A pill that does what injections did
On December 22, 2025, the FDA approved Wegovy tablets (semaglutide 25mg) for weight management in adults. It was the first oral GLP-1 receptor agonist cleared for treating obesity in the United States. Before that date, anyone who wanted semaglutide for weight loss had to inject it once a week.
Oral semaglutide had existed since 2019 under the brand name Rybelsus, but only at lower doses (up to 14mg) and only for type 2 diabetes. The December 2025 approval covered a higher-dose tablet aimed at weight reduction, carrying two overlapping indications: reducing major cardiovascular events in adults with established heart disease and obesity or overweight, and reducing excess body weight in adults with obesity (BMI 30+) or overweight (BMI 27+) with at least one weight-related comorbidity.
Wegovy tablets come in four strengths -- 1.5mg, 4mg, 9mg, and 25mg -- to allow gradual dose escalation. The approval rested on the Phase III OASIS 4 trial, which enrolled 307 adults without diabetes who had overweight or obesity. Commercial availability began in early January 2026.
A pill version of an existing drug wouldn't normally generate this much interest. But needle aversion keeps a real number of patients from starting GLP-1 treatment at all. Some delay for months. Others refuse outright. A daily pill removes that specific barrier. Whether it delivers the same weight loss as the injection is a separate and more complicated question.
The engineering trick that makes oral semaglutide possible
Putting a peptide drug into a pill sounds simple. It is not. Peptides are proteins, and your digestive system exists to dismantle proteins. Stomach acid, pepsin, and the thick mucus lining of the GI tract all work against a peptide that needs to reach your bloodstream intact. This is why insulin still requires injection over a century after its discovery -- attempts at oral insulin have been failing since 1923.
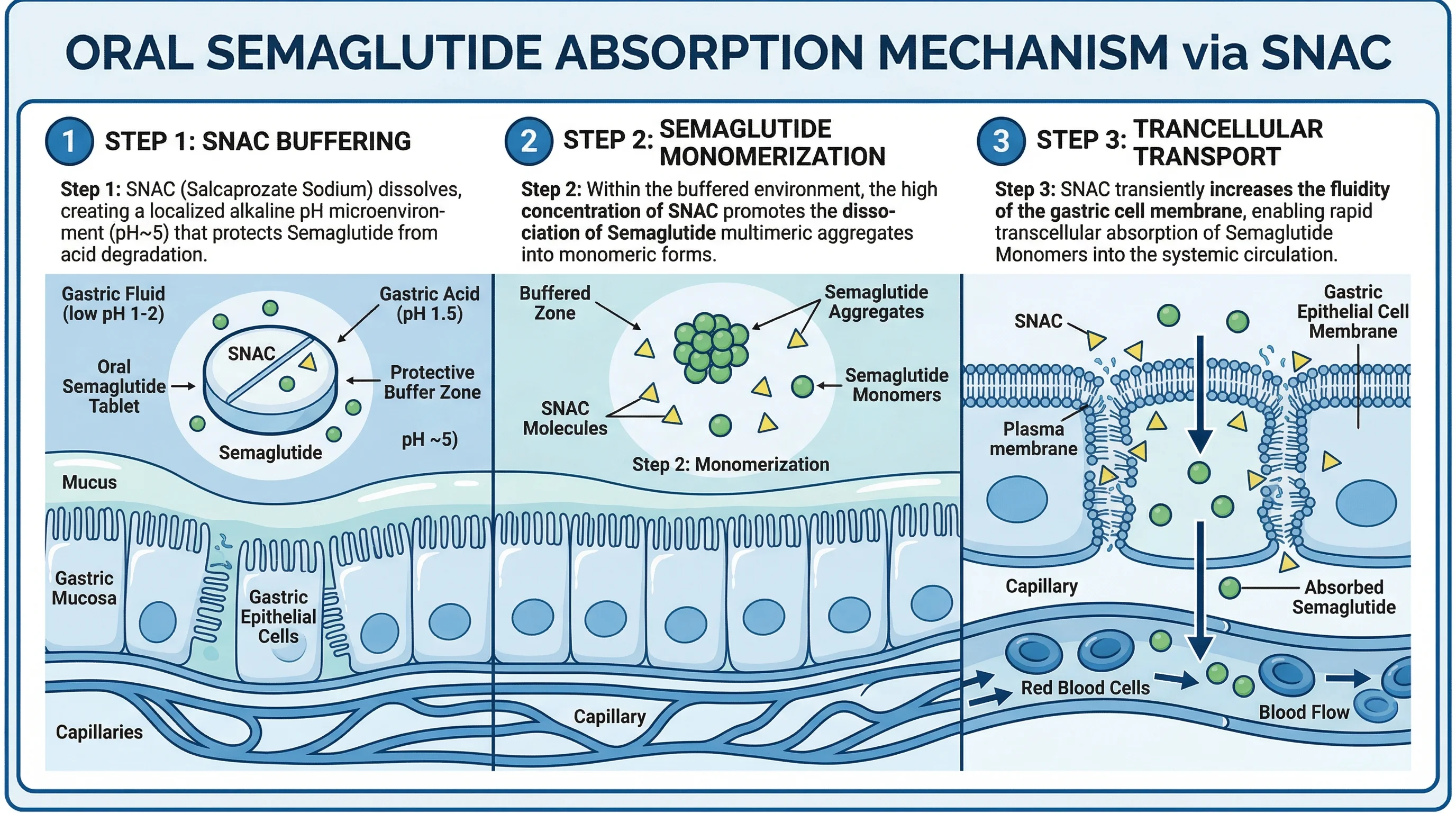
