You hit your weekly dose, you eat the same way you have for months, the scale moved fast for a while, and now it has not moved in eight weeks. You read about a peptide stack on Reddit. Someone is selling a vial of tesamorelin online. A friend at the gym is "cycling" CJC-1295 with their semaglutide. The question hanging over all of this is not whether stacking works. It is whether the kind of stack that actually moves the needle in clinical trials looks anything like the kind of stack being sold on Instagram.
The honest answer matters because plateaus on GLP-1 medicines are normal, predictable, and partly biological — and most "stacks" being marketed online have no human trial data behind them. A few do. Telling those apart is the entire point of this guide.
Important: Stacking peptides off-label carries real risk, including hypoglycemia, accelerated lean mass loss, and theoretical cancer concerns from growth-hormone-releasing peptides. Talk to an obesity medicine specialist or endocrinologist before changing your regimen. This article is editorial, not medical advice.
Why a GLP-1 plateau is biology, not personal failure
Plateaus are the rule, not the exception. About 85% of dieters hit one, and people on GLP-1 medicines are not exempt. They just hit theirs later — typically between six and twelve months in, where lifestyle-only plateaus tend to land closer to the six-month mark. One obesity medicine physician writing for The Conversation put a clean definition on it: eight to twelve weeks of essentially no movement, while still on a stable dose, is a plateau.
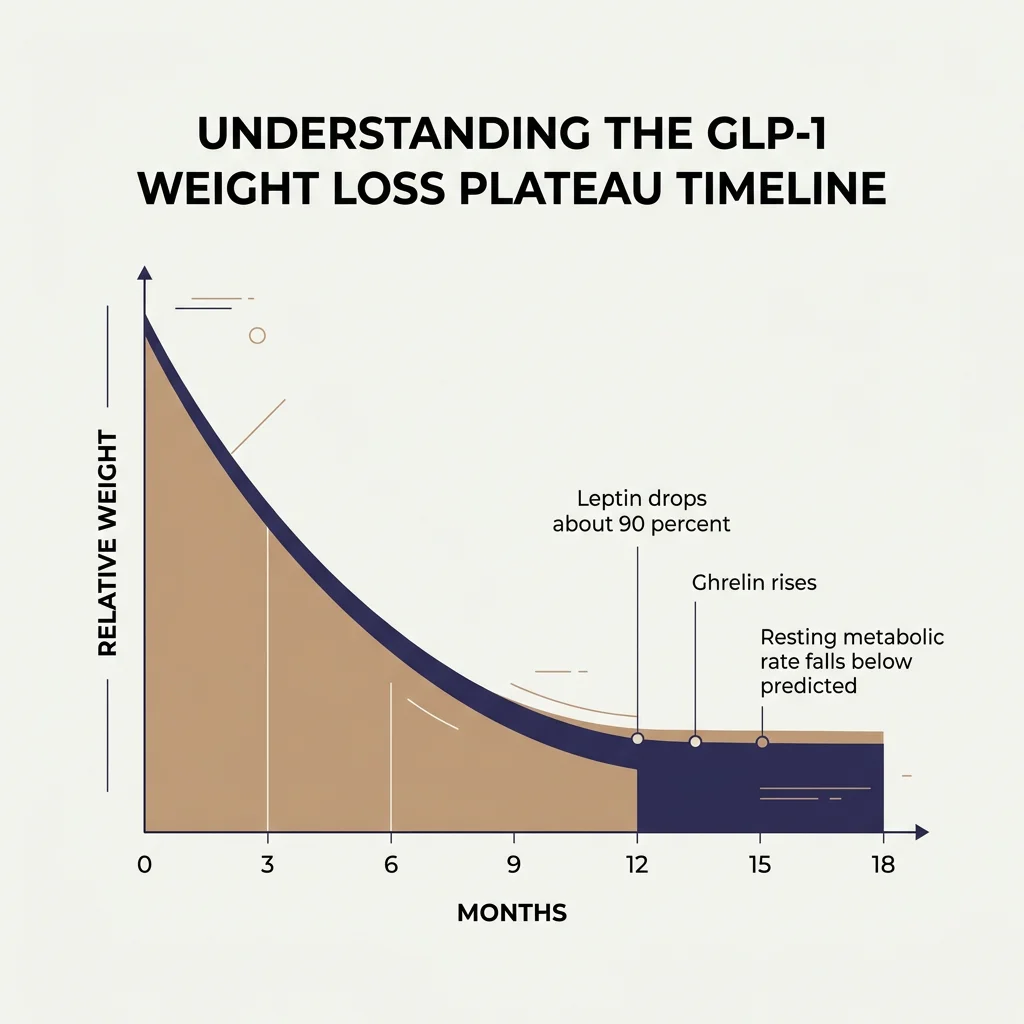
The reason is not mystery. The body defends a previous body weight. Kevin Hall's work at the National Institutes of Health is the clearest demonstration of how stubborn that defense is. Hall and colleagues followed sixteen Biggest Loser contestants for six years after they ended the show. The participants regained, on average, 41 kilograms of the weight they had lost. But the surprise was what their resting metabolism did. Six years out, their metabolic rates were still running about 499 kilocalories per day below what their body composition predicted.
