The 2026 Peptide Landscape — Why Doctors Are Rethinking Weight Loss
Walk into an obesity clinic in 2018, and the conversation usually started with calorie counts and stalled there. Walk into the same clinic in 2026, and a clinician is more likely to start with hormones — GLP-1, GIP, glucagon receptor signalling, and how a weekly injection can do what a decade of dieting did not. Obesity medicine has not changed this fast in fifty years.
Two regulatory turning points define the current moment. The FDA removed tirzepatide from the national shortage list in December 2024 and semaglutide in February 2025, ending a window where compounding pharmacies could legally produce knockoff versions of the brand-name drugs. The same regulator then announced it intended to act against non-FDA-approved GLP-1 products, leaving patients who had relied on $49-a-month compounded versions scrambling for the $149-and-up brand pens. Compounded medications, the FDA has been clear, are not reviewed for safety, quality, or effectiveness.
The economics are unforgiving. A 2025 KFF survey found 27 percent of insured GLP-1 users still paid the full out-of-pocket cost, and that number does not include the millions without coverage at all. Some patients have re-enrolled in research-grade peptide clinics; others have shifted to the older, cheaper liraglutide; and a small but growing group is asking about peptides that never made it to FDA approval at all — tesamorelin, AOD-9604, the CJC-1295/ipamorelin stack — all of which doctors prescribe quietly, off-label, often in small specialty clinics rather than primary-care offices.
Cleveland Clinic puts the typical first-year weight loss on FDA-approved medications at 3 to 12 percent of body weight, but the new generation of multi-receptor agonists has redrawn that ceiling. The seven peptides below are the ones moving the needle in 2026 — four hold FDA approval for weight management or a related metabolic indication, three remain investigational or are used off-label. None work without lifestyle change. All sit in a regulatory and clinical landscape that looks very different from the one we had eighteen months ago.
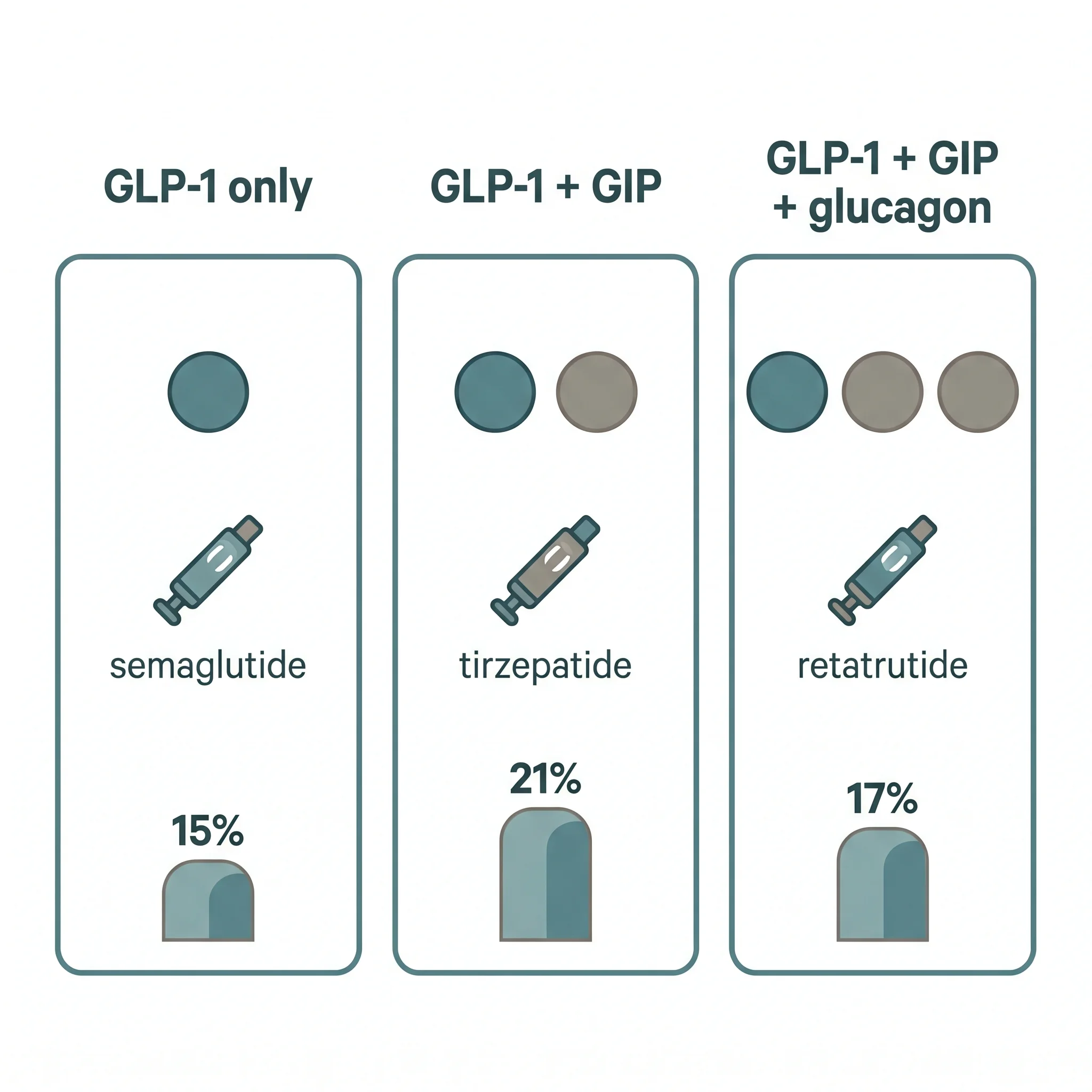
The seven peptides below cover most clinical conversations about weight-loss biologics in 2026. Four are FDA-approved for weight management or a metabolic indication — semaglutide, tirzepatide, liraglutide, and tesamorelin (the last for HIV-associated lipodystrophy). The cagrilintide-semaglutide combination CagriSema is in late-stage regulatory review. Retatrutide, AOD-9604, and CJC-1295 / ipamorelin remain investigational or off-label.