Semaglutide gets around this through a molecule called SNAC (sodium N-[8-(2-hydroxybenzoyl)amino] caprylate). SNAC works as a chemical bodyguard doing three things at once. First, as the tablet dissolves in the stomach, SNAC raises the local pH around the dissolving tablet, which slows the conversion of pepsinogen to pepsin -- the enzyme that would otherwise chop semaglutide into useless fragments. Second, SNAC prevents semaglutide molecules from clumping together (oligomerization) by changing the polarity of the surrounding fluid. Individual semaglutide molecules are small enough to cross cell membranes; clumps are not. Third, SNAC gets incorporated into the lipid membrane of nearby stomach cells and temporarily fluidizes it, opening a window for semaglutide molecules to pass through the gastric wall and into the bloodstream.
The stomach, not the intestine, is where oral semaglutide gets absorbed. Scintigraphic imaging in human volunteers and pyloric ligation studies in dogs both confirmed that absorption occurs through the gastric lining. Most oral drugs are absorbed in the intestines, making semaglutide's stomach-based absorption unusual.
The membrane-fluidizing effect is transient and fully reversible. SNAC itself has been used in pharmaceutical applications for decades and received FDA "generally recognized as safe" (GRAS) status in 2014 when it was first approved in an oral vitamin B12 formulation.
Even with all this engineering, oral semaglutide achieves bioavailability of roughly 0.8% under recommended dosing conditions. By contrast, subcutaneous semaglutide has bioavailability of about 89%. That is not a typo -- less than 1% of the drug you swallow actually reaches your bloodstream. The rest gets destroyed by digestion. This is why the oral dose (25mg daily) is far larger than the injection dose (2.4mg weekly). The pill compensates for massive absorption losses with a massive dose increase.
What saves this from being a dealbreaker is semaglutide's long half-life. Both oral and subcutaneous semaglutide share an elimination half-life of approximately one week. Daily dosing with a long half-life means the drug accumulates to stable steady-state levels over 4-5 weeks, smoothing out the day-to-day variability in absorption. Your Tuesday dose might absorb slightly differently from your Wednesday dose, but the cumulative effect stays consistent.
Oral vs. injection: what the trials show
The data that convinced the FDA came from two trials in Novo Nordisk's OASIS program.