The deeper finding from that study is the one that should change how anyone thinks about plateaus. Contestants who maintained the most weight loss six years later were the ones with the deepest ongoing metabolic slowing. The correlation went the wrong way for tidy stories about willpower. Sustained weight loss meant sustained adaptation. Hall calls this the constrained model of human energy expenditure. The body works hard to keep total energy balance steady. If you reduce intake, it reduces output. If you exercise more, it slows metabolism. There is no clean lever.
Hormones do the wiring. After weight loss, leptin, the satiety signal, falls fast. In the Biggest Loser cohort, leptin dropped from about 41 nanograms per milliliter at baseline to under 3 at the end of the competition. Six years later, even after substantial weight regain, leptin had only partially recovered to about 28. Ghrelin, the hunger signal, climbs. Neuropeptide Y, a potent appetite stimulator, goes up too. The brain thinks the body is starving. It is not subtle.
What this means for someone stalled on Ozempic or Mounjaro: a plateau is not a sign that the medication has stopped working in some pharmacologic sense. It is a sign that the medication has done its job, the body has adapted, and the next move depends on what is actually causing the adaptation. That is what Step 1 is for.
Step 1 before any stack: rule out the four fixable causes
Before considering anything you would buy on the internet, an honest plateau workup goes through four fixable causes. Three of them are common. The fourth is uncomfortable.
Under-titrated dose. Many people sit at a maintenance dose that was never optimized. If you are on semaglutide 1.0 mg or tirzepatide 5.0 mg and your plateau started months before you reached the higher therapeutic doses, you are not at the medication's ceiling. Your prescriber has a real lever here.
Adherence and product quality. Real-world adherence to GLP-1s is brutally low. A 2026 UT Southwestern analysis of insurance claims from nearly 127,000 U.S. adults found that only one in four were still on any GLP-1 a year after starting. A joint advisory in the American Journal of Clinical Nutrition put the trial-to-real-world gap in starker terms: trial adherence sits at 83 to 88% at about 68 weeks, but in real-world settings it falls to 33-50% at one year and only 15% at two years. Missed doses, paused doses, and inconsistent injection days quietly erase gains. So does compounded product. Compounded GLP-1s are not all the same, and quality varies in ways that affect outcomes.
Underlying medical drift. Hypothyroidism, perimenopause, new SSRIs, beta-blockers, and steroid courses can all blunt weight loss. Dr. Chika Anekwe of the Massachusetts General Hospital Weight Center, writing for The Conversation, lists these as the standard plateau workup before assuming the medication has stopped working.
Calorie creep. The most uncomfortable cause. GLP-1s reduce hunger powerfully at first. As the body adapts, hunger returns partially, and intake quietly creeps back up — often through liquid calories, snacks, and alcohol that bypass the satiety signal entirely. Most people do not notice this without measuring.
If even one of these four is in play, no peptide stack is going to outperform fixing it. This is the boring part nobody wants to read. It is also where the real gains live.

Stack #1: The clinical-evidence path — switching, escalating, or trial-enrolling
The first "stack" worth considering is not a stack at all. It is the set of moves that obesity medicine specialists actually use, in roughly the order they use them.
The most decisive of these is switching molecules. The SURMOUNT-5 phase 3b trial, led by Louis J. Aronne, MD, and published in the New England Journal of Medicine, randomized 751 adults with obesity to either tirzepatide or semaglutide at maximum tolerated doses. At 72 weeks, tirzepatide produced 20.2% body weight loss compared with 13.7% on semaglutide — an average of 22.8 kilograms versus 15.0. The same trial showed a smaller GI burden on tirzepatide: 2.7% discontinued for gastrointestinal reasons versus 5.6% on semaglutide. Tirzepatide adds GIP receptor activity to GLP-1, and that mechanism appears to matter for both efficacy and tolerability.
