One receptor or two: why the pharmacology matters
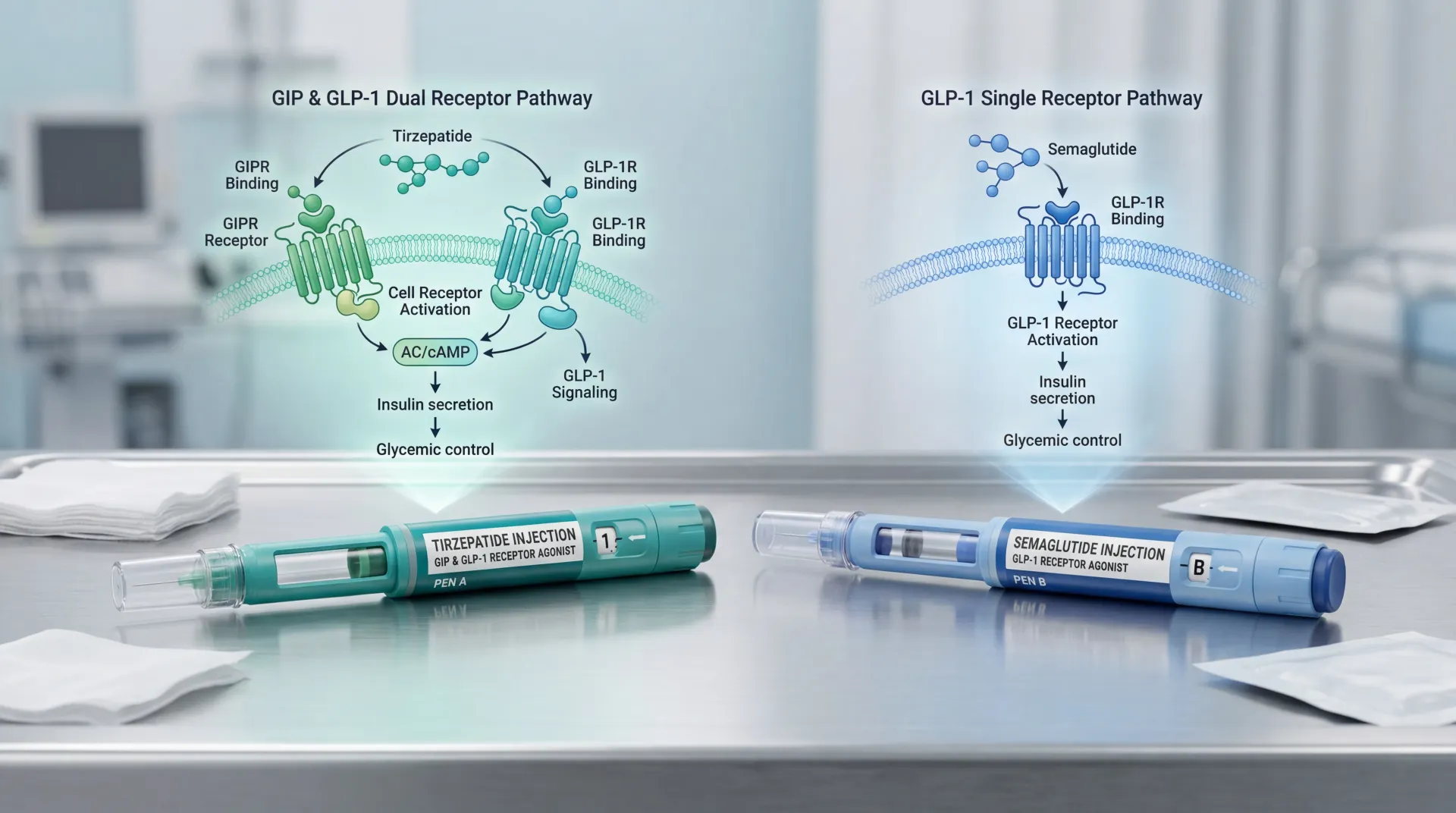
Your gut releases two hormones after a meal that tell your brain you're full and signal your pancreas to produce insulin: GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). Semaglutide -- the active ingredient in both Ozempic and Wegovy -- mimics GLP-1 only. Tirzepatide, sold as Mounjaro for type 2 diabetes and Zepbound for obesity, activates receptors for both GIP and GLP-1. That dual action is the single biggest pharmacological difference between these medications.