OASIS 1 tested the higher 50mg dose. Over 68 weeks, participants taking oral semaglutide 50mg lost an average of 15.1% of their body weight compared to 2.4% in the placebo group -- a gap of 12.7 percentage points. Among those on the drug, 85% lost at least 5% of their body weight, 69% lost at least 10%, and 34% reached the 20% threshold.
OASIS 4 tested the 25mg dose that the FDA ultimately approved for Wegovy tablets. Over 64 weeks, the oral 25mg group lost 13.6% of body weight versus 2.2% for placebo -- a gap of 11.4 percentage points. About 76.3% achieved at least 5% weight loss compared to 31.3% on placebo.
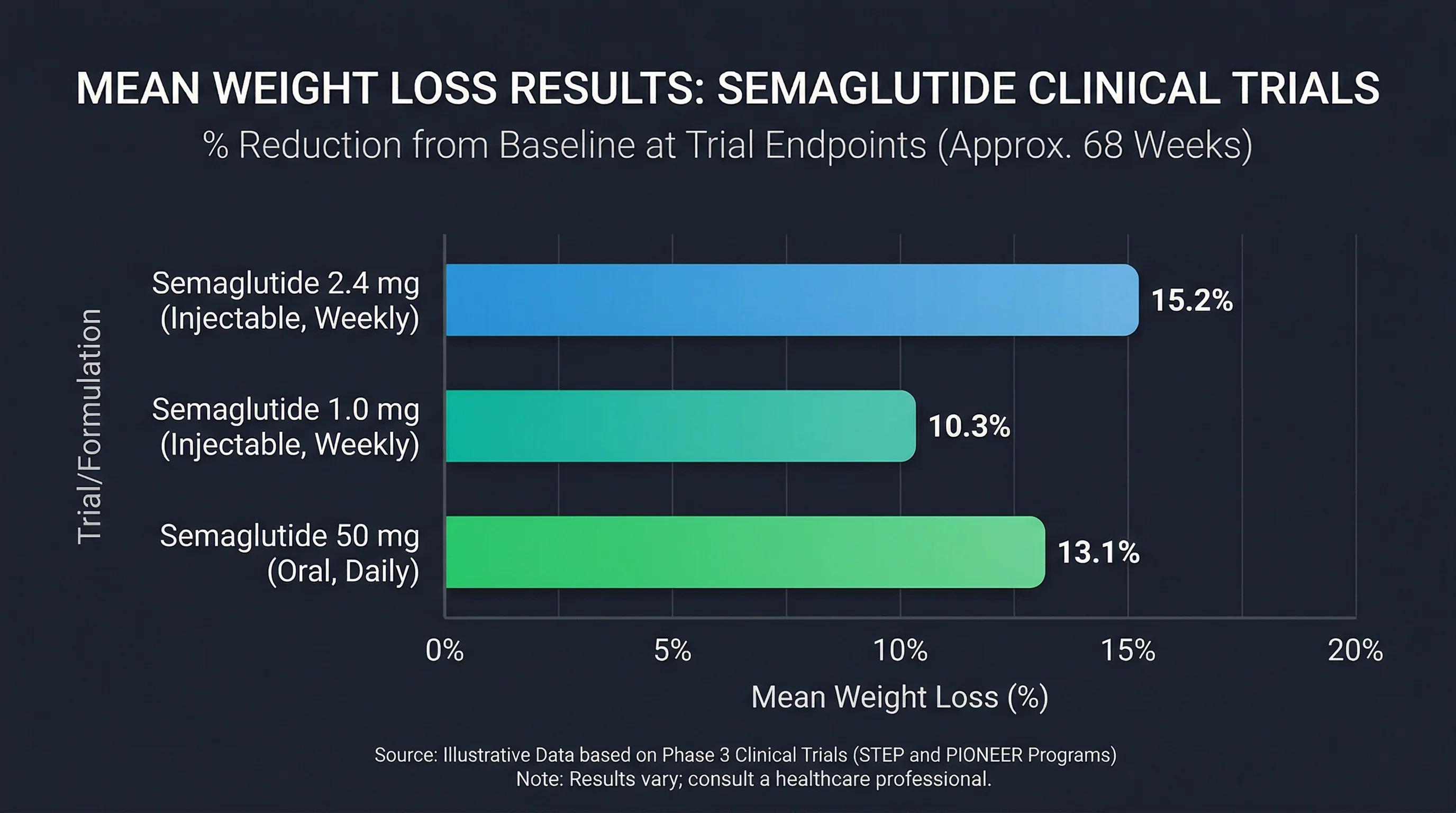
The comparison that matters most: how does the pill stack up against the injection? Dr. Sean Wharton and colleagues noted in their OASIS 4 analysis that the weight loss with oral 25mg (11.4pp above placebo) was comparable to both oral 50mg in OASIS 1 (12.7pp above placebo) and subcutaneous 2.4mg in STEP 1 (12.4pp above placebo). These are cross-trial comparisons, which carry inherent limitations, but the pattern is consistent: the approved oral dose produces weight loss in the same range as the approved injectable dose.
| Trial | Formulation | Dose | Duration | Weight loss vs. placebo |
|---|---|---|---|---|
| OASIS 4 | Oral tablet | 25mg daily | 64 weeks | -11.4 percentage points |
| OASIS 1 | Oral tablet | 50mg daily | 68 weeks | -12.7 percentage points |
| STEP 1 | Subcutaneous injection | 2.4mg weekly | 68 weeks | -12.4 percentage points |
For blood sugar control specifically, the picture is slightly different. In diabetes-focused trials, oral semaglutide reduced HbA1c by 1.0-1.4% in the PIONEER program, while subcutaneous semaglutide achieved 1.5-1.8% reductions in the SUSTAIN trials. The injectable version holds a modest edge for glycemic control, likely because of more consistent plasma concentrations.
