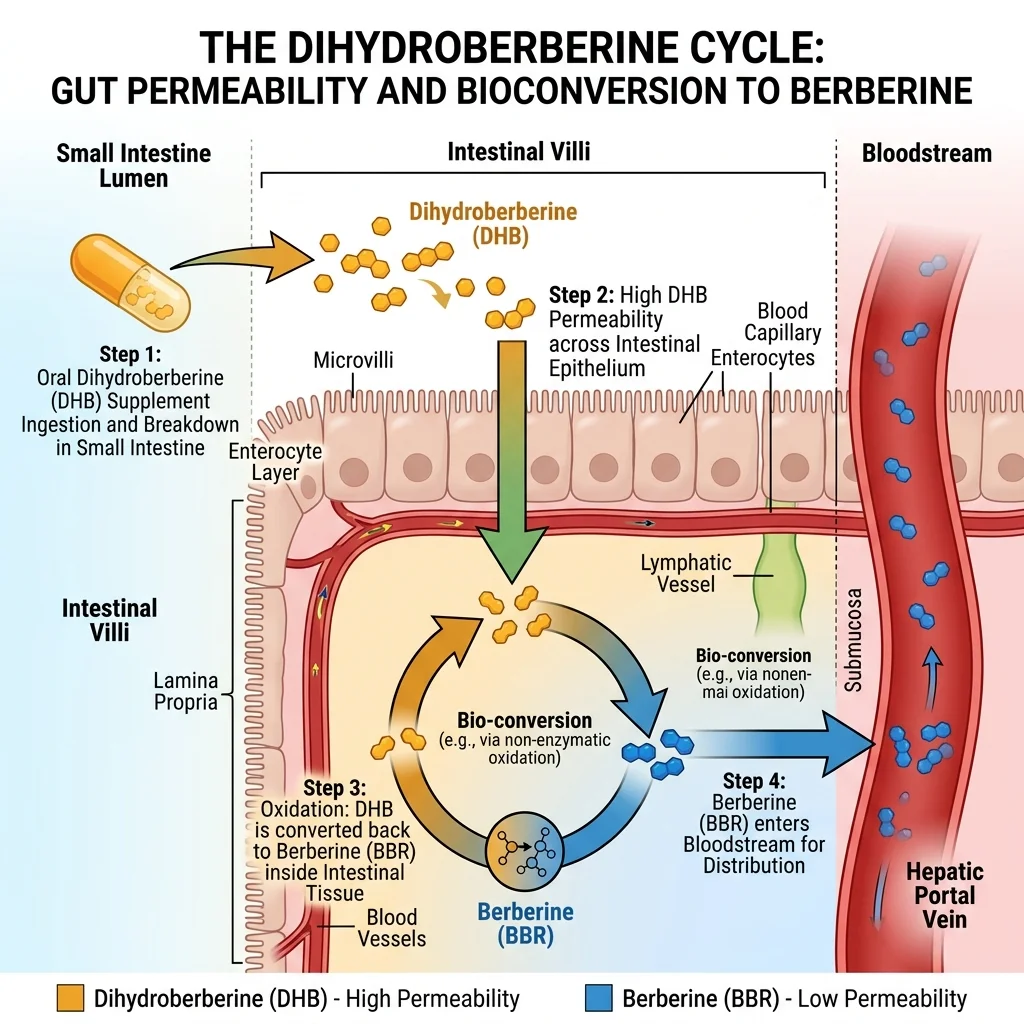
The permeability difference is substantial. In Caco-2 cell models (a standard test for intestinal absorption), dhBBR showed 11.9-fold higher permeability than berberine. The efflux ratio -- how aggressively cells pump the compound back out -- dropped from 32.39 for berberine to just 1.58 for dhBBR. Berberine gets actively rejected by intestinal cells. DHB does not.
So the mechanism works like a shuttle system. DHB enters because its structure avoids the cellular pumps that reject berberine. Once past the intestinal barrier, oxidation reverts it to the pharmacologically active form. Your body ends up with berberine in the blood either way -- DHB just gets more of it there.
