A $66 Billion Bet That Your Diet Should Be Yours Alone
Two people eat the same bowl of white rice. One person's blood sugar barely moves. The other's spikes to levels associated with prediabetic risk. Same food, same portion, wildly different biological responses. That gap between what a food does to you versus what it does to your neighbor is the entire reason personalized nutrition exists.
The global personalized nutrition market was valued at USD 15.35 billion in 2025 and is projected to reach USD 66.95 billion by 2034, growing at a compound annual growth rate of 18.11%. In the United States alone, the market is expected to hit USD 13.24 billion by 2032. Those numbers reflect something real: millions of people are tired of dietary advice that treats everyone identically.
The frustration is justified. Standard dietary guidelines are built on population averages, and population averages describe almost nobody. A 2026 review in the journal Nutrients put it bluntly: one-size-fits-all guidelines "frequently yield modest and heterogeneous responses at the individual level, and long-term adherence is often poor." The same review noted that up to one in three adults may meet criteria for metabolic syndrome, a cluster of conditions that standard dietary advice has failed to meaningfully dent.
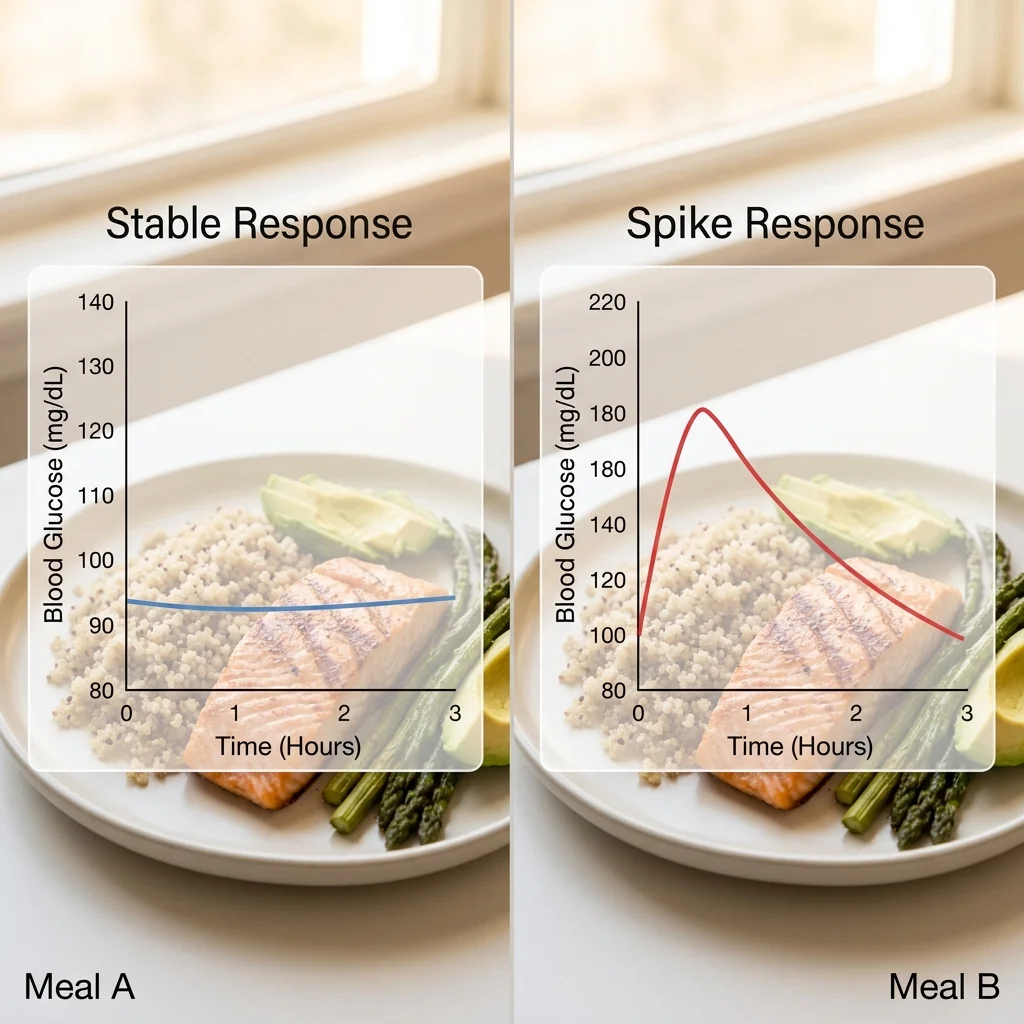
Personalized nutrition tries to close that gap by tailoring dietary recommendations to individual biology. At its most basic level, that means using some combination of genetic data, blood biomarkers, gut microbiome analysis, and continuous glucose monitoring to figure out which foods work for your specific body. At its most ambitious, it means feeding all that data into machine learning algorithms that predict how you will respond to foods you have never eaten.