Cardiovascular protection is part of the picture too. The SUSTAIN-6 trial showed subcutaneous semaglutide reduced major adverse cardiovascular events by 26% in patients with type 2 diabetes and elevated CV risk. The oral form's approval for CV risk reduction extends this benefit to the pill format, though the supporting data comes primarily from the injection trials.
Who qualifies and what it costs
The FDA label for oral Wegovy follows the same weight criteria as injectable Wegovy: adults with a BMI of 30 or higher, or a BMI of 27 or higher with at least one weight-related medical condition. Those conditions include hypertension, type 2 diabetes, high cholesterol, and obstructive sleep apnea. For the cardiovascular indication, the requirement is established CV disease (prior heart attack, stroke, or peripheral artery disease) plus obesity or overweight.
Meeting the medical criteria and getting your insurance to pay are two different problems.
A Tufts Medical Center analysis of 18 major US commercial health plans found that 7 didn't even issue a public coverage policy for semaglutide. Among the 11 that did, requirements varied widely. Most imposed BMI thresholds matching the FDA label, but two set stricter limits. Eight required documented diet and exercise efforts, and seven required enrollment in a behavioral modification program before approving coverage. One plan demanded patients try a different weight loss medication first.

Medicare is its own mess. Federal law has historically prohibited Medicare from covering drugs used specifically for weight loss. The cardiovascular indication for Wegovy opened a pathway -- KFF estimates approximately 3.6 million Medicare beneficiaries (about 7%) have established cardiovascular disease plus obesity or overweight and could qualify under this indication. But at a list price of roughly $1,300 per month, even covered beneficiaries face monthly out-of-pocket costs of $325 to $430 before hitting the annual spending cap.