A useful analogy: semaglutide turns up one channel -- GLP-1 -- and gets strong results from that channel alone. Tirzepatide turns up two channels at once, and the combined signal produces effects that neither channel achieves on its own.
The molecular details are counterintuitive. Tirzepatide was engineered by building GLP-1 activity into the GIP molecule's backbone, not the other way around. The resulting drug binds the GIP receptor with the same affinity as the body's own GIP hormone but binds the GLP-1 receptor with roughly 5-fold weaker affinity than native GLP-1. Researchers Francis Willard, Kyle Sloop, and colleagues at Eli Lilly characterized this as an "imbalanced" agonist -- it leans harder on GIP than GLP-1.
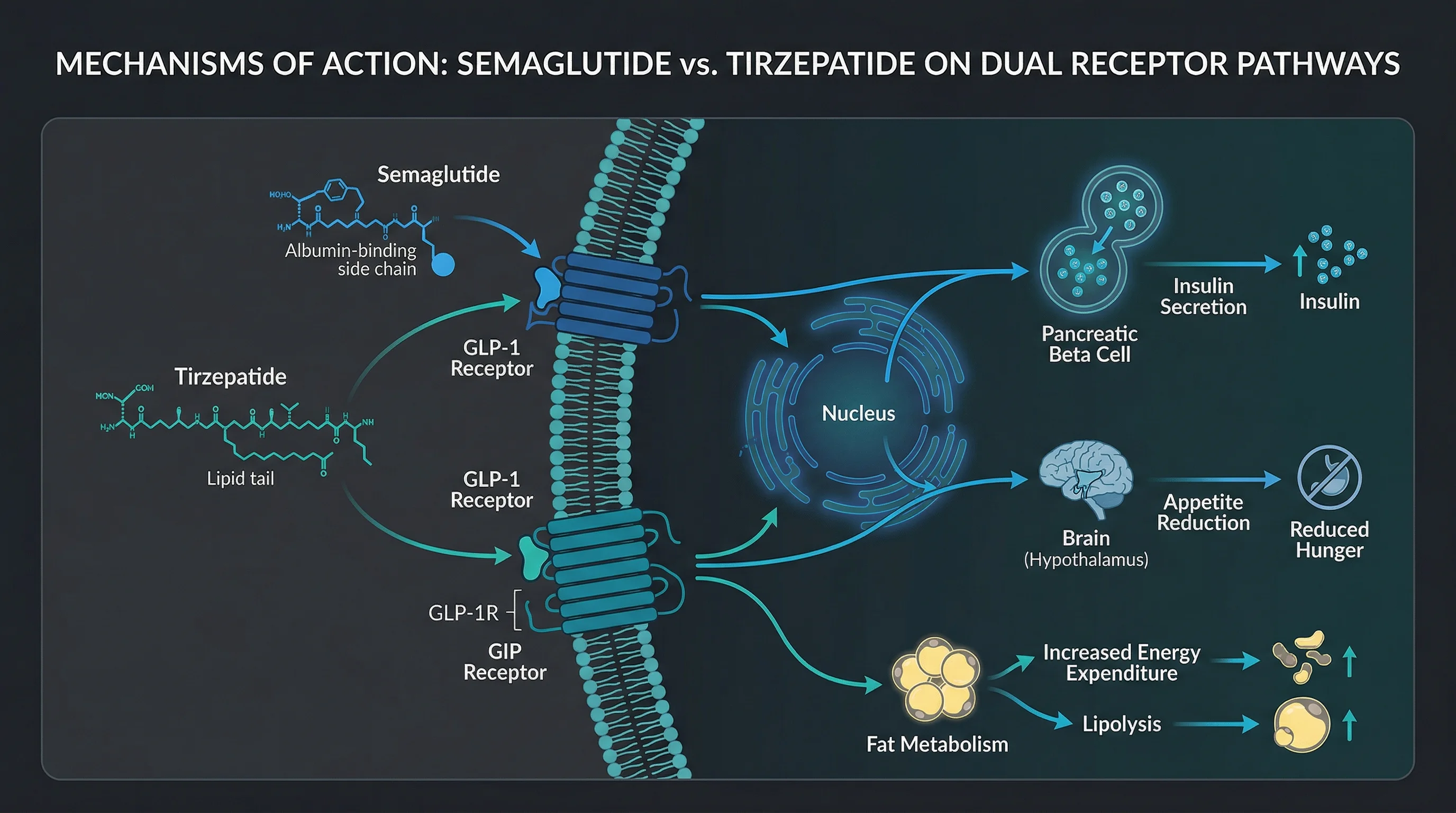
That imbalance may actually be advantageous. The Willard et al. study published in JCI Insight found that tirzepatide shows "biased agonism" at the GLP-1 receptor -- it preferentially triggers cAMP signaling (which drives insulin release) while largely avoiding beta-arrestin recruitment (which pulls the receptor off the cell surface and dampens the signal). In practical terms, tirzepatide gets more insulin-releasing activity per unit of GLP-1 receptor engagement than semaglutide does, and the GIP receptor engagement adds effects on fat tissue metabolism and appetite that GLP-1 alone doesn't fully address.