Semaglutide (Ozempic, Wegovy) — The GLP-1 That Started Everything
Semaglutide is the molecule that turned a niche diabetes drug class into a household word. In the pivotal STEP 1 trial — a 68-week double-blind study published in the New England Journal of Medicine — 1,961 adults with a BMI of 30 or higher (or 27-plus with weight-related comorbidities) lost an average of 14.9 percent of body weight on once-weekly 2.4 mg semaglutide versus 2.4 percent on placebo. The treatment difference of 12.4 percentage points was the largest seen in obesity pharmacology to that point. Half the participants — 50.5 percent — hit the 15 percent loss threshold, a number that rivals what bariatric surgery achieves at the lower end of its outcome range.
Semaglutide is a synthetic version of glucagon-like peptide-1, the hormone your gut releases after eating. Think of GLP-1 as the "I'm full" signal a healthy gut sends after dinner. Semaglutide turns that signal up and keeps it on. It also slows stomach emptying, which is the part most users notice first — the way food simply stops calling. A fatty-acid side chain that binds to albumin gives the molecule a half-life long enough for one injection a week.
The catch is what happens when the message stops. The STEP 1 extension followed 327 participants for a year after they discontinued semaglutide and lifestyle intervention; they regained roughly two-thirds of the weight they had lost, with net loss falling from 17.3 percent at week 68 to 5.6 percent at week 120. Cardiometabolic gains — blood pressure, lipids, A1c — drifted back toward baseline. The investigators framed it bluntly: obesity behaves like a chronic condition, and like high blood pressure or hypothyroidism, the medication treats it only as long as you keep taking it.
The next chapter of the semaglutide story is already published. CagriSema — a fixed-dose combination of cagrilintide (an amylin analog) and semaglutide — produced a 22.7 percent mean weight reduction in the REDEFINE 1 trial published in NEJM on June 22, 2025. Among adherent patients, 50.7 percent of obese participants reached a non-obese BMI under 30, and 23.1 percent lost more than 30 percent of their starting weight. Those numbers are now the benchmark Novo Nordisk is competing against — including against itself.