But the term "personalized nutrition" covers a spectrum so wide that it borders on meaningless. A quiz-based supplement subscription and a clinically validated AI model predicting your postprandial glycemic response both claim the label. The companies selling these services range from serious research operations publishing in Nature Medicine to supplement brands that slap "personalized" on a multivitamin. Separating the science from the marketing requires looking at three distinct pillars: what your DNA actually tells you about food, what your biomarkers reveal that genes cannot, and whether AI can reliably stitch those data streams into something useful.
Your Genes Explain Less Than You Think
The pitch for DNA-based diets sounds elegant: spit in a tube, get your genome analyzed, and receive a diet plan optimized for your genetic makeup. The reality is messier. Genetics matter for nutrition, but they matter less than most direct-to-consumer companies imply, and the mechanisms are more nuanced than a simple gene-to-food mapping.
The largest and most rigorous test of this question came from the PREDICT 1 study, which enrolled 1,002 adults including 230 identical twin pairs. By studying twins, the researchers could isolate what genetics actually explains. The findings were sobering for anyone selling DNA diets: genes accounted for roughly 30% of the variation in glucose responses and just 4% of the variability in triglyceride responses. Even identical twins, sharing virtually all their DNA, often had dramatically different metabolic responses to the exact same meal.
That does not mean genes are irrelevant. Several well-characterized genetic variants have practical dietary implications. Think of your genome like the floor plan of a house: it establishes the basic layout, but the furniture arrangement, the temperature, and who lives there all change the experience of being inside.
A comprehensive 2026 review in Ecology and Evolution cataloged 15 genes with clinically meaningful nutrient interactions. Among the most actionable: FTO variants (linked to obesity risk with odds ratios of 1.3-1.7), TCF7L2 rs7903146 (which increases type 2 diabetes risk by 40-50%), and APOE ε4 (which triples cardiovascular disease risk and demands stricter saturated fat limits).
The CYP1A2 gene offers one of the clearest examples of how genetics shape nutritional needs. People with the rs762551 variant metabolize caffeine at rates that differ by as much as four-fold. Slow metabolizers who drink several cups of coffee per day face meaningfully higher cardiovascular risk, while fast metabolizers may actually see protective effects. That is the kind of specific, actionable insight DNA testing can deliver.
The MTHFR gene provides another concrete case. People with the C677T variant in the TT genotype need roughly twice the recommended daily allowance of folate (about 800 micrograms) to maintain normal homocysteine levels. Without that information, they might follow standard guidelines and still develop deficiency-related problems.
Where DNA-based nutrition runs into trouble is the jump from individual gene variants to comprehensive dietary plans. A 2024 review in Nutrients identified a core problem: "Many diseases linked to nutrition are intricate and involve multiple genes, indicating that single polymorphisms are insufficient to comprehensively elucidate the associated conditions." The same review noted that when PREDICT 1 researchers looked at known SNPs for glucose levels, those variants explained only about 20% of the estimated heritability. There is a large gap between knowing that a gene matters and knowing exactly what to eat because of it.
So what should you actually do with a DNA test result? Use it for the handful of well-validated gene-nutrient interactions (caffeine metabolism, folate needs, saturated fat sensitivity via APOE) while treating broader dietary recommendations from DNA-only services with healthy skepticism. Your genome sets certain boundaries, but your microbiome, blood biomarkers, sleep patterns, and meal timing all operate within those boundaries in ways that DNA alone cannot predict.
What Blood Tests, Stool Samples, and Glucose Monitors Actually Reveal
If DNA is the floor plan, biomarkers are the real-time weather report inside the house. They tell you what is happening in your body right now, not what your genes say might happen theoretically. That makes biomarker testing the layer where personalized nutrition starts generating genuinely individual recommendations.