All of this berberine -- whether from direct supplementation or from the DHB shuttle route -- acts primarily through one metabolic pathway. It inhibits mitochondrial respiratory complex I, the same target hit by metformin, according to research by Nick Turner's team. This inhibition raises the AMP:ATP ratio inside cells, which activates an enzyme called AMPK. AMPK is sometimes described as the cell's fuel gauge -- when energy runs low, it flips switches that increase glucose uptake, boost fat burning, and reduce new fat production.
Blood Sugar and Metabolic Benefits: What the Research Shows
The evidence base for berberine's blood sugar effects is extensive. The evidence base for DHB specifically is thin. Separating the two matters, because marketing materials often blur them together.
For berberine, the data is strong and growing. An overview of 54 systematic reviews published in 2025 by Lanjun Shi's team found that berberine improved 92.59% of type 2 diabetes outcomes and 90.91% of metabolic syndrome outcomes across the included studies. The Examine.com database, drawing on 11 studies with 912 participants, rates berberine's effect on blood glucose in type 2 diabetes as a "Large Improvement."
Some of the most cited work comes from Yin and colleagues, who ran two separate studies giving berberine at 500 mg three times daily. Over three months, hemoglobin A1C, fasting blood glucose, postprandial glucose, and triglycerides all improved -- and the magnitude of improvement was similar to metformin. The Cleveland Clinic and Mayo Clinic both acknowledge this comparison while cautioning that more high-quality trials are needed.
Key finding: A meta-analysis of berberine monotherapy found significant improvements across six metabolic markers: triglycerides, total cholesterol, LDL, HDL, HOMA-IR (a measure of insulin resistance), and fasting plasma glucose -- all with statistical significance (p < 0.05).
But nuance matters. A large 2024 JAMA Network Open randomized clinical trial with 337 participants tested berberine at 1 g/day for six months in diabetes-free individuals with obesity. The result: berberine did not reduce visceral fat or liver fat compared to placebo. It did reduce LDL cholesterol by 7.72 mg/dL and lowered the inflammatory marker hs-CRP, but it didn't deliver the fat-loss results some had hoped for. The safety profile was excellent -- adverse events matched the placebo group.
This suggests berberine's metabolic benefits may be most pronounced in people who already have metabolic dysfunction -- those with elevated blood sugar, insulin resistance, or type 2 diabetes -- rather than in metabolically normal individuals looking for fat loss. As Mayo Clinic dietitian Tara Schmidt put it, berberine is a "promising supplement" but "not a magical weight-loss solution."
For DHB specifically, the clinical outcomes data barely exists yet. The Moon et al. crossover trial showed no significant differences in glucose or insulin response -- but the study only supplemented for one day in five healthy young men who already had normal blood sugar. The researchers themselves stated that "follow-up efficacy studies on glucose and insulin changes should be completed to assess the impact of berberine and dihydroberberine supplementation in overweight, glucose intolerant populations." Animal data from Turner's team showed dhBBR improved glucose tolerance and reduced insulin resistance in diet-induced obese mice, but animal models don't reliably predict human outcomes.
Dosing, Side Effects, and Safety Considerations
The dosing difference between berberine and DHB is stark, and it comes directly from the absorption data.
| Factor | Standard Berberine | Dihydroberberine (DHB) |
|---|---|---|
| Typical daily dose | 900-1,500 mg (sometimes up to 2,000 mg) | 100-200 mg |
| Doses per day | 3-4 | 1-2 (based on trial protocol) |
| Timing | With meals | With meals (trial protocol) |
| Clinical evidence level | Multiple RCTs, meta-analyses | 1 human PK trial (n=5) |
The crossover trial tested DHB at 100 mg and 200 mg against 500 mg of berberine. Both DHB doses produced significantly more plasma berberine than the 500 mg berberine dose. If DHB achieves equal or greater blood levels at one-fifth to one-half the dose, the logic goes, side effects driven by high intestinal berberine concentrations should diminish.
Regarding safety: berberine interacts with multiple medications through liver enzyme inhibition. Examine.com identifies CYP3A4, CYP2D6, and CYP2C9 as affected enzyme pathways. Practically, this means berberine can increase blood levels of drugs processed by these enzymes, including cyclosporine (anti-rejection), certain statins, diabetes medications, and sedatives like zolpidem. The Cleveland Clinic and Mayo Clinic both stress talking to a provider before combining berberine with prescription drugs.