Practically, this means that if you are starting semaglutide in 2026, the realistic conversation is not "lose weight, then stop." It's "this is likely a long-term medication, and we'll plan for that — including how to taper, how to maintain with diet and resistance training, and how to budget for it." If your insurance plan denies coverage and the brand price is unworkable, your clinician will probably suggest tirzepatide as the next conversation rather than gambling on a compounded version.
Tirzepatide (Mounjaro, Zepbound) — The Dual-Agonist Outperforming Semaglutide
Tirzepatide changed the ceiling. Where semaglutide hit a single receptor — GLP-1 — Eli Lilly's molecule activates both GLP-1 and GIP, the second incretin hormone your gut releases after eating. The pharmacological logic is straightforward: hit two satiety pathways instead of one. The clinical results have been more striking than most doctors expected.
SURMOUNT-1 was the registrational trial. Over 72 weeks in 2,539 adults with obesity, the 15 mg dose produced a 20.9 percent mean weight loss, the 10 mg dose 19.5 percent, and the 5 mg dose 15.0 percent, versus 3.1 percent on placebo. 91 percent of patients on the highest dose lost at least 5 percent of body weight; between 50 and 57 percent of those on the 10 mg or 15 mg arms lost more than 20 percent. For context, those weight loss percentages put tirzepatide in territory previously reserved for sleeve gastrectomy.
The head-to-head trial published this past summer settled the question most patients ask their doctors. SURMOUNT-5, a 72-week open-label study of 751 adults with obesity but without type 2 diabetes, found tirzepatide produced a 20.2 percent mean weight reduction versus 13.7 percent for semaglutide (p<0.001). Waist circumference fell 18.4 cm on tirzepatide versus 13.0 cm on semaglutide — a meaningful difference for cardiometabolic risk. For most patients without specific reasons to favor one molecule, tirzepatide is now the more potent option.
For most patients choosing between the two: if you can tolerate higher gastrointestinal effects (tirzepatide produces more, particularly during dose escalation) and Zepbound is covered or affordable, tirzepatide tends to win. If you already tolerate semaglutide and the weight is coming off steadily, switching to chase a few percentage points means restarting the dose-escalation grind for marginal gain. The honest decision is rarely about which drug "won" a trial. It's about which side-effect curve your body fits.

Retatrutide — The Triple-Agonist Pushing Past Tirzepatide
If tirzepatide raised the ceiling, retatrutide is poised to break through it. The molecule, in late-stage development at Eli Lilly, is the first triple-receptor agonist — it activates GLP-1, GIP, and the glucagon receptor. The first two suppress appetite and slow gastric emptying. The third does something neither semaglutide nor tirzepatide does: it directly stimulates energy expenditure and lipolysis. In effect, retatrutide tells your body to eat less and burn more.
The numbers are the kind that make endocrinologists sit up. A 2025 systematic review and meta-analysis pooling three RCTs (878 patients) found retatrutide produced a mean weight loss of 14.33 percent versus placebo, with the highest dose driving even larger effects: 12 mg produced 16.90 percent loss; 8 mg 16.31 percent; 4 mg 9.86 percent. At 36 weeks, 63 percent of retatrutide patients had lost at least 15 percent of body weight, compared to 40 percent on tirzepatide at the same time point. The 12 mg dose pushed weight loss to magnitudes that overlap with the lower end of bariatric surgery outcomes — a comparison most pharmacology has never been able to make.
The metabolic side effects look as good as the weight numbers. Hemoglobin A1c dropped 0.91 percentage points; systolic blood pressure fell 9.88 mm Hg; fasting plasma glucose dropped 23.51 mg/dL. For context, those blood-pressure changes alone would qualify retatrutide as an antihypertensive in another life. The waist-circumference reduction averaged 10.5 cm.