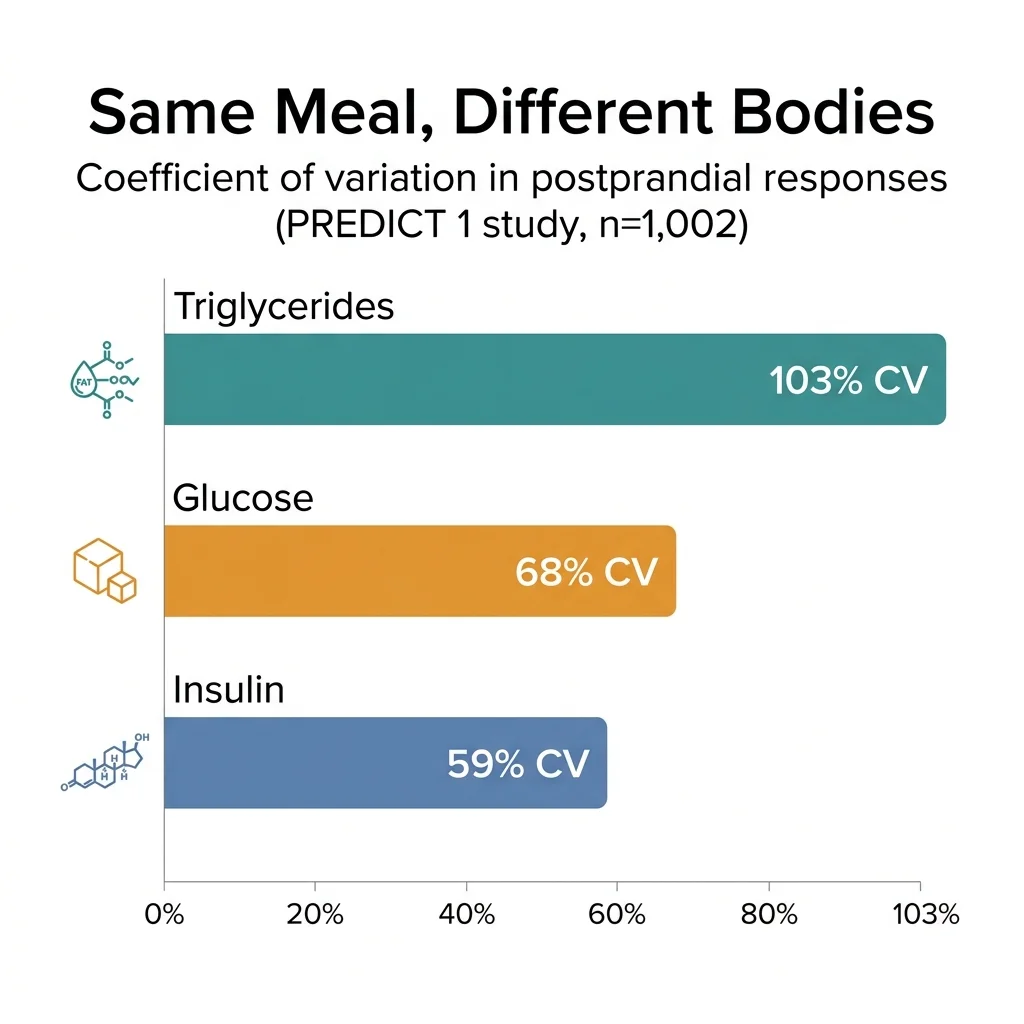
The foundational finding driving all of this came from the PREDICT 1 study's measurement of postprandial responses. When 1,002 people ate identical meals under controlled conditions, the coefficient of variation across individuals was 103% for triglycerides, 68% for glucose, and 59% for insulin. Those numbers describe people whose bodies handle the same food in completely different ways.
What was surprising was the relative importance of different factors. For blood fat responses after eating, the gut microbiome explained 7.1% of the variance while meal macronutrients explained only 3.6%. In other words, who you are mattered more than what you ate for predicting how your blood fats would behave. For blood sugar, the picture flipped: meal macronutrients accounted for 15.4% of the variation versus 6.0% for person-specific factors. That split has practical implications. It means blood sugar management can lean more heavily on food choices, while blood fat management may require understanding your individual microbiome.