Some relief may come from price negotiations. CMS selected Ozempic, Rybelsus, and Wegovy for its second round of Medicare drug price negotiations, with negotiated prices taking effect in 2027. Until then, the cost barrier is real. Anyone considering oral Wegovy should check their specific plan's formulary and prior authorization requirements before assuming coverage.
| Coverage type | Status | Key requirements |
|---|---|---|
| Commercial insurance | Varies by plan | BMI thresholds, diet/exercise documentation, sometimes behavioral program |
| Medicare Part D (CV indication) | Eligible for ~3.6M beneficiaries | Established CV disease + obesity/overweight; plan must add to formulary |
| Medicare Part D (weight loss only) | Not covered | Federal law prohibits coverage for weight loss indication alone |
Side effects: what to actually expect
GI problems dominate the side effect profile, and the numbers are higher than many patients anticipate. In OASIS 4, 74% of participants on oral semaglutide 25mg reported gastrointestinal adverse events compared to 42.2% on placebo. The higher 50mg dose in OASIS 1 pushed that to 80% versus 46% on placebo. Nausea, vomiting, and diarrhea are the most common complaints.
Those numbers need context, though. The majority of these events were mild to moderate and transient -- they peaked during the dose escalation period and eased as the body adjusted. In OASIS 4, treatment discontinuation due to adverse events was 7% with oral semaglutide versus 6% with placebo. A 1 percentage point difference in dropout rates suggests that while side effects are common, they rarely get severe enough to stop treatment.
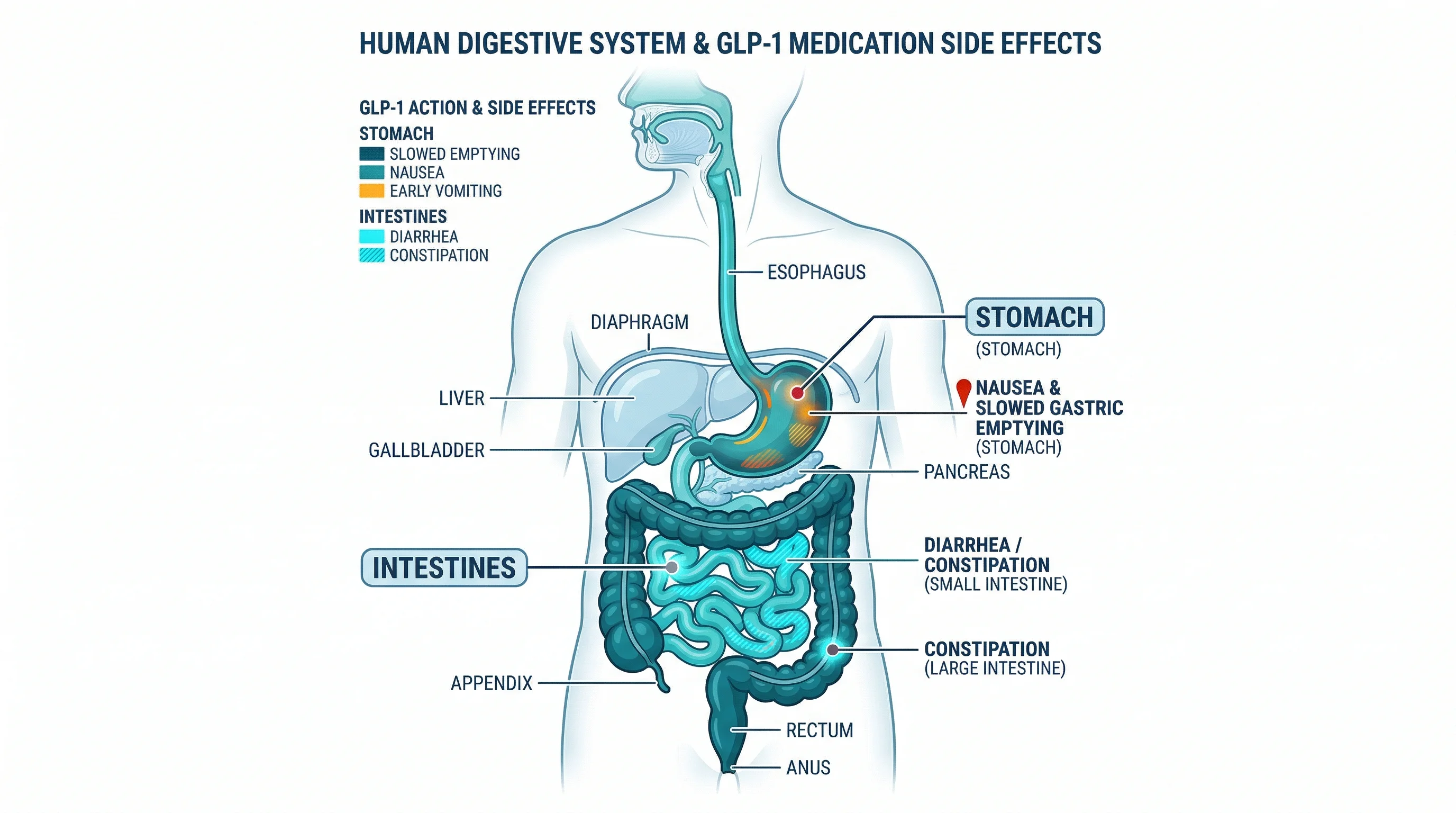
Beyond the GI tract, semaglutide carries several less common but more serious risks. The prescribing information includes a boxed warning for thyroid C-cell tumors. It is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2, and in pregnancy. Other documented risks include acute pancreatitis, acute kidney injury, and gallbladder disease.
One effect with practical implications beyond discomfort: semaglutide delays gastric emptying. This is part of how it drives weight loss (you feel full longer), but it has raised concerns in surgical settings. Patients scheduled for procedures requiring anesthesia should tell their surgical team about their semaglutide use, since delayed gastric emptying can increase the risk of pulmonary aspiration under anesthesia.