Switching is not failure. UT Southwestern's analysis of 127,000 patients showed that those who switched between GLP-1 medications had higher long-term adherence than those who stayed on their first drug. Senior author Sarah Messiah's framing is worth keeping: switching should be viewed as a normal part of long-term obesity care, not a sign of failing treatment.
The other path inside Stack #1 is enrollment in a clinical trial. Both CagriSema and retatrutide phase 3 programs are still active across multiple indications. Trial enrollment is the only legal route to stacking-style efficacy with safety monitoring attached.
Stack #2: GLP-1 + amylin — what CagriSema actually showed
The most studied "stack" is not a stack you assemble. It is one molecule. CagriSema combines cagrilintide, a long-acting amylin analog, with semaglutide in a single weekly injection. Amylin is a hormone co-secreted with insulin that slows gastric emptying and acts on hypothalamic appetite pathways. Pairing it with GLP-1 hits two complementary brakes.
The REDEFINE-1 trial, led by W. Timothy Garvey, MD, published in the New England Journal of Medicine, randomized 3,417 adults without type 2 diabetes. At 68 weeks, CagriSema produced 20.4% weight loss compared with 3.0% on placebo, a 17.3 percentage point difference. REDEFINE-2, led by Melanie J. Davies, MD, ran in 1,206 adults with type 2 diabetes. CagriSema produced 13.7% weight loss versus 3.4% on placebo, and 74% of CagriSema patients reached an HbA1c at or below 6.5%, compared with 15.9% on placebo. Garvey's framing for why two molecules outperform one: complementary action on appetite regulation in brain regions handling both hedonic and homeostatic eating. Different brakes, different roads.
Then came the head-to-head reality check. REDEFINE-4 pitted CagriSema directly against tirzepatide. Eight hundred nine adults with obesity, 84 weeks of treatment. CagriSema achieved 23.0% weight loss. Tirzepatide 15 mg achieved 25.5%. CagriSema failed its non-inferiority primary endpoint. The combination is real. It is just not, on average, better than maxed-out tirzepatide.
For someone stalled on semaglutide, the practical implication is twofold. CagriSema is real and FDA review is in progress, with a decision anticipated by late 2026. And until it is approved, it is not something you can build yourself. DIY pairing of a GLP-1 with separately sourced cagrilintide does not exist as a legal off-label option, and the clinical trial dosing was tightly co-titrated.
Stack #3: The triple-receptor pathway — what TRIUMPH-4 hints at
Eli Lilly's retatrutide is the second case where "stack" is misleading. Retatrutide is a single molecule that activates three receptors at once: GIP, GLP-1, and glucagon. The glucagon arm matters because, despite the name, glucagon receptor activation increases energy expenditure rather than reducing it.

TRIUMPH-4 reported its results in late 2025. In an obesity-and-knee-osteoarthritis cohort, patients on the 12 mg dose lost 28.7% of their body weight at 68 weeks, a placebo-adjusted 26.6% reduction. That is the largest weight loss reported in any phase 3 GLP-1-class trial to date. But the trial also surfaced a new safety signal that did not show up in phase 2: dysesthesia, an abnormal sense of touch where ordinary sensations feel unusual or painful. It hit 20.9% of patients on the 12 mg dose and 8.8% on 9 mg, compared with 0.7% on placebo. Lilly reported that dysesthesia did not lead to discontinuation, but signals like this matter for long-term tolerability.
The standard GI burden was also higher: nausea 43%, vomiting 21%, diarrhea 33%. Treatment discontinuation reached 18.2% on 12 mg compared with 4% on placebo. Some of those discontinuations were attributed to perceived excessive weight loss rather than side effect intolerance. The TRIUMPH program has enrolled 5,800 participants since 2023.
For someone on a plateau, the practical takeaway is the same as with CagriSema: this is a regulated, single-molecule approach in late-stage development, not a do-it-yourself protocol. Combining a GLP-1 with separately sourced glucagon analogs or growth hormone analogs in an attempt to replicate retatrutide's effect is not equivalent and is not safe.
Stack #4: GLP-1 + tesamorelin and other GHRH analogs
This is where the wellness market lives, and where almost none of the human evidence does. Tesamorelin is a growth-hormone-releasing hormone analog. The FDA approved it for one narrow indication: central adiposity in people living with HIV, also called HIV-associated lipodystrophy. The trials were run in HIV cohorts that were 87% male and 83% Caucasian, on stable antiretroviral therapy. Approximately 70% of those patients responded with at least an 8% reduction in visceral fat. The other 30% did not respond. Tesamorelin's effect on insulin and glucose homeostasis was neutral.