Three types of biomarker testing currently dominate the consumer market:
Blood biomarker panels measure circulating markers like cholesterol fractions, inflammatory markers (CRP, IL-6), hormone levels, vitamin and mineral status, and metabolic indicators (HbA1c, fasting glucose, insulin). These provide a snapshot of current metabolic health and can identify deficiencies or imbalances that dietary changes might address. The advantage over genetic testing is directness: a blood test tells you your vitamin D level is low, not that you might be at risk for low vitamin D.
Gut microbiome testing analyzes the trillions of bacteria, viruses, fungi, and archaea living in your digestive tract. The composition of this community influences how you extract calories from food, how your immune system functions, and how you metabolize specific nutrients. A 2026 review identified the key mechanisms: short-chain fatty acid production, bile acid signaling, gut barrier integrity, and regulation of low-grade systemic inflammation. High-fiber and prebiotic dietary strategies can improve glycemic control alongside microbiome shifts, but the relationship is bidirectional: what you eat changes your microbiome, and your microbiome changes what your body does with what you eat.
Continuous glucose monitoring (CGM) uses a small sensor inserted under the skin to measure interstitial glucose levels every few minutes for days or weeks. Originally designed for diabetes management, CGMs have entered the wellness market as a way for non-diabetic people to see how specific foods, meals, exercise, and sleep affect their blood sugar in real time. The PREDICT study found that people had, on average, a two-fold higher glycemic response to the same meal eaten at lunch compared to breakfast, though individual variation was significant.
One encouraging finding from the research: your responses are consistent. The 2015 Weizmann Institute study found that within-person postprandial responses were highly reproducible, with correlation coefficients of 0.77 for both glucose and bread-with-butter meals. That means once you learn how your body responds to certain foods, that information remains useful over time, assuming your overall health status and microbiome composition stay relatively stable.
From Raw Data to Your Dinner Plate
Your DNA, blood biomarkers, microbiome data, and CGM readings generate more variables than any human dietitian can integrate simultaneously. Machine learning handles that integration, and the results from controlled trials are more credible than most wellness-tech claims, though gaps remain.