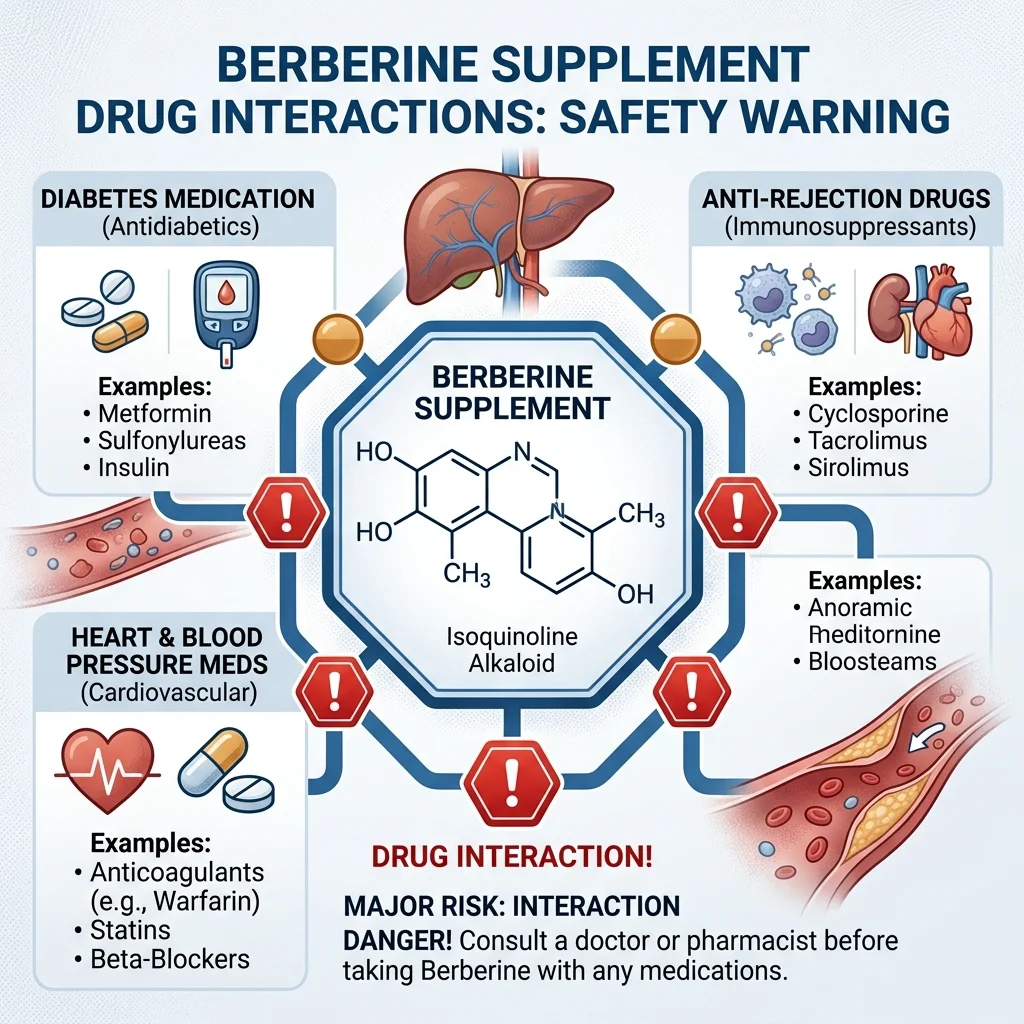
The NCCIH warns explicitly against berberine during pregnancy and breastfeeding, citing bilirubin toxicity risk to infants. This isn't a theoretical concern -- berberine decreases bilirubin binding to proteins, and high bilirubin can cause jaundice and brain damage in newborns. Examine.com and UCLA Health reinforce this warning.
One more quality concern: studies conducted in the United States and France found that berberine supplements frequently did not contain the labeled amount when independently tested. This is a problem common to dietary supplements generally -- the FDA does not regulate them for safety and effectiveness the way it does pharmaceuticals -- but it's particularly relevant here because accurate dosing matters for metabolic effects.