| Side effect category | Frequency (OASIS 4) | Severity |
|---|---|---|
| Any GI event (nausea, vomiting, diarrhea) | 74% (vs 42% placebo) | Mostly mild-moderate, transient |
| Treatment discontinuation from side effects | 7% (vs 6% placebo) | -- |
| Thyroid C-cell tumors | Rare (boxed warning) | Serious |
| Acute pancreatitis | Rare | Serious |
| Gallbladder events | Uncommon | Moderate to serious |
The daily reality of the Wegovy pill
Taking oral semaglutide is not like popping a vitamin. The dosing protocol demands specific conditions. You must take the tablet on an empty stomach with no more than 120mL of water (about 4 ounces), then wait at least 30 minutes before eating, drinking anything else, or taking other oral medications. This is a clinical requirement, not a suggestion. Food and excess fluid reduce absorption, and given that only 0.8% of the drug gets absorbed under ideal conditions, any further reduction can undermine the whole point.
Why so finicky? The SNAC mechanism depends on direct contact between the dissolving tablet and the stomach lining. Food creates a physical barrier. More water dilutes the localized pH buffer that SNAC creates. Other medications taken at the same time can interfere with semaglutide absorption. The 30-minute window gives the tablet enough time to erode, release its SNAC shield, and move semaglutide molecules across the gastric wall.
This daily ritual contrasts with injectable Wegovy's once-weekly schedule. With the injection, you pick a day, inject, and forget about it for a week. No food timing, no water volume limits. BMO Capital Markets analysts noted that the Wegovy pill's food restriction "is likely to limit uptake" once competing oral GLP-1s with more convenient dosing reach the market.

Dose escalation also takes patience. For the weight management indication, the climb from starting dose to the full 25mg target takes several months to minimize GI side effects. You start low and increase gradually. The pill is not delivering its full weight-loss effect from day one. Meaningful results take 3-4 months to become visible, and the OASIS trials measured outcomes at 64-68 weeks.