The proof of concept arrived in 2015 from the Weizmann Institute of Science in Israel. Researchers Eran Segal and Eran Elinav monitored 800 people wearing continuous glucose monitors for a full week, recording responses to 46,898 meals. They then built a machine learning algorithm that integrated blood parameters, dietary habits, body measurements, physical activity data, and gut microbiota composition. The algorithm predicted personalized glycemic responses to foods the participants had not yet eaten, and those predictions were validated in an independent 100-person cohort. A blinded randomized controlled trial using the algorithm to guide dietary choices produced significantly lower postprandial glucose responses and measurable changes in gut microbiota composition. That was a decade ago, and the technology has only gotten more sophisticated.
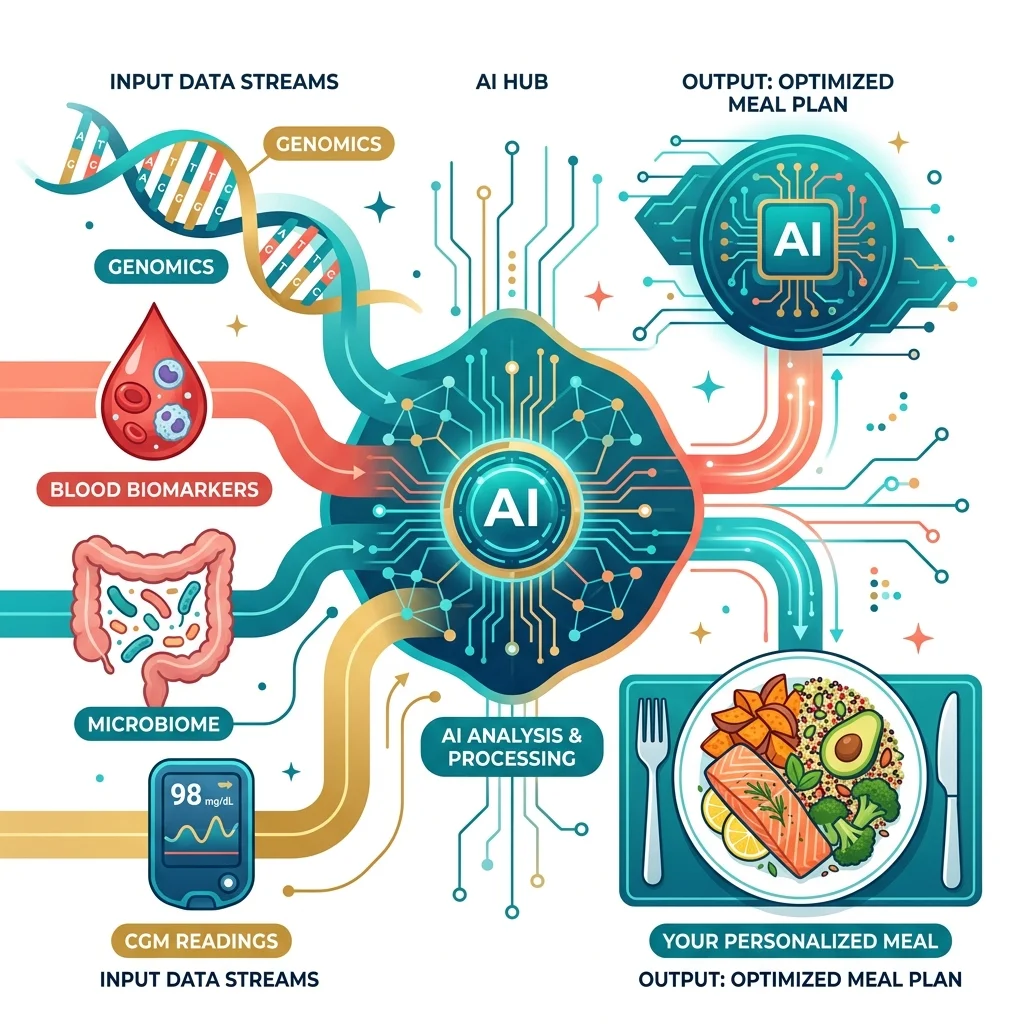
The PREDICT studies built on this foundation with substantially larger datasets. PREDICT 1's machine learning model achieved prediction accuracy of r=0.77 for glycemic responses and r=0.47 for triglyceride responses. That glycemic prediction accuracy is strong enough to be clinically useful. The triglyceride prediction is weaker, reflecting the greater complexity of fat metabolism and the larger role of hard-to-measure microbiome factors. Across the full PREDICT programme, ZOE has accumulated over 4 million blood glucose recordings, 56,000 triglyceride readings, and 12 terabytes of gut microbiome data, feeding ever-larger training datasets into their models.
The AI approaches used by these services differ meaningfully. Viome's system uses metatranscriptomic sequencing, analyzing RNA rather than DNA. The distinction matters: DNA shows which organisms are present (including dead ones), while RNA shows which organisms are alive and actively doing things. Viome pairs this with machine learning that gets more accurate as more people use the service, since each new dataset refines the model. InsideTracker takes a different approach, combining blood biomarker data with genetic information and cross-referencing results against a database of over 7,500 possible dietary and lifestyle actions.
Academic research is pushing the boundaries further. At the University of Alberta, gastroenterologist Heather Armstrong is running clinical trials at seven sites across Canada, funded by a $10 million grant from the Weston Family Foundation, for an AI-powered tool that cultures a person's gut microbes with different carbohydrate types and generates a precision diet plan. The tool was designed for inflammatory bowel disease patients but has shown results in people with other inflammatory conditions and even healthy individuals. Armstrong reported increased rates of disease remission from subtle dietary changes, with microbiome shifts visible in as little as a few weeks.
AI in personalized nutrition is not vaporware. The algorithms produce measurable improvements in metabolic outcomes when tested in controlled trials. What remains unclear is whether those improvements are large enough, compared to simpler approaches, to justify the cost for people without specific metabolic problems.