In the DHB crossover trial, 11 adverse events were reported across all conditions, with 90.9% classified as mild. Six occurred in the 100 mg DHB group, three in placebo, one in the 200 mg DHB group, and one in the 500 mg berberine group. The numbers are too small to draw conclusions about relative safety. The most important disclosure: one study author, C.M. Kerksick, serves as a paid advisor to NNB Nutrition, the manufacturer of GlucoVantage. A conflict-of-interest management plan was in place, but readers should be aware of this relationship.
Berberine vs. Dihydroberberine: A Direct Comparison
The pharmacokinetic data tells a clear story. The clinical outcomes data doesn't exist yet. That gap is the central tension with DHB.
| Measurement | 500 mg Berberine | 100 mg DHB | 200 mg DHB | Placebo |
|---|---|---|---|---|
| Peak berberine concentration (CMax) | 0.4 ng/mL | 3.76 ng/mL | 12.0 ng/mL | 0.22 ng/mL |
| Berberine AUC (ng/mL x 120 min) | 42.3 | 284.4 | ~500-600 (trend) | 20.2 |
| Statistical significance vs B500 | -- | p = 0.045 | p = 0.06 (trend) | -- |
The Moon et al. crossover trial found that just 100 mg of DHB produced roughly 5-7 times more plasma berberine than 500 mg of standard berberine over a two-hour window. The 200 mg dose showed an even higher peak concentration of 12.0 ng/mL, though the AUC difference between the two DHB doses only trended toward significance (p = 0.073) rather than reaching it.
The study did not show any difference in glucose or insulin response. Neither glucose (p = 0.97) nor insulin (p = 0.24) differed across conditions. The researchers attribute this to the short supplementation period (participants took only four doses total before testing) and the fact that all five participants were healthy young men with normal insulin sensitivity. A glucose-lowering supplement is hard to evaluate in people whose glucose is already normal.
The study's limitations deserve direct acknowledgment. Five participants is not enough to establish clinical efficacy -- it's a proof-of-concept for absorption. All participants were male, all were young (mean age 26), and all were metabolically healthy. The study was funded by NNB Nutrition. These aren't disqualifying flaws, but they constrain how much weight to put on the findings.
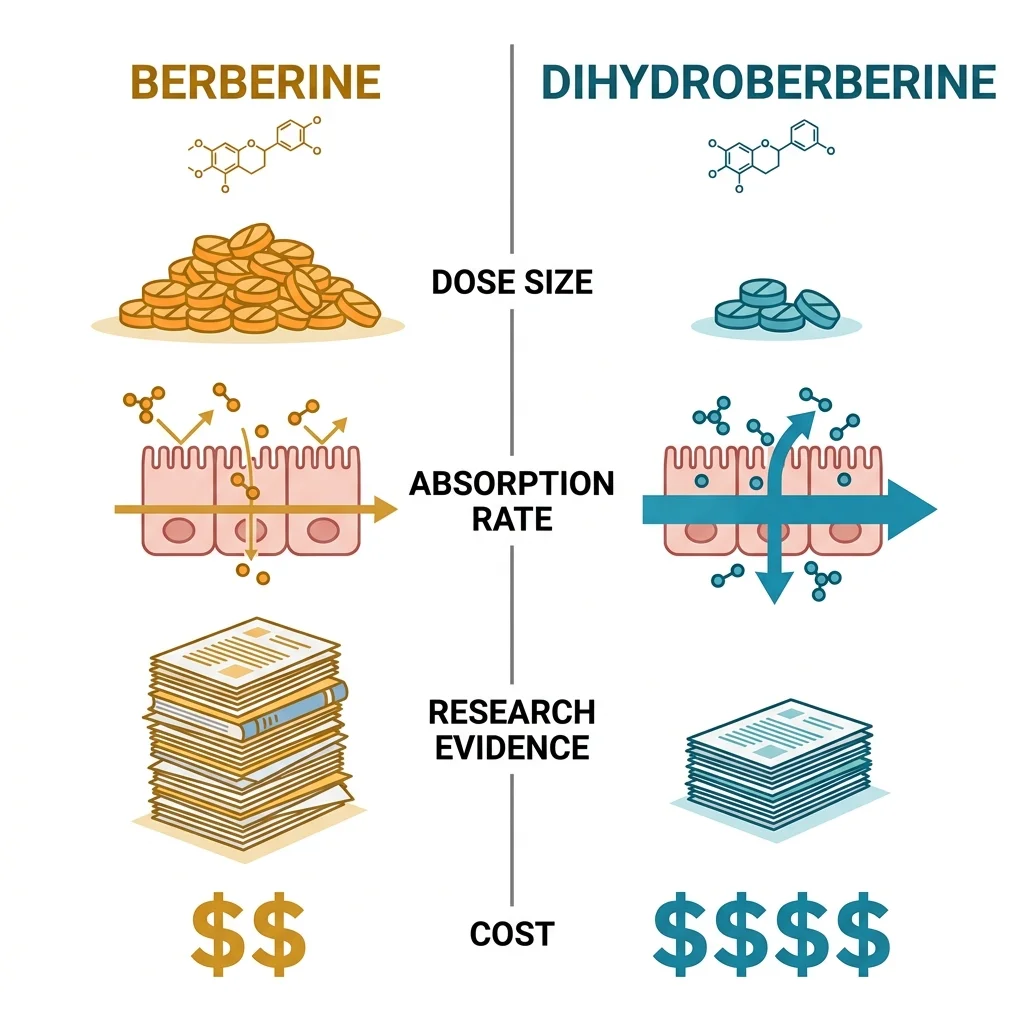
The honest assessment: DHB demonstrably gets more berberine into your blood at lower doses. Whether more berberine in the blood at lower oral doses translates to better metabolic outcomes -- or just equivalent outcomes with fewer GI side effects -- remains unproven in humans. The pharmacokinetic case is strong. The clinical outcomes case needs trials that don't yet exist.
| Factor | Berberine | Dihydroberberine |
|---|---|---|