None of those trials were run in people with obesity who were stalled on a GLP-1. The off-label use being marketed online (adding tesamorelin to semaglutide or tirzepatide to "break a plateau" or "preserve muscle") is not what tesamorelin was approved for, not what it was tested in, and not what the trial population looked like.
Eric Topol, director of the Scripps Research Translational Institute, summarized the landscape with unusual directness in a July 2025 critical review: "There is no evidence from randomized trials in humans that any of these peptides provide the benefits that are advocated." He was talking about the broader category: sermorelin, CJC-1295, ipamorelin, BPC-157, AOD-9604, MOTS-c. Sermorelin, frequently prescribed by wellness clinics, was FDA approved in 1997 and withdrawn in 2008. It has no current FDA approval. Topol also raised the theoretical cancer concern that applies to every growth-hormone-axis peptide: by inducing broad cellular growth, they could plausibly accelerate dormant tumor growth, the way TB-500 was shown to do in animal studies.
The CNN piece on the peptide craze captured the same medical consensus through a different door. Dr. Elan Goldwaser, a sports medicine physician at Columbia University Vagelos College of Physicians and Surgeons, told the network that most of the available research stems from laboratory studies and limited animal studies. Dr. Panagis Galiatsatos at Johns Hopkins was more measured: peptides are tools, but they need supervision, an end goal, and a way to monitor adverse effects. None of which is happening when someone is stacking ingredients sourced from a research-chemicals website.
If "stack #4" sounds dismissive, it is meant to. There is no human trial showing that GLP-1 + tesamorelin or GLP-1 + CJC-1295 produces better weight loss, better body composition, or fewer side effects than dose-optimizing a GLP-1 alone. Until that trial exists, the upside is hypothesis and the downside is real.
Stack #5: The lifestyle stack — boring, evidence-based, and actually works
Saving the actual evidence-based stack for last is deliberate. It is the one almost nobody wants to hear about.
The numbers from the joint advisory in the American Journal of Clinical Nutrition are uncomfortable. Roughly 38% of weight lost on GLP-1s in trials is lean body mass, not fat. Without resistance training, modeling work suggests men lose 20-25% of their total weight loss as muscle and women lose 10-15%. That is not a minor cosmetic problem. Lean mass loss is what drives metabolic adaptation. Less muscle means a lower resting metabolic rate, which means a deeper plateau, which means easier regain when the medication ends or fades.
The fix is not exotic. The advisory recommends 1.2 to 1.6 grams of protein per kilogram body weight per day during active weight loss, with 80 to 120 grams as a practical absolute target. StatPearls lands in a similar range at 1.2 to 1.5 grams per kilogram. On a GLP-1, hitting this almost always requires deliberate planning, because reduced appetite collides with the higher-protein target. Protein-forward smoothies, cottage cheese, eggs, and lean meat at every meal are the workhorses.
Resistance training is the other half. A randomized trial cited in the advisory found that one year of combined GLP-1 therapy with structured exercise training preserved bone mineral density, while GLP-1 therapy alone reduced it. That is a quietly significant finding. It means the medication is not just losing fat from your hips; it is also costing bone, and the cost is offset by training, not by the drug.
The evidence for combining lifestyle work with GLP-1s is also harder than the wellness market admits. The STEP 3 trial paired semaglutide with intensive lifestyle intervention: 30 counseling visits, structured nutrition, physical activity, plus eight weeks of meal replacements. Result: 16% weight loss, versus 5.7% with the lifestyle work alone. SURMOUNT 3 used a staged design (12 weeks of intensive lifestyle intervention before starting tirzepatide) and produced 25% weight loss, the largest reduction reported in any GLP-1 trial. The pattern is consistent. The medication does more when paired with meaningful lifestyle work, and the lifestyle work does more when paired with the medication. Neither replaces the other.
Sleep matters too. Poor sleep raises ghrelin, lowers leptin, and elevates cortisol. That is exactly the hormonal signature that makes a plateau worse. The National Weight Control Registry, established in 1993, has consistently shown that physical activity is one of the strongest predictors of long-term maintenance among people who keep significant weight off.
The practical version of stack #5 is: protein at every meal, resistance training three times a week, 150-200 minutes of aerobic activity, and seven hours of sleep. That is the unsexy stack with the strongest evidence base of anything in this article.

When to stop fighting the plateau and accept maintenance
Some plateaus are not problems to solve. They are end states.