Three Different Philosophies of Personalization
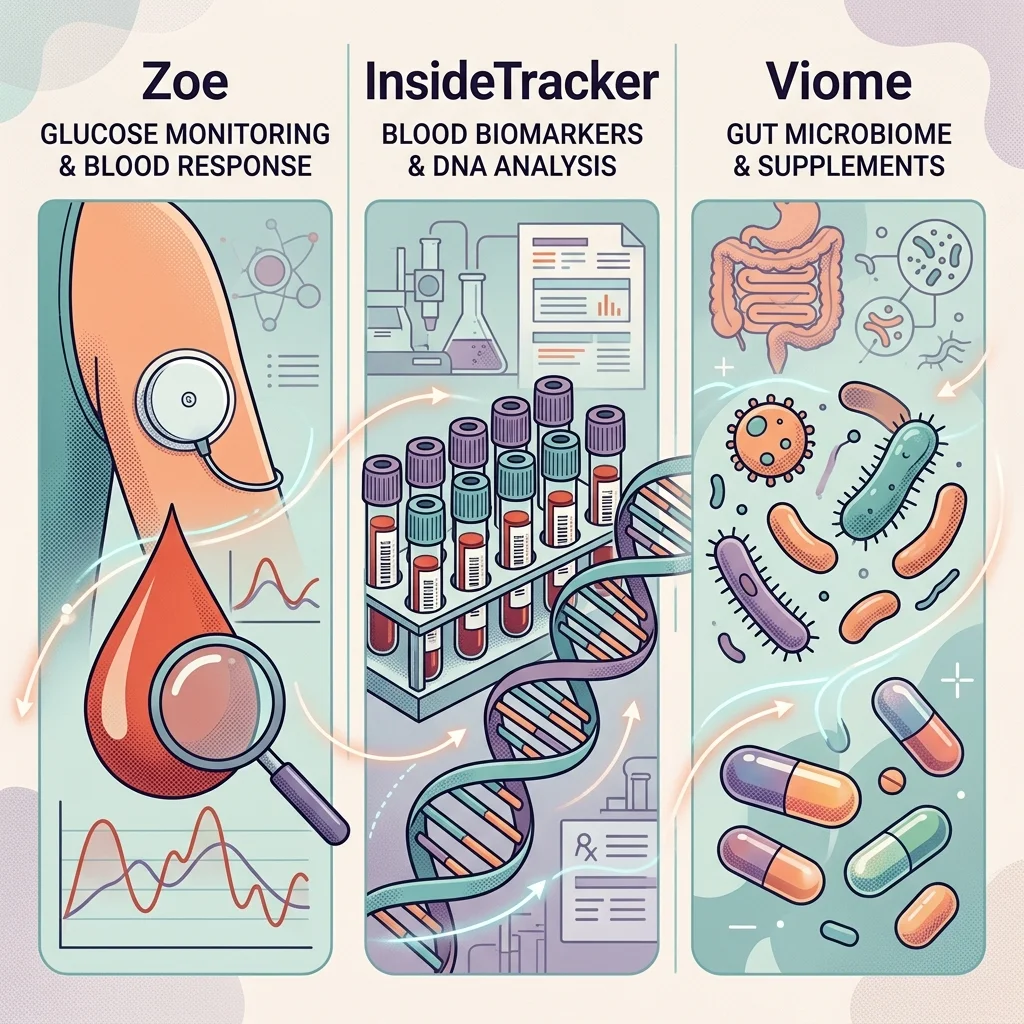
Zoe, InsideTracker, and Viome represent three genuinely different approaches to the same problem. Choosing between them is less about which is "best" and more about which matches your biology questions and budget.
| Feature | Zoe | InsideTracker | Viome |
|---|---|---|---|
| Primary data source | CGM + blood fat + microbiome | Blood biomarkers + DNA | Gut microbiome RNA + blood |
| Number of biomarkers | Glucose (CGM), triglycerides, microbiome | Up to 48 blood + 261 gene variants | Thousands of microbial RNA transcripts |
| Testing method | CGM sensor, blood spot, stool sample | Blood draw (Quest Diagnostics), cheek swab | Stool sample, finger-prick blood, saliva |
| AI/algorithm basis | PREDICT studies ML model (Nature Medicine) | 7,500+ action database cross-referenced with biomarkers | Metatranscriptomic AI (proprietary) |
| Research backing | Multiple published RCTs including Nature Medicine | Scientific Reports study (1,032 users) | FDA breakthrough device (cancer detection) |
| Custom supplements | No | No (recommends, doesn't sell) | Yes (personalized prebiotics, probiotics) |
| Retesting | Ongoing membership | Recommended quarterly ($699+ per test) | Free annual retest with subscription |
| Starting price | ~$354 (membership) | $699 (Ultimate Plan) | ~$149-$349 (depending on test) |
Zoe has the strongest published research foundation. The PREDICT programme involved scientists from Massachusetts General Hospital, King's College London, Stanford Medicine, and the Harvard T.H. Chan School of Public Health. The resulting ML model integrates meal composition, habitual diet, meal context, body measurements, genetics, microbiome, and clinical parameters. Its weakness is that the evidence base for the method is largely generated by people affiliated with the company, which creates a legitimate conflict of interest concern even when the research itself is rigorous.