For patients, the mechanistic difference boils down to a concrete question: does activating two pathways produce meaningfully better outcomes than activating one? The trial data from the past two years answers that.
20% vs 14%: what the head-to-head trial found
Until 2025, comparisons between tirzepatide and semaglutide relied on cross-referencing separate clinical trials -- the SURMOUNT series for tirzepatide and the STEP series for semaglutide. Both showed impressive weight loss, but comparing results across different trials with different patient populations is imprecise by nature. SURMOUNT-5 changed that.
Published in the New England Journal of Medicine in May 2025, SURMOUNT-5 was the first head-to-head randomized trial pitting tirzepatide directly against semaglutide. The study enrolled 751 adults with obesity but without type 2 diabetes at 32 sites across the U.S. and Puerto Rico. Participants were randomly assigned to receive either the maximum tolerated dose of tirzepatide (10 or 15 mg) or semaglutide (1.7 or 2.4 mg) via weekly injection for 72 weeks. Both groups received diet and exercise counseling.
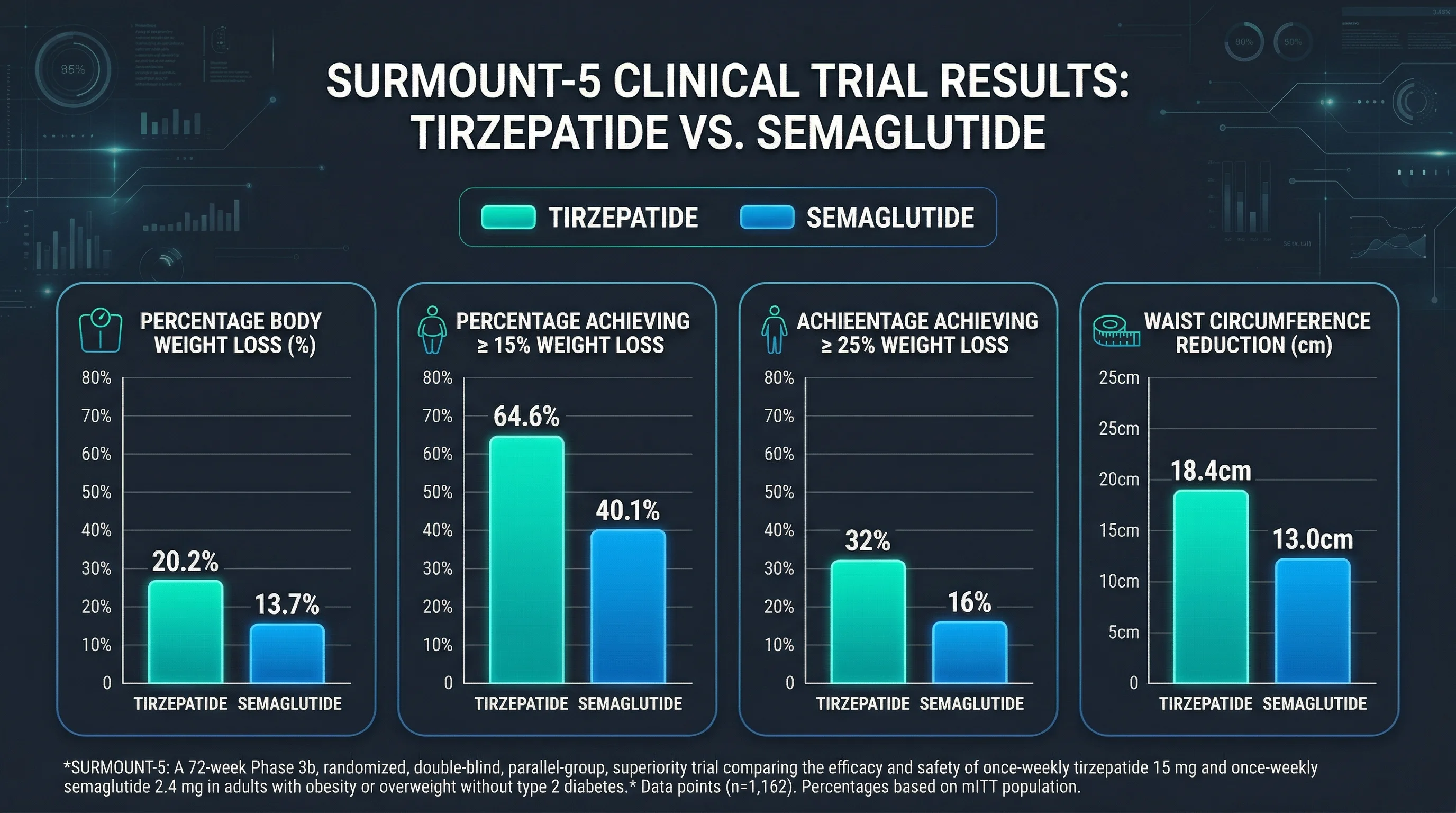