| Years of research | Decades, hundreds of studies | Limited, primarily since 2008 |
| Human clinical trials | Multiple RCTs, meta-analyses | 1 PK trial (n=5) |
| Proven metabolic effects | Blood glucose, lipids, insulin sensitivity | Absorption only (no clinical outcomes) |
| Dose required | 900-2,000 mg/day | 100-200 mg/day |
| GI side effect profile | Well-documented (34.5% in trials) | Insufficient data |
| Cost per month | Lower (commodity supplement) | Higher (patented ingredient) |
| FDA regulated | No (dietary supplement) | No (dietary supplement) |
Frequently Asked Questions
Is dihydroberberine better than berberine for blood sugar control?
DHB gets more berberine into your bloodstream at a fraction of the dose, but no human study has directly compared blood sugar outcomes between the two. The one published human trial measured absorption only, not metabolic effects. If you respond well to berberine but struggle with the GI side effects of high doses, DHB may be worth trying. If standard berberine works for you without issues, there's no proven clinical reason to switch.
Can I take dihydroberberine with metformin or other diabetes medications?
Berberine (which DHB converts into) can interact with diabetes medications and potentially amplify their blood-sugar-lowering effects. While this likely doesn't increase hypoglycemia risk based on current data, the Cleveland Clinic and Mayo Clinic both recommend consulting your healthcare provider before combining berberine products with any prescription medication, especially drugs metabolized by CYP3A4 enzymes.
What dose of dihydroberberine should I take?
The only human pharmacokinetic trial tested 100 mg and 200 mg doses. Both produced significantly more plasma berberine than 500 mg of standard berberine. Most DHB supplements on the market contain 100-200 mg per serving. There are no long-term dosing studies, and the optimal dose for metabolic benefits hasn't been established in clinical trials.
Does dihydroberberine have fewer side effects than berberine?
Theoretically, yes -- because you need much less of it to achieve equal or greater blood berberine levels, so less compound sits in the gut causing irritation. But the safety data is too limited (one trial, five participants) to make definitive claims. Standard berberine's 34.5% GI adverse event rate in clinical trials is well-documented. DHB's side effect profile at scale remains unknown.
Is dihydroberberine safe during pregnancy?
No. DHB converts to berberine in your body, and the NCCIH states that berberine is likely unsafe for infants due to bilirubin toxicity risk, which can cause brain damage. Avoid both berberine and DHB during pregnancy and breastfeeding.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.