One important caveat: retatrutide is not yet FDA-approved as of early 2026. Phase 3 data has been positive, and Lilly has signaled an approval pathway, but until that decision lands the drug remains research-grade. If your physician brings up retatrutide today, they are usually talking about either expanded-access trial enrollment or — much more often — a gray-market compound sold by online peptide vendors. Trial enrollment is reasonable medicine. The vendor route is how patients end up in poison-control reports.
Liraglutide (Saxenda) — The Original, Still in the Toolkit
Liraglutide is the molecule that proved the GLP-1 thesis worked in obesity, and it's the one most people forget about now that semaglutide and tirzepatide dominate the headlines. Marketed as Saxenda at the 3.0 mg dose for weight management and Victoza at 1.8 mg for diabetes, it received FDA approval for chronic weight management in December 2014 — the first GLP-1 to clear that bar.
The five SCALE trials — the registrational program for Saxenda — established a consistent picture. Across phase III studies, liraglutide 3.0 mg produced 4 to 6 kg greater weight loss than placebo, with 46 to 63 percent of patients achieving at least 5 percent loss and 22 to 37 percent reaching the 10 percent threshold. The pivotal SCALE Obesity and Prediabetes trial enrolled 3,731 participants and produced an 8.0 percent mean weight reduction — modest by today's standards but durable, with most patients maintaining loss for two years on continuous treatment.
Where liraglutide actually shines in 2026 is in adjacent indications. The SCALE Sleep Apnea trial showed a 5.7 percent weight reduction and improvement in apnea-hypopnea index of 12.2 events per hour — a meaningful number for anyone with moderate obstructive sleep apnea. SCALE Obesity and Prediabetes also showed a 79 percent reduction in progression to type 2 diabetes, the strongest diabetes-prevention signal in any weight-loss medication studied to date.
Then there's the cardiovascular data. The LEADER trial enrolled 9,340 type 2 diabetes patients and followed them for 3.8 years on liraglutide 1.8 mg. Cardiovascular death fell 22 percent (HR 0.78), all-cause mortality fell 15 percent (HR 0.85), and the composite primary outcome (CV death, MI, or stroke) fell from 14.9 percent on placebo to 13.0 percent on liraglutide (HR 0.87, p=0.01 for superiority). The weight-management 3.0 mg dose has not been studied in a comparable cardiovascular outcomes trial, so the LEADER findings extrapolate imperfectly — but they remain the strongest hard-endpoint evidence for any GLP-1 drug.