The body's defense of a previous body weight does not vanish at 12% loss. It does not vanish at 20%. The mean weight loss in the Biggest Loser cohort six years out was 11.9%, and 57% of the participants had still maintained at least a 10% loss. By comparison, the Look AHEAD trial, the largest intensive lifestyle intervention ever run, saw 27% of participants maintain a 10% loss at 8 years. So 11.9% sustained from a starting BMI of nearly 50 is actually a strong outcome.
If you hit a plateau on a GLP-1 after losing 15-20% of your starting weight, you are at or near the trial-average outcome. The decision is not how to get to the next 5%. The decision is whether the weight you have lost is worth the dose, side effects, and cost of staying on the medication indefinitely. STEP 4 showed that within 48 weeks of stopping semaglutide, participants regained approximately two-thirds of their prior weight loss. That is biology talking, not failure. Long-term GLP-1 therapy is now treated, in most clinical guidance, as ongoing chronic-disease management, not a finite course.

Red flags: the stacks not to touch
A short, blunt list. None of the following should be added to a GLP-1 regimen on the basis of internet protocols:
- BPC-157. The FDA's 2023 statement was explicit: "the agency lacks sufficient information to know whether the drug would cause harm when administered to humans." Not an endorsement. A warning.
- AOD-9604. Marketed as a "fat-burning fragment." No robust human RCT data for weight loss. Repeatedly investigated; never approved.
- DNP (2,4-dinitrophenol). Causes hyperthermia and death. Has killed multiple users in the past decade.
- T3 / thyroid hormone for non-hypothyroid plateau. Causes muscle loss, atrial fibrillation, bone loss. Not a plateau strategy. A medical risk.
- MOTS-c, 5-amino-1MQ, NAD+ injections marketed for weight loss. No human RCT data showing weight loss benefit.
- Anything sold as "research chemicals." A peptide-testing startup in Texas, Finnrick Analytics, has run nearly 9,900 quality tests since late 2024. Twenty-two percent failed. Twelve percent had labeled doses off by more than 20%. Even when the molecule is what it says it is, dosing is often not.
The pattern across the red-flag list is the same one Topol named: animal data, anecdote, social media, and a marketplace built on the absence of FDA oversight. The FDA classifies many of these peptides as Category 2 substances — meaning they pose potential significant safety risks and cannot legally be compounded for human use.
Frequently Asked Questions
How long should I wait before deciding I've truly hit a plateau on Ozempic?
Most obesity medicine specialists define a true plateau as eight to twelve weeks of essentially no weight movement on a stable, properly used dose. Less than that is normal week-to-week variation, especially around hormonal cycles, hydration shifts, or travel. If the scale has been flat for two months and your dose, adherence, and product source are all confirmed, that is the point at which a workup makes sense.
Should I switch from semaglutide to tirzepatide if I plateau?
It's a reasonable conversation with your prescriber, not an automatic move. The SURMOUNT-5 trial showed tirzepatide produced about 6.5 percentage points more weight loss than semaglutide on average over 72 weeks, with somewhat lower GI dropout. But your specific tolerance, insurance coverage, and the dose you're actually on matter. If you're not yet at therapeutic-target dose on semaglutide, dose escalation may achieve more than switching.
Is CagriSema available now? Can I get it through my doctor?
Not yet in the United States. Novo Nordisk submitted CagriSema to the FDA in December 2025, with a decision anticipated by late 2026. Outside of clinical trial enrollment, there is no legitimate route to access it before approval. DIY combinations of separately sourced semaglutide and cagrilintide are not equivalent to the co-formulated trial product and carry meaningful risks, including unpredictable dosing.
What about adding tesamorelin to my GLP-1 to lose belly fat?
There are no human trials testing tesamorelin alongside GLP-1s for weight loss in people without HIV. The trial data for tesamorelin come from people living with HIV-associated lipodystrophy, and 30% of those patients did not respond. Growth-hormone-axis peptides also carry theoretical cancer concerns flagged by independent researchers including Eric Topol. This is not a stack with evidence behind it.
What's actually the highest-yield change for someone stalled on a GLP-1?
Three things, in order of evidence quality. First, audit dose, adherence, and product source. Second, add structured resistance training and protein at 1.2 to 1.6 grams per kilogram body weight per day. Third, talk to your prescriber about switching molecules or escalating dose if you're not at target. Everything beyond that (DIY peptide stacks, exotic supplements, extreme caloric restriction) has either thinner evidence or worse risk profiles.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.