At 72 weeks, participants on tirzepatide lost an average of 20.2% of their body weight compared to 13.7% for semaglutide -- a 47% greater relative weight loss. In absolute terms, that translated to 50.3 pounds (22.8 kg) lost with tirzepatide versus 33.1 pounds (15.0 kg) with semaglutide.
The secondary endpoints told a consistent story. 64.6% of tirzepatide users achieved at least 15% body weight loss versus 40.1% on semaglutide. And nearly a third (32%) of tirzepatide participants lost at least 25% of their body weight, compared with 16% on semaglutide.
Dr. Louis Aronne, the principal investigator and director of the Comprehensive Weight Control Center at Weill Cornell Medicine, noted that the results were "consistent with -- in fact, almost identical to -- what we've seen in trials in which these drugs were evaluated independently." The head-to-head data confirmed what cross-trial comparisons had suggested rather than producing any surprises.
A separate meta-analysis pooling data from two randomized controlled trials and five retrospective cohort studies by Ahmad Bin Aamir, Rabia Latif, and colleagues at Imam Abdulrahman Bin Faisal University found tirzepatide produced a mean 4.23 kg greater weight loss than semaglutide (95% CI: 3.22-5.25, p<0.01). They identified a dose-response relationship too: doses above 10 mg showed a mean difference of 6.50 kg versus 3.89 kg for doses at or below 10 mg.