The reason most patients in 2026 still get tirzepatide or semaglutide rather than liraglutide is not a question of efficacy. It's the daily injection. A daily needle versus a weekly one is the kind of compliance gap that tilts most patients (and most prescribers) toward the newer molecules. But for a patient with prediabetes plus moderate sleep apnea who can't get insurance coverage for the once-weekly drugs, liraglutide is the workhorse — cheaper, generic-track, and backed by a decade of cardiovascular outcomes data the newer drugs are still accumulating.

Tesamorelin — The Visceral Fat Specialist Used Off-Label
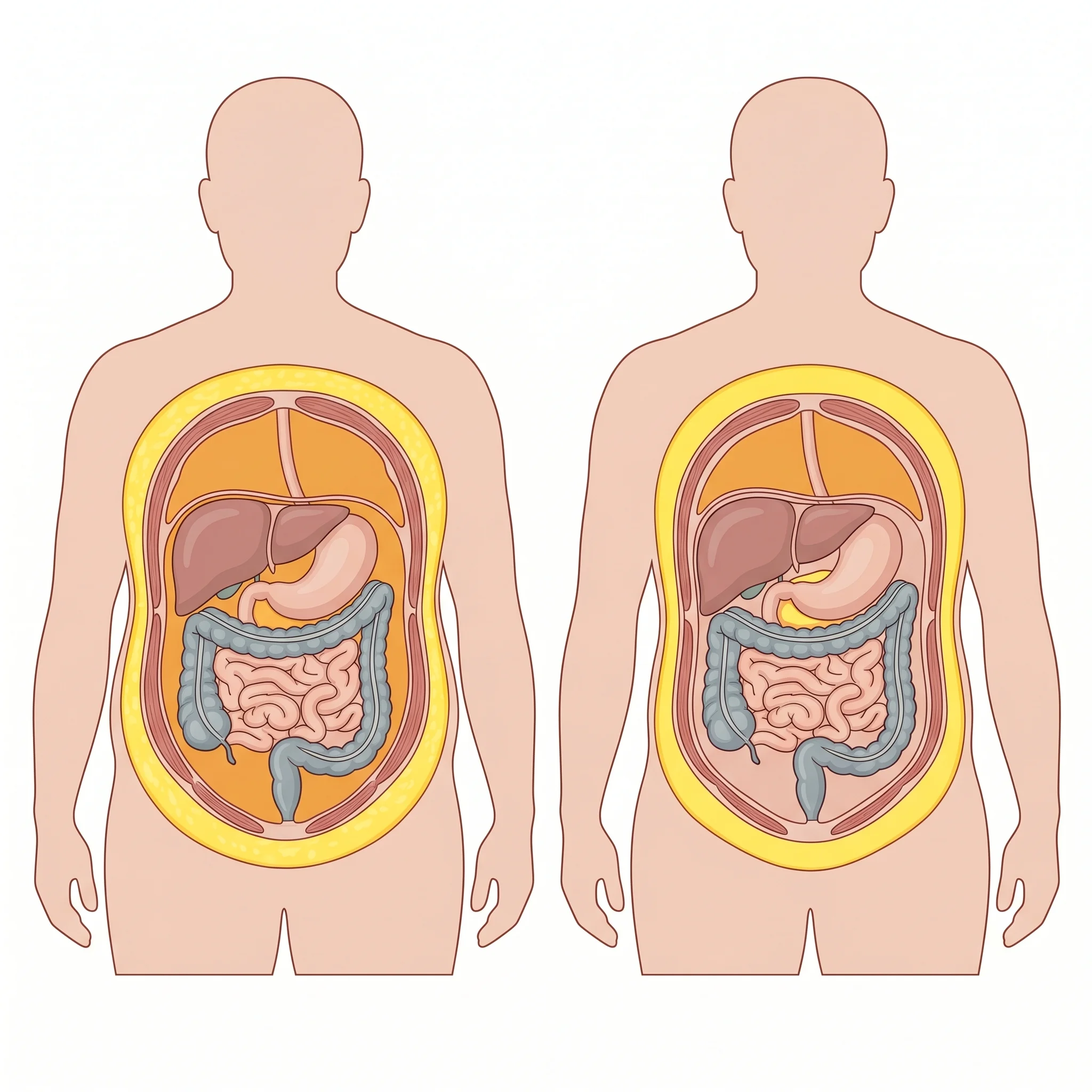
Tesamorelin is the peptide most patients have never heard of and most endocrinologists know exactly what to do with. Sold as Egrifta, it's a synthetic analog of growth hormone–releasing hormone — meaning it tells your pituitary to release more of your own growth hormone in natural pulses, rather than flooding the system with synthetic GH directly. The FDA approved it for HIV-associated lipodystrophy, where antiretroviral therapy can cause a striking accumulation of belly fat.
The clinical evidence on visceral fat reduction is genuinely impressive. Takara Stanley and colleagues at Massachusetts General Hospital ran a double-blind randomized trial of 50 HIV-infected patients with abdominal fat accumulation, comparing tesamorelin 2 mg subcutaneously daily to placebo for 6 months. Visceral adipose tissue dropped 9.9 percent versus a placebo gain of 6.6 percent; liver fat fell with a net treatment effect of -2.9 percent in lipid-to-water ratio (P=0.003). Crucially, lean mass did not change (P=0.23). The drug strips visceral fat — the dangerous, organ-encasing kind — without touching the muscle you actually want to keep.
A more recent 12-month trial in HIV patients on integrase inhibitor antiretrovirals — the modern standard of care that itself causes adipose tissue dysfunction — confirmed the effect. 67 percent of tesamorelin recipients achieved at least 8 percent visceral fat reduction, versus 13 percent on placebo.