InsideTracker focuses on blood biomarkers with DNA as a secondary layer. Founded in 2009 by scientists from Harvard, MIT, and Tufts, its distinguishing feature is the "optimal zone" concept: rather than just flagging whether your results fall within normal clinical ranges, it calculates tighter optimal ranges based on your age, sex, and activity level. A young female athlete gets different ferritin targets than a sedentary older man. The platform is particularly strong for athletes focused on performance optimization. The downside is cost. The Ultimate Plan runs $699 per test, and InsideTracker recommends quarterly retesting, which means annual costs can easily pass $2,000. The company also recently removed its lower-cost test options, making the service less accessible.
Viome bets heaviest on the gut microbiome. Its use of RNA rather than DNA sequencing gives it a more accurate picture of which microorganisms are actually active in your gut at the time of testing. It is also the only major player that manufactures and ships personalized probiotic, prebiotic, and supplement blends based on your results. Innerbody Research testers reported increased bowel regularity and decreased brain fog within a week, and one tester with mild GERD saw symptom improvement within two to three weeks. Viome's AI model improves as its user base grows, creating a data flywheel that competitors without custom supplement offerings cannot easily replicate.
None of these platforms is a clear winner for everyone. If you want the most research-validated approach to understanding food responses, Zoe has the edge. If you are an athlete or biohacker who wants deep blood work with actionable optimization targets, InsideTracker fits better despite the cost. If gut health is your primary concern and you want a single service that tests, analyzes, and ships you exactly what to take, Viome is the most integrated option.
The Honest State of the Science
Does personalized nutrition produce better health outcomes than standard dietary guidelines? Probably. But the margin is smaller than the marketing suggests, and it depends on what you measure.
The strongest clinical evidence comes from the ZOE METHOD trial, published in Nature Medicine. This 18-week randomized controlled trial assigned 347 adults (ages 41-70) to either a personalized dietary program or standard USDA dietary guidelines. The personalized group saw significantly greater reductions in triglycerides (mean difference of -0.13 mmol/L, P=0.016), body weight (-2.46 kg), waist circumference (-2.35 cm), and HbA1c (-0.05%). Diet quality scores improved by 7.08 points on the Healthy Eating Index.
But the same study demands honest accounting of what did not improve. LDL cholesterol changes were not significant between groups (P=0.521). Blood pressure, fasting insulin, fasting glucose, C-peptide, and apolipoproteins A1 and B all showed no difference. The personalized approach was better than standard advice on some metrics and indistinguishable on others.
| Outcome | Personalized Program | Standard USDA Advice | Difference |
|---|---|---|---|
| Triglycerides | -0.21 mmol/L | -0.07 mmol/L | Significant (P=0.016) |
| Body weight | -2.17 kg | +0.30 kg | Significant (P<0.001) |
| Waist circumference | -2.94 cm | -0.59 cm | Significant (P=0.008) |
| HbA1c | -0.02% | +0.03% | Significant |
| LDL cholesterol | -0.01 mmol/L | +0.04 mmol/L | Not significant (P=0.521) |
| Blood pressure | — | — | Not significant |
| Fasting glucose/insulin | — | — | Not significant |
The study also had important limitations. 86% of participants were female with a mean BMI of 34, placing them squarely in the obese category. Whether the results generalize to men, to people with normal weight, or to diverse ethnic populations remains untested. The 18-week duration also leaves the long-term durability of these improvements unknown.
On the genetics side, the evidence picture is both older and more fragmented. A study by Arkadianos and colleagues found that patients who received a nutrigenetic test screening 24 variants across 19 metabolism-related genes and then received tailored dietary guidance showed better long-term BMI reductions and blood glucose outcomes compared to those without genetic testing. The NOW (Nutrigenomics, Overweight/Obesity and Weight Management) trial found that nutrigenomics-guided interventions produced greater reductions in body fat percentage than standard approaches.