Real-world data backs up the trial findings, though with smaller effect sizes -- as you'd expect outside controlled conditions. A retrospective cohort study by Huong Trinh, Anthony Donovan, and Carrie McAdam-Marx at the University of Nebraska Medical Center examined 945 patients in an ambulatory care setting and found tirzepatide users lost 5.3% of body weight at 6 months versus 2.7% for semaglutide (p<0.001). 48.6% of tirzepatide patients lost more than 5% of their body weight compared with 26.0% on semaglutide. One finding worth flagging: the advantage was most pronounced in patients without diabetes. In patients with diabetes, the difference between the two drugs was not statistically significant at 6 months.
Both drugs produce medically meaningful weight loss. Semaglutide's 13.7% average in SURMOUNT-5 exceeds what any prior obesity medication achieved. Tirzepatide goes further. But 13.7% is not a consolation prize -- five years ago, that number on its own would have been front-page medical news.
Beyond the scale: cardiovascular risk and metabolic health
Weight loss alone doesn't capture the full clinical picture. Both tirzepatide and semaglutide affect cardiovascular risk factors, insulin resistance, and metabolic markers in ways that matter separately from the number on the scale.
Semaglutide has the stronger cardiovascular evidence right now because of the SELECT trial -- a dedicated cardiovascular outcomes trial that demonstrated a 20% reduction in major adverse cardiovascular events (MACE-3) compared with placebo in people with obesity and established cardiovascular disease. That trial led the FDA to approve Wegovy for cardiovascular risk reduction in March 2024. Tirzepatide doesn't have an equivalent completed outcomes trial yet, though one is underway.
A post-hoc analysis of SURMOUNT-5 data does provide the first direct comparison of cardiovascular risk reduction between the two drugs. Mamas Mamas, Harold Bays, and colleagues used the Framingham risk equation to calculate predicted 10-year cardiovascular disease risk among SURMOUNT-5 participants without pre-existing cardiovascular disease. The average baseline 10-year CVD risk was 9.3%. After 72 weeks, tirzepatide was associated with an absolute reduction of 2.4% in predicted 10-year CVD risk compared with 1.4% for semaglutide (p<0.001).
Scaled to the roughly 85 million Americans who would meet treatment eligibility criteria, the researchers estimated approximately 2 million cardiovascular events could potentially be prevented over 10 years with tirzepatide versus 1.15 million with semaglutide. Those are modeled projections, not observed outcomes -- but the gap between the two is substantial.
Tirzepatide has also shown particular promise for heart failure. In a separate trial of patients with heart failure with preserved ejection fraction (HFpEF) and obesity, worsening heart failure events occurred in 8.0% of the tirzepatide group versus 14.2% in the placebo group.
Waist circumference reduction -- a strong independent predictor of cardiovascular risk -- also favored tirzepatide: -18.4 cm versus -13.0 cm for semaglutide over 72 weeks. That 5.4 cm difference in abdominal fat loss may partly explain the divergence in predicted cardiovascular risk.
For patients weighing these two medications, the cardiovascular picture has an important wrinkle. Semaglutide has proven cardiovascular benefit from a completed outcomes trial. Tirzepatide shows larger improvements in cardiovascular risk factors but hasn't finished its own outcomes trial yet. If you have established heart disease, the proven SELECT data for semaglutide carries real weight. If your concern is primary prevention through maximum risk factor improvement, tirzepatide's larger effects on weight, waist circumference, and predicted CVD risk make a strong case.
The GI question: side effects are more similar than different
Both medications cause gastrointestinal side effects. GLP-1 receptor activation slows gastric emptying, and that produces nausea, vomiting, and diarrhea in a meaningful percentage of patients. The question is whether tirzepatide's additional GIP receptor activation makes those side effects better or worse.