That selectivity for visceral fat is why some metabolic specialists use tesamorelin off-label in patients with non-HIV metabolic syndrome — particularly those with disproportionate central obesity, fatty liver, and insulin resistance who haven't responded well to GLP-1s alone. The trade-offs are real. HbA1c rose modestly in the Stanley trial: +0.20 percent on tesamorelin versus +0.02 percent on placebo (P=0.03), and discontinuation due to paresthesias or arthralgias is more common than with GLP-1s. The cost is also significant — Egrifta carries a list price most insurers will not cover for off-label use.
The practical implication: if a clinician is considering tesamorelin for non-HIV metabolic disease, you are in highly specialized territory. This is a peptide for patients with specific phenotypes — usually visceral-predominant obesity with hepatic steatosis — and the conversation should include realistic expectations about glucose monitoring, cost, and the modest effect size on total body weight (BMI did not change significantly in the Stanley trial). Tesamorelin is not a weight-loss drug in the sense most patients want one. It is a body-composition drug.
AOD-9604 and CJC-1295 / Ipamorelin — Body-Recomposition Peptides With Limited Human Data
This is the section where doctors get cautious and patients get curious. AOD-9604, CJC-1295, and ipamorelin are the peptides you see advertised on wellness-clinic websites and discussed in the more enthusiastic corners of biohacking forums. They are also the peptides with the thinnest human evidence base. Honesty about what is and isn't known matters here, because the marketing claims and the trial data are not in the same neighborhood.
AOD-9604 is a synthetic fragment of human growth hormone — specifically residues 177-191, the section believed to drive GH's lipolytic (fat-burning) effects without its growth-promoting effects. The original case for the molecule comes from a 2000 study in Hormone Research in obese Zucker rats. Daily oral AOD-9604 at 500 µg/kg for 19 days reduced body-weight gain by over 50 percent compared to controls, while euglycemic clamp testing showed no impact on insulin sensitivity. The lipolytic activity of adipose tissue increased.
That sounds compelling. Then it didn't translate. Subsequent human clinical development pursued AOD-9604 as an anti-obesity agent and it never received FDA approval — the human weight-loss data did not match the rat data, and it is now sold primarily through compounding pharmacies and "research peptide" vendors. If a clinic is recommending AOD-9604 for fat loss in 2026, the evidence base behind that recommendation is largely the rat study above plus mechanistic plausibility.
CJC-1295 and ipamorelin are usually prescribed as a stack for body recomposition rather than weight loss per se. Both increase the body's own growth hormone secretion, but through different mechanisms. CJC-1295 is a long-acting GHRH analog with an estimated half-life of 5.8 to 8.1 days; a single subcutaneous injection produced 2- to 10-fold sustained increases in plasma GH lasting 6 days or more, and 1.5- to 3-fold increases in IGF-1 lasting 9 to 11 days. After multiple weekly doses, IGF-1 levels stayed above baseline for up to 28 days.
Ipamorelin is the partner molecule. Originally developed by Novo Nordisk in 1998, it is a pentapeptide that selectively stimulates GH release without elevating cortisol, prolactin, ACTH, FSH, LH, or TSH. That selectivity is the entire point — older GH secretagogues like GHRP-2 and GHRP-6 raise stress hormones along with GH, which negates many of the metabolic benefits. Ipamorelin doesn't.
The pairing makes pharmacological sense — CJC-1295 raises baseline GH/IGF-1 tone, ipamorelin adds clean pulses on top — and clinics that prescribe the combination for "fat loss and recovery" are not necessarily wrong about the biology. They are, however, prescribing largely on mechanistic plausibility rather than weight-loss RCT data. Neither molecule has a registrational obesity trial on the scale of STEP or SURMOUNT. Anecdotal user reports (improved sleep, modest changes in body composition over months) are consistent with what the GH/IGF-1 axis would be expected to do, but "consistent with" is not the same as "proven."