Critical voices raise valid points. The Gene Food team pointed out that many PREDICT study authors are affiliated with ZOE, creating a conflict of interest. They also argued that fasting lipid levels, which are more heritable than postprandial ones, are a better predictor of cardiovascular risk, and that ZOE's research "way oversells the importance of postprandial lipids in health and nutrition decisions."
The regulatory environment adds another layer of uncertainty. Personalized nutrition services are currently considered unregulated across the world. In the U.S., the FDA regulates genetic test kits sold to multiple laboratories, but laboratory-developed tests, which are more common, are not currently regulated by the FDA. Europe has no specific legal instruments for personalized nutrition. This means the quality and accuracy of what different companies deliver varies significantly, with no standardized way for consumers to verify claims.
A 2026 review of microbiome-informed personalization offered a measured conclusion: algorithms predicting postprandial glycemic responses have improved short-term glycemic outcomes compared with standard advice in controlled trials. But "clinical implementation will require standardized measurement, rigorous external validation on clinically meaningful endpoints, interpretable decision support, and equity-focused evaluation across diverse populations." The science works under controlled conditions. Whether it holds up when real people with real budgets try to follow it for years is a different question.
What does this mean if you are considering personalized nutrition right now? The approach appears to produce real but modest improvements over standard dietary guidelines, particularly for blood sugar management, weight loss, and triglyceride reduction. For LDL cholesterol, blood pressure, and fasting metabolic markers, the evidence for superiority over conventional advice is not yet there. If you have specific metabolic concerns (prediabetes, insulin resistance, elevated triglycerides), personalized nutrition is worth exploring. If you are generally healthy and eating reasonably well, the extra benefit over a good diet may not be worth the price tag.
Frequently Asked Questions
How much do personalized nutrition tests cost, and does insurance cover them?
Costs range from about $149 for a basic Viome gut microbiome test to $699 or more for InsideTracker's Ultimate blood and DNA panel. Zoe memberships start around $354. No major insurance plans currently cover these tests because they are classified as wellness services rather than medically necessary diagnostics. Annual costs can reach $2,000 or more if you follow the recommended retesting schedules, so budget accordingly.
Can a DNA test really tell me what to eat?
DNA tests can identify specific gene variants with well-validated dietary implications, like caffeine metabolism speed (CYP1A2), folate requirements (MTHFR), or saturated fat sensitivity (APOE). For those handful of interactions, the guidance is clinically meaningful. But for building a complete dietary plan, DNA alone falls short. The PREDICT 1 study found that genetics explained only about 30% of glucose response variation and 4% of triglyceride variation, leaving the majority of your food responses determined by non-genetic factors like your microbiome, sleep, and meal timing.
How accurate are gut microbiome tests?
Accuracy depends heavily on the testing method. Companies using RNA sequencing (like Viome's metatranscriptomic approach) capture a more accurate picture of active microorganisms than DNA-based methods, which can detect dead organisms that are no longer affecting your health. However, microbiome composition can shift within days based on diet changes, stress, and antibiotic use. A single test provides a snapshot, not a permanent profile. Retesting periodically gives a more reliable picture of your gut health trajectory.
Is personalized nutrition worth it if I am generally healthy?
The strongest evidence for personalized nutrition comes from people with metabolic concerns like elevated blood sugar, high triglycerides, or excess weight. The ZOE METHOD trial showed meaningful improvements in these areas compared to standard guidelines. For generally healthy people without specific metabolic issues, the marginal benefit over a well-balanced diet rich in whole foods, fiber, and vegetables is harder to quantify and may not justify the cost. Starting with standard evidence-based dietary patterns and considering personalized testing if you hit a plateau or develop specific health concerns is a reasonable approach.
How often should I retest with these services?
Blood biomarkers change relatively quickly in response to dietary and lifestyle changes, so InsideTracker recommends quarterly retesting to track progress. Gut microbiome composition can shift within weeks but tends to stabilize over months, making annual retesting reasonable for microbiome services like Viome (which includes free annual retesting with subscriptions). CGM data from services like Zoe provides real-time feedback, so ongoing monitoring is built into the membership model. The right frequency depends on whether you are actively making changes and want to measure their impact or just want a periodic health check.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.