Physical function improvements go beyond the scale. In OASIS 4, patients on oral semaglutide showed physical function scores improving by 16 points versus 8 points on placebo, measured by a validated quality-of-life tool. Patients reported finding daily physical activities easier as they lost weight. Combined with improvements in waist circumference, blood pressure, and lipid levels, the clinical picture extends well beyond pounds lost.
What comes next for oral GLP-1s
Novo Nordisk's head start with oral Wegovy may not last long. Eli Lilly's oral GLP-1 candidate orforglipron has a PDUFA goal date of April 10, 2026, and the company has indicated it expects to launch early in the second quarter of 2026. That gives Novo roughly three to four months of exclusive positioning in the oral GLP-1 weight loss market.
Orforglipron brings a different profile. In Phase III data, it achieved 12.4% average weight loss compared to 13.7% on injectable Wegovy. The weight loss is somewhat lower, but orforglipron is a small molecule rather than a peptide, which could mean simpler manufacturing, lower costs, and fewer dosing restrictions than semaglutide's SNAC-dependent absorption system.

Not every contender has survived. Pfizer halted its oral GLP-1 candidate danuglipron in April 2025 after encountering liver injury signals and tolerability problems. Converting a GLP-1 from injection to pill does not work for every molecule.
The broader pipeline is crowded. IQVIA counts roughly 193 obesity therapies in development, and industry projections suggest oral formulations could capture around 20% of the projected $80 billion obesity GLP-1 market by the end of the decade. For patients, that should translate to more options and more pricing pressure over the next few years.
Will oral GLP-1s kill off weight loss injections? Probably not. For patients who refuse needles, the pill is the difference between treatment and no treatment. For patients already comfortable with weekly injections who prefer more consistent drug exposure, the injection still makes sense. Both forms will coexist, serving different preferences within the same drug class. The real gain is that patients now get to choose.
Frequently asked questions
Is the oral Wegovy pill as effective as the Wegovy injection for weight loss?
Clinical trial data suggests comparable results. In cross-trial comparisons, oral semaglutide 25mg produced weight loss 11.4 percentage points above placebo, while subcutaneous semaglutide 2.4mg produced 12.4 percentage points above placebo. These are similar magnitudes, though direct head-to-head trial data between the two formulations at weight-management doses is limited. Both forms share the same active molecule and the same half-life of approximately one week.
Why do I have to take oral semaglutide on an empty stomach?
The absorption mechanism depends on SNAC, a compound that creates a protective buffer zone around the dissolving tablet in your stomach. Food physically blocks contact between the tablet and your stomach lining, and extra fluid dilutes the protective effect. Since less than 1% of the oral dose reaches your bloodstream even under ideal conditions, any interference with absorption can reduce the drug's effectiveness. The 30-minute fasting window gives the tablet time to dissolve and move the medication through the gastric wall.
Can I switch from Wegovy injections to the Wegovy pill?
This is a conversation for your prescribing physician. The oral and injectable forms use different dosing schedules and escalation protocols. A switch would typically involve starting the oral form at a low dose and titrating up over several months, rather than jumping directly to the 25mg target dose. Your doctor can determine the appropriate timing and transition strategy based on your current dose and response.
Does Medicare cover the oral Wegovy pill?
It depends on your specific Part D plan and why you are taking it. Medicare cannot cover medications prescribed solely for weight loss, but the cardiovascular indication for Wegovy has opened a coverage pathway for beneficiaries with established heart disease and obesity or overweight. Individual Part D plans decide whether to add Wegovy to their formularies and what prior authorization requirements to impose. Negotiated Medicare pricing is expected to take effect in 2027.
What happens if I eat within 30 minutes of taking the pill?
Eating too soon interferes with absorption. Clinical studies showed that food intake significantly reduced or eliminated measurable absorption of oral semaglutide. If this happens occasionally, it is unlikely to be dangerous, but consistently failing to follow the dosing protocol can mean the drug never reaches therapeutic levels in your bloodstream. If the fasting requirement proves unmanageable, discuss alternatives with your doctor.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.