The short answer: roughly the same, with a slight tolerability edge for tirzepatide overall.
| Side effect | Tirzepatide | Semaglutide | Source |
|---|---|---|---|
| Nausea | 17-22% | 18% | SURPASS-2 |
| Diarrhea | 13-16% | 12% | SURPASS-2 |
| Vomiting | 6-10% | 8% | SURPASS-2 |
| Decreased appetite | 7-9% | 5% | SURPASS-2 |
| GI-related discontinuation | 2.7% | 5.6% | SURMOUNT-5 |
| Overall adverse event discontinuation | 6.1% | 8.0% | SURMOUNT-5 |
Individual symptom rates are strikingly similar. The difference shows up in discontinuation rates. In SURMOUNT-5, 6.1% of tirzepatide participants stopped treatment due to adverse events compared with 8.0% on semaglutide. GI adverse events specifically caused discontinuation in 2.7% of the tirzepatide group versus 5.6% of the semaglutide group. Fewer people quit tirzepatide because of stomach problems, even though the rates of individual symptoms are comparable.
Dr. Supriya Rao, a board-certified gastroenterologist and obesity medicine provider, notes that side effect experiences are "very individualized". A patient who can't tolerate semaglutide might do fine on tirzepatide, and the reverse is also true.
Both drugs carry the same class-level warnings: thyroid C-cell tumor risk (based on animal studies), pancreatitis, gallbladder problems, and kidney issues related to dehydration from vomiting and diarrhea. The FDA has also added warnings about depression and suicidal thoughts to both drug classes, though large-scale pharmacovigilance data hasn't established a causal link.
The most consistent advice from clinicians: GI side effects peak during dose escalation, not at maintenance doses. Most adverse events were mild to moderate in severity and occurred during dose escalation in both SURMOUNT-5 treatment groups. Slower titration often reduces nausea substantially, and most patients who push through the first few weeks at a new dose find symptoms fade.
What you'll pay: cost, coverage, and access
Sticker prices for these medications are high enough to override clinical data in many treatment decisions.
| Medication | Brand names | FDA-approved for | Average retail price | Direct-to-consumer cash price |
|---|---|---|---|---|
| Tirzepatide | Mounjaro (T2D), Zepbound (obesity) | Type 2 diabetes, chronic weight management, OSA | ~$1,480/month | $299-$449/month (Lilly vials) |
| Semaglutide | Ozempic (T2D), Wegovy (obesity) | Type 2 diabetes, chronic weight management, CV risk reduction | ~$1,846/month | $349-$499/month (Novo Nordisk) |
Those retail prices rarely reflect what patients actually pay, but they illustrate the baseline cost problem. Insurance coverage is the dominant variable, and it depends almost entirely on why the medication is prescribed.

With a diabetes diagnosis, insurance is the least complicated part of the process. More than 90% of commercial and Medicare Part D plans cover GLP-1 medications when prescribed for type 2 diabetes. Prior authorization is common but approval rates are high.
The weight loss indication is where coverage falls apart. 36% of employers now cover GLP-1s for weight loss, up from about 25% in 2023. Progress, but two-thirds of employer plans still exclude it -- and some employers who previously covered these drugs have reversed course because of cost projections.
Medicare is a hard no. Federal law explicitly excludes weight-loss medications from Part D coverage. A proposed rule change in late 2024 would have opened the door, but the current administration declined to finalize it. A pilot program -- the BALANCE Model, a Medicare GLP-1 payment demonstration -- is scheduled to begin in July 2026, but that's not standard coverage.
Medicaid is a patchwork. Fee-for-service Medicaid covers weight-loss medication in only 15 states, with a handful of others covering them through managed care plans.
Both manufacturers have responded with direct-to-consumer programs. Eli Lilly cut cash prices for Zepbound vials to $299-$449 per month depending on dose. Novo Nordisk offers the two lowest Wegovy doses at $199/month for new patients' first two months, then $349/month. Starting in January 2027, Novo Nordisk will reduce the U.S. list price of both Wegovy and Ozempic to $675 per month.
More than 20 states are considering legislation related to GLP-1 coverage for weight loss, though most bills are still in committee as of early 2025. The direction is toward broader access, but it's moving slowly.