If you are considering this stack, ask the prescribing clinic two questions: where does the peptide actually come from (a 503A pharmacy with traceable sourcing, or an unregistered overseas manufacturer), and what specific endpoint are you targeting. "General body composition" usually means the clinic doesn't have a measurable goal in mind, and that's the kind of treatment that tends to stretch on indefinitely without clear stopping rules.
Safety, Side Effects, and the FDA Compounding Reversal

Every peptide above carries a side-effect profile that matters. Doctors don't quietly recommend these molecules because they're harmless — they recommend them because the benefits, in the right patient, outweigh known risks. Knowing what those risks are is what separates an informed conversation from a marketing pitch.
The gastrointestinal load
Roughly 50 to 60 percent of GLP-1 patients experience nausea, vomiting, diarrhea, or constipation during the initial weeks of treatment. Most cases are mild to moderate and dose-dependent — they fade as the body adjusts and as dose escalation proceeds slowly. The STEP 1 trial saw 4.5 percent of semaglutide recipients discontinue treatment due to GI events versus 0.8 percent on placebo. SURMOUNT-1 reported similar numbers for tirzepatide.
Gallbladder and biliary risk
Rapid weight loss in any context — not just GLP-1s — increases gallstone risk. A meta-analysis of 76 randomized trials found GLP-1 use is associated with a pooled relative risk of 1.37 (95% CI 1.23-1.52) for gallbladder and biliary disorders. The risk was higher in studies using weight-loss doses than in diabetes-management doses, and it scaled with treatment duration. The mechanism is partly the rapid weight loss itself (which supersaturates bile with cholesterol) and partly direct GLP-1 effects on gallbladder motility.
Surgery and anesthesia
Delayed gastric emptying is the GLP-1 mechanism of action — it's also a problem if you need anesthesia. The American Society of Anesthesiologists currently recommends discontinuing short-acting GLP-1s one day before procedures and long-acting formulations at least one week beforehand. A semaglutide ultrasound study found 70 percent of recipients still had retained solid gastric contents after an overnight fast, versus 10 percent of controls. If you're scheduled for anything requiring sedation, including endoscopy, your prescriber and your anesthesiologist need to coordinate.
Eyes — diabetic retinopathy and NAION
Two ocular signals deserve attention. The SUSTAIN-6 trial found semaglutide associated with a hazard ratio of 1.76 (95% CI 1.11-2.78) for diabetic retinopathy complications, mostly in patients with pre-existing retinopathy. The mechanism is thought to be the rapid drop in blood glucose itself rather than direct retinal toxicity, but the practical advice — get a baseline eye exam if you have diabetes before starting — is the same either way.
The newer concern is non-arteritic anterior ischemic optic neuropathy (NAION), an uncommon cause of sudden vision loss. Hathaway and colleagues reported in 2024 that NAION incidence in semaglutide users was 4.28 per 1,000 person-years in T2D patients and 7.64 per 1,000 in overweight or obese patients without diabetes, over 36 months. A retrospective cohort study using the TriNetX database did not find an increased risk over five years, so the signal is being investigated rather than considered settled. Anyone reporting sudden vision change on a GLP-1 should be evaluated immediately.
Mental health
The suicidal ideation question prompted an EMA inquiry in mid-2023, but the randomized evidence has been reassuring. A pooled analysis of the STEP 1, 2, 3, and 5 trials found semaglutide 2.4 mg did not increase depressive symptoms or suicidal ideation versus placebo; a TriNetX retrospective cohort actually found semaglutide reduced both new and recurrent suicidal ideation compared to other anti-obesity medications. The label still requires monitoring, and any patient with active depression should be evaluated by their prescriber.
Muscle loss
The "Ozempic muscle" headlines are real but have been somewhat oversimplified. Studies show lean mass losses ranging from 15 to 60 percent of total weight lost — a wide range explained by population differences, drug, and how researchers measure "lean mass" (which includes organs, water, and bone, not just skeletal muscle). MRI-based studies suggest the changes are largely adaptive — the kind of muscle loss that accompanies any significant weight loss, not a unique drug effect. Resistance training and adequate protein intake (1.2-1.6 g/kg of body weight is the typical recommendation) substantially blunt the effect. Older patients are more vulnerable to sarcopenia and need more careful monitoring.