Picking the right one for your situation
Tirzepatide produces more weight loss, on average, than semaglutide. The data on that point is clear. But average weight loss is one variable in a decision with several.
| Consider tirzepatide (Mounjaro/Zepbound) if... | Consider semaglutide (Ozempic/Wegovy) if... |
|---|---|
| Maximum weight loss is the primary goal | You have established cardiovascular disease (proven CV benefit from SELECT trial) |
| You have type 2 diabetes and want dual metabolic benefit | Your insurance covers semaglutide but not tirzepatide |
| Previous GI side effects with semaglutide were intolerable | You want an oral option (Rybelsus available for T2D) |
| You're focused on insulin sensitivity improvement | You prefer more long-term safety data (semaglutide has been on market longer) |
| Heart failure with preserved ejection fraction is a concern | Cost without insurance is the deciding factor (Wegovy direct pricing is competitive) |

Both medications require a prescription from a healthcare provider who knows your medical history. That prescribing conversation should cover not just efficacy data but insurance coverage (which can override clinical preference), existing cardiovascular conditions, diabetes status, and your tolerance for GI side effects during dose escalation.
There are real gaps in what we know. The longest head-to-head data comes from 72-week trials. Neither drug has published data on what happens when patients switch from one to the other. Long-term weight maintenance data beyond two years is limited for both. Dr. Aronne's comment about the next generation of medications captures the current state well: "Even though drugs like tirzepatide and semaglutide work really well, better than anything we have ever seen, we still have people who don't respond to them."
The next generation is already in trials. Eli Lilly's retatrutide -- a triple GIP/GLP-1/glucagon agonist -- showed even larger weight loss numbers in early data. For now, tirzepatide and semaglutide are the two strongest options available by prescription, and choosing between them comes down to your medical profile, insurance situation, and what your doctor recommends after reviewing both.
Frequently asked questions
Can I switch from Ozempic or Wegovy to Mounjaro or Zepbound?
Yes, with your prescriber's guidance. Switching between GLP-1 medications is common in clinical practice, though there are no formal studies on the switch itself. Your doctor will typically restart the dose escalation process with the new medication to minimize GI side effects. Insurance authorization for the new medication may require documentation of why the switch is medically necessary.
Is the weight loss permanent, or does it come back when you stop?
Clinical trials consistently show partial weight regain after stopping both medications. The SURMOUNT series data indicates that a substantial portion of lost weight returns within a year of stopping tirzepatide, and the STEP series shows similar patterns for semaglutide. Current medical guidelines treat obesity as a chronic condition requiring ongoing management -- most patients who respond well will need to stay on the medication long-term.
Are compounded versions of tirzepatide or semaglutide safe?
The FDA has issued multiple warnings about compounded versions of both medications. Compounded drugs are not FDA-approved and are not held to the same testing standards for safety, efficacy, and dosing consistency. Adverse events linked to purity and dosing problems have been reported. If cost is the barrier, the manufacturer vial programs ($299-$449/month for Zepbound, starting at $199/month for Wegovy) are a regulated lower-cost alternative.
Do these medications work for people without much weight to lose?
FDA approval for weight management requires a BMI of 30 or higher, or 27 or higher with at least one weight-related medical condition. Trial participants had average BMIs around 35-40. The percentage of body weight lost tends to be proportionally similar regardless of starting weight -- someone starting at 200 lbs can expect a similar percentage loss as someone starting at 300 lbs. Neither medication is approved or recommended for cosmetic weight loss in people at a healthy weight.
Why does tirzepatide produce more weight loss if it has weaker GLP-1 receptor activity?
This gets at the core pharmacological insight. Tirzepatide compensates for its weaker GLP-1 receptor binding by also activating GIP receptors, which contribute to appetite suppression and fat metabolism through a separate pathway. The biased signaling at the GLP-1 receptor also means tirzepatide may extract more insulin-releasing activity per unit of receptor engagement. Two partially activated pathways, it turns out, can exceed the effect of one fully activated pathway.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.