The compounding situation
The compounding question deserves its own paragraph because it shapes what patients actually receive in 2026. With semaglutide and tirzepatide off the FDA shortage list, 503A pharmacies legally can no longer mass-produce identical-formulation knockoffs. Some are continuing to produce "modified" versions (with added B vitamins or levocarnitine) to argue continued legality — claims that are theoretical and untested. The FDA in 2024 warned about overdoses, including documented cases of patients injecting 0.5 mL when instructions specified 0.05 mL — a tenfold dosing error. A Partnership for Safe Medicines review found 239 semaglutide and tirzepatide shipments produced at unregistered overseas factories. If cost is the barrier to brand-name GLP-1s, the safer alternatives are patient-assistance programs, switching to liraglutide (cheaper, generic-track), or revisiting whether non-pharmacological interventions can produce enough benefit alone.
Important: The information in this article is for educational purposes only and is not medical advice. Peptide therapy carries real risks. Decisions about prescription weight-loss drugs, off-label use, or research-grade peptides must be made with a licensed clinician who knows your medical history. Do not start, stop, or change any peptide regimen based on what you read in an article.
Frequently Asked Questions
Which weight-loss peptide produces the most weight loss?
By the longest-term published data, tirzepatide currently leads among FDA-approved options, with the SURMOUNT-1 trial showing 20.9 percent mean loss at 72 weeks on the 15 mg dose, and the head-to-head SURMOUNT-5 trial confirming superiority over semaglutide (20.2 percent vs 13.7 percent). The investigational triple-agonist retatrutide and the cagrilintide-semaglutide combination CagriSema (22.7 percent in REDEFINE 1) are pushing the ceiling further as they move through approval pathways. None of these comparisons account for which patient phenotype responds best to which drug.
What happens if I stop taking semaglutide or tirzepatide?
Most people regain a substantial portion of lost weight. The STEP 1 extension followed semaglutide patients for a year after discontinuation; they regained roughly two-thirds of their weight loss, with net loss falling from 17.3 percent to 5.6 percent, and most cardiometabolic improvements drifted back toward baseline. Researchers have framed the implication clearly: obesity behaves like a chronic condition, and these medications treat it only as long as you take them.
Are compounded peptides safe to use instead of brand-name versions?
Compounded versions are not FDA-reviewed for safety, quality, or effectiveness. With tirzepatide off the FDA shortage list as of December 2024 and semaglutide as of February 2025, mass production of identical-formulation compounds is no longer legally permitted. The FDA has warned about dosing errors (including tenfold overdoses) and overseas sourcing of active ingredients from unregistered factories. If brand-name cost is prohibitive, patient assistance programs and switching to generic-track alternatives like liraglutide are the safer paths.
Will GLP-1 drugs make me lose muscle?
Some lean mass loss is unavoidable with any significant weight loss, drug-induced or otherwise. Studies show lean mass loss ranging from 15 to 60 percent of total weight lost on GLP-1 drugs, with MRI evidence suggesting the changes are largely adaptive — meaning muscle quality often improves even as raw mass declines. Resistance training two to three times weekly and protein intake around 1.2-1.6 g per kg of body weight substantially blunt sarcopenic effects. Older patients face higher baseline sarcopenia risk and warrant closer monitoring.
Can my regular doctor prescribe peptides like tesamorelin or CJC-1295?
FDA-approved options — semaglutide, tirzepatide, liraglutide — are routinely prescribed by primary-care physicians, endocrinologists, and obesity-medicine specialists. Tesamorelin (Egrifta) is FDA-approved only for HIV-associated lipodystrophy, so off-label prescription happens almost exclusively in specialty settings. CJC-1295, ipamorelin, and AOD-9604 are not FDA-approved for weight loss; they're prescribed by specialty wellness or anti-aging clinics, typically through compounding pharmacies. The further you move from FDA-approved indications, the more important it becomes to verify your provider's credentials and the sourcing of the actual peptide